1. Background
Polychlorinated biphenyls (PCBs) are man-made chemicals, characterized by the replacement of hydrogen atoms by chlorine atoms in two interlocked benzene rings. Theoretically, there are 209 different congeners of PCBs (1). PCBs are very stable chemically and show good thermal conductivity; besides, they are electrically insulating. These features make them very useful for production of the liquid components of transformers and capacitors (2).
The PCBs enter the environment through various forms such as leakage from old electrical equipment in use; and are easily absorbed by sediments and soils (3). Due to their low solubility and high resistance to chemical reactions, they have a relatively long shelf-life in the environment (4). In the 1970’s, the use of PCBs was banned worldwide. However, since these organic contaminants are highly persistent in the soil, and their presence poses a risk of toxicity and diseases in humans, they must be refined or removed from the environment. To remove PCBs from contaminated soils, various technologies, such as solvent extraction, soil washing, biodegradation and etc., are used (5). One of the biodegradation methods is the biological vermicomposting process that was used in the present study.
Biological methods are usually used for removing low concentrations of pollutants (6). Zhang et al. (7) evaluated PCBs at a concentration of 1 mg/kg and reported an elimination efficiency of 43.8%. Lu et al. (8) investigated the effect of earthworms on low concentrations of PCBs. He et al. (9) also investigated the effect of earthworms on 500 μg/kg PCB congeners. The novelty of our method is that earthworms were added to a mixture of contaminated soil and biological sludge to eliminate higher concentrations of PCBs compared to previous methods (10). Ekperusi and Aigbodion (11) and Abdollahinejad et al. (12) examined the removal of petroleum hydrocarbons from contaminated soil using the vermicomposting process. Also the vermicomposting method, in which biological sewage sludge is used to feed worms and microorganisms, has not been studied to refine soil contaminated with power plant Ascarel oil, and can be considered as an important and innovative step. Also, Earthworms can remove heavy metals from sewage sludge and prevent entering the environment by absorbing them in their bodies (13) which is a positive environmental advantage on its own. The removal of PCBs by bioremediation is a low-cost and environmentally friendly method (14), in which earthworms can increase the dispersion of bacteria and improve the performance of microorganisms destroying PCBs (8). Our method can be applied for large amounts of contaminated soil in situ (15), which is not possible in other methods.
2. Objectives
During power plant operations in the electric power industry, a variety of wastes, including large amounts of soils contaminated with PCBs, are generated, causing severe environmental pollution. In this study, to decontaminate and treat PCB-contaminated soils in the electric power industry, the effect of biological sludge on contaminated soils in the removal of PCBs was investigated in a vermicomposting process at different mix ratios.
3. Methods
For the experiments, soil from the ground surface (0 - 20 cm) was used. After drying, the soil passed through a 2-mm sieve to separate large particles and obtain a uniform soil texture. The measurements taken from the soil did not indicate any PCBs. Next, Aroclor 1260 oil was dissolved in n-hexane and mixed with soil to simulate contaminated soil conditions; the oil was provided by a company responsible for the maintenance of distribution transformers in north of Iran. Besides, earthworms were purchased from Sharifabad Agro-Industry Company (Iran). Biological sludge was also obtained from the wastewater treatment plant of the same power plant. After drying, it passed through a 2-mm sieve and was used to feed the worms.
To degrade PCBs in contaminated soil, mixtures of PCB-contaminated soil and biological sludge at different mix ratios (ratios of 1:1, 1:2, 1:3, and 1:4) were prepared in sample containers. For accuracy, each mix ratio was prepared in three containers; the final result for each mix ratio was the average value of these three containers. After mixing, each mixture was used in the specified pilot. The specifications of our study pilots are presented in Tables 1 and 2.
