The great Khorasan province, made up of three provinces, is located in the neighborhood of Afghanistan and Pakistan, two of countries that are among 22 TB high-burden countries; according to WHO report (2015), the TB prevalence rate in Afghanistan and Pakistan was 380 per 100,000 population in 2014 (
2). Also, high rates of immigration to this region from other parts of Iran as well as its neighboring countries such as Afghanistan are reported. However, despite the high prevalence of TB, there is limited information on the strains prevalent in this area. In the present study, the 140
M. tuberculosis isolates from Northeast of Iran were identified by using Spoligotyping and MIRU-VNTR methods.
The molecular typing of strains by Spoligotyping could not act as a specific indicator for other members of
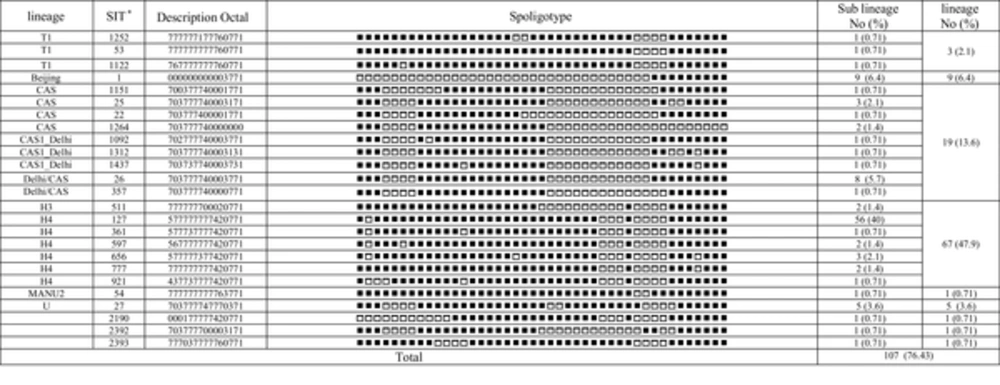
M. tuberculosis complex. The Haarlem family was recognized as the most common type, and the major shared types of Haarlem family were SIT127. It was followed by the CAS family that was ranked as the second most prevalent Spoligotype. Previously published studies from Iran also reported that Haarlem and CAS were the most predominant families (
6,
21). Also, in another study performed in Iran, it was shown that Haarlem was the most frequent lineage but the second most prevalent Spoligotype was reported the Beijing genotype (23.5%), followed by the CAS family (
22). This difference may be due to the used strains, which were obtained from patients with MDR-TB and were highly associated with the Beijing genotype. Nine of the 140 studied strains (6.4%) in the present study were the Beijing genotype. This genotype was the third most predominant family in the present study. Also, other studies from Iran showed nearly similar corresponding results, e.g. 6.3% in Ramazanzadeh and colleagues (
23), 5.3% in Merza et al. (