| Specifications | A | B | C | D |
|---|---|---|---|---|
| Percentage of contaminated soil (%) | 50 | 33 | 25 | 20 |
| Amount of biological sludge (%) | 50 | 67 | 75 | 80 |
| PCBs (mg/kg) | 45 | 29.7 | 22.5 | 18 |
| C/N ratio | 25.4 | 26.6 | 24.8 | 27.3 |
| pH | 8.6 | 8.4 | 8.1 | 7.9 |
| Moisture (%) | 65 | 65 | 65 | 65 |
| Number of containers per pilot | 3 | 3 | 3 | 3 |
The Initial Specifications of Each Pilot
| Characteristics | Values |
|---|---|
| PCBs (mg/kg) | 0 |
| C/N ratio | 5.1 |
| pH | 6.5 |
| Moisture (%) | 61% |
The Basic Characteristics of Biological Sludge
A total of 1000 Eisenia fetida worms (approximately 1 kg) were distributed in each container. The sample containers were covered by a mesh plate to prevent the worms from escaping. After the initial mixing and adding the worms, the vermicomposting process was initiated and continued for 80 days to expose the content of each container to microbial degradation. All treatments were evaluated simultaneously, each in three replicates. The reactors were prepared aerobically in rectangular wooden boxes with an inner polyethylene coating (L = 100 cm, W = 50 cm, H = 30 cm). To provide oxygen and create aerobic conditions, aeration of masses was done by hand stirring, alternating three times a week. The vermicomposting process was performed at normal pH of the mass and ambient temperature. The moisture content of the mass was also adjusted to 65%, and if reduced, it was adjusted with distilled water.
The samples were tested on days 0, 20, 40, 60, and 80. To extract PCBs from the soil, the frozen sample was poured into a mortar and crushed; then, it passed through a 250-μm sieve and added to a thimble. Next, 1 mL of an internal standard was added. Extraction was performed for eight hours with a mixture of extraction solvents. To prepare the blank, a Soxhlet procedure was performed; the yielded Soxhlet extract was concentrated. Next, it was dried with sodium sulfate anhydrous. For separation, a Florisil column was used. The sample was mixed with sodium sulfate and added to a burette; finally, a fraction exited the column. At this point, the fraction obtained from the cleanup step was concentrated and injected into the instrument (16).
The samples were sent to laboratory for measuring the PCBs, fixed solids percentage, volatile solid content, nitrogen content, C/N ratio and pH. The number of E. fetida earthworms was measured manually. Temperature was also measured daily. Standard methods were used to measure different parameters, such as temperature, pH, organic carbon content, and nitrogen content. The PCB content was measured by the GC/ECD method (17) with a capillary column. A gas chromatograph (Agilent, USA), equipped with an ECD and HP-5 capillary column (60 m × 0.25 mm × 0.25 μm), was used for the measurements.
4. Results and Discussion
According to Table 3, the proportion of PCBs in all pilots (A, B, C, and D) decreased. However, the highest reduction of PCBs occurred in pilot D (1:4 volume ratio of contaminated soil to biological sludge) and more than a 47% reduction was observed in the total PCBs. Zhang et al. (7) revealed that in their study on PCBs at a concentration of 1 mg/kg, the degradation rate was 43.8%; whereas the present study was performed at higher concentrations of PCBs and indicated a higher rate of PCB removal. In pilots A and B, due to the high ratios of contaminated soil to biological sludge, the reduction of PCBs was not significant. However, in pilot C, due to the addition of more biological sludge in the presence of E. fetida worms and microorganisms, the amount of PCBs reduced more than pilots A and B. To remove PCBs from contaminated soil by the vermicomposting process, parameters, such as pH, volatile solids and fixed solids, were also considered effective and studied.
| Pilot | Day 0 | Day 20 | Day 40 | Day 60 | Day 80 | Rate of Reduction (%) |
|---|---|---|---|---|---|---|
| A | 45 | 40.4 | 36.7 | 32.3 | 29.5 | 34.3 |
| B | 29.7 | 26.6 | 23.5 | 21.1 | 18.9 | 36.1 |
| C | 22.5 | 20.2 | 17.9 | 15.5 | 12.9 | 42.9 |
| D | 18 | 16.2 | 13.8 | 11.9 | 9.5 | 47.4 |
The PCB Changes in the Pilots (Experimental Conditions: Ambient Temperature, 19°C; Moisture, 65%; Pressure, 1 atm; Season, Winter; Location, Mazandaran Province).a
4.1. Trend of pH Changes in the Pilots
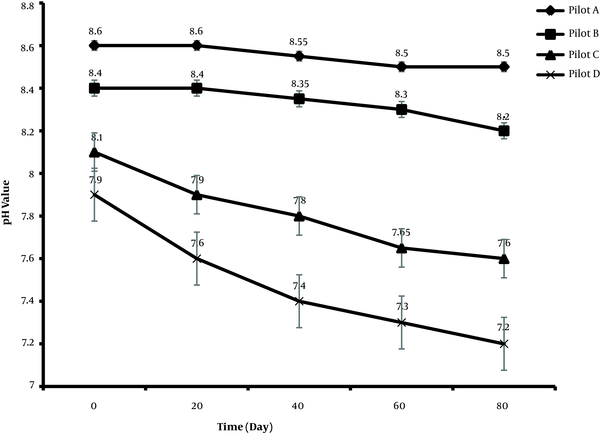
Soil earthworms and microorganisms can change the soil pH. The ability of microorganisms to access nutrients is also affected by pH. The optimal pH for the vermicomposting process is between 5.5 and 8. The pH is usually reduced during this process due to the activity of acid-forming bacteria and biodegradation of raw materials to intermediates, including organic acids (18). Figure 1 presents the trend of pH changes in all treatments.
High pH at the beginning of degradation in pilots A, B, C, and D, was related to the nature of waste (soil contaminated with transformer oil) and degraded over time as the microorganisms grew active. The outcome of degradation reaction under aerobic conditions (even a possible anaerobic process in the lower part of the mass) was the production of CO2 (19), which could significantly reduce pH during degradation as the most important weak acid. However, the pH change was not significant in pilots A and B, showing less biological activity. But in pilots C and D, as compared to pilots A and B, due to the active presence of bacteria, microorganisms, and worms, the rate of pH changes was higher. In other words, as the growth rate of earthworms and the activity of microorganisms increased in a sample, the content of volatile solids decreased further, and the organic nitrogen compounds were partly converted to ammonia. Ammonia was then removed by submerging and mixing the vermicomposting mass, and the pH was slightly reduced. In other studies, the pH in the vermicomposting process showed acidic conditions, compared to the initial pH (20). According to Figure 1, in pilots C and D, where the activity of worms was higher, the pH reached a neutral level, while in pilots A and B, it remained in the range of 8.2 - 8.5.
4.2. Trend of Changes in the Volatile Solid Content in the Pilots
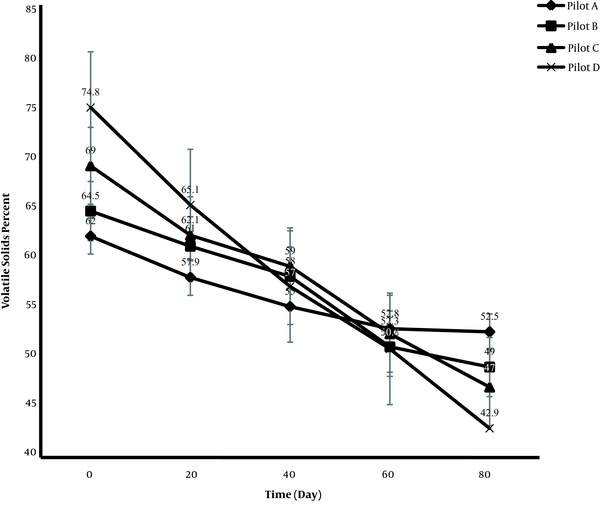
According to the Figure 2, degradation of organic matter in the mixture of biological sludge and contaminated soil by microorganisms was accompanied by a decrease in the volatile solid content in the pilots. As shown, the greatest reduction in the volatile organic matter occurred in pilot D. In three pilots with lower amounts of biological sludge in the mixture (i.e., pilots A, B, and C), the rate of reduction in the organic matter was lower, which was related to very low biodegradation by microorganisms in the contaminated soil. Therefore, the reduction of volatile solids in pilot D was more than other pilots due to the higher activity of bacteria, microorganisms, and worms in the biological sludge, which in turn increased the rate of degradation and removal of organic matter. Because microorganisms use carbon as a source of energy, the increased growth of microorganisms and mobility of worms led to the further degradation of organic solids in the mass (21). Based on the analysis of the degradation process during 80 days, changes in the percentage of volatile solids in the pilots, especially pilot D, showed a decreasing trend. Overall, measurement of changes in volatile solids is important, as it represents the process of degradation and removal of PCBs.
4.3. Trend of Changes in Fixed Solids in the Pilots
Fixed solids are the inorganic part of sludge. During vermicomposting process, the organic matter was partly converted to minerals; therefore, in samples with more biological activity, the percentage of fixed solids increased; these changes are shown in Table 4. Other factors include C/N ratio, temperature, and moisture, as described below.
| Pilot | Day 0 | Day 20 | Day 40 | Day 60 | Day 80 |
|---|---|---|---|---|---|
| A | 41.5 | 45 | 46.5 | 48 | 49.7 |
| B | 36.8 | 39.7 | 42.2 | 48.8 | 51 |
| C | 28.1 | 35.9 | 40.1 | 48 | 56 |
| D | 24.9 | 35.2 | 43.4 | 48.4 | 59.5 |
The Trend of Changes in the Fixed Solid Percent in the Pilots (Experimental Conditions: Ambient Temperature, 19°C; Moisture, 65%; Pressure, 1 atm; Season, Winter; Location, Mazandaran Province).a
4.3.1. C/N Ratio
An appropriate C/N ratio is essential for the optimal nutrition of earthworms. The ideal C/N ratio of containers for crude vermicomposting should be about 25 - 30 (22) that was adjusted in our study by using straw. During the process, the C/N ratio decreased in all pilots, although it decreased further in pilot D; because of the presence of a higher percentage of biological sewage sludge.
4.3.2. Temperature
An optimal temperature between 15 and 25°C is suitable for the growth of E. fetida (23). Therefore, the temperature of the room was kept at 19°C using heating systems. During the process, the temperature increased in all pilots, although the highest temperature increase occurred in pilot D; due to that there was a higher percentage of biological sewage sludge, which led to the increased growth and number of worms and resulted in further biological activity.
4.3.3. Moisture
In the vermicomposting process, low moisture leads to dehydration and decelerates the biological process. Also, high moisture, with obstruction of pores, causes disturbance in aeration and leads to the predominance of anaerobic conditions (24). Therefore, as mentioned earlier, the moisture content of masses was increased to 65% and adjusted with distilled water if reduced.
5. Conclusions
Based on the comparison of our results with similar research (7), it was found that the method used in our study was very efficient and yielded better results. Therefore, the mixture of contaminated soil with biological sludge has a great potential for degradation of toxic substances, including PCBs, due to the presence of some microorganisms, especially heterotrophic bacteria. The earthworm activity maintained the aerobic conditions and increased the microbial activity. In pilots with a higher biological sewage sludge content, the amount of microorganisms was higher, which indicated a higher biological activity, resulting in the further reduction of pH. The greatest decrease in the PCBs and volatile solids also occurred in the same pilot, indicating the greatest decrease in pH (Pilot D). The reason for this decrease, similar to previously mentioned parameters, was the higher microbial activity and growth, which increased the rate of degradation and removal of organic matter.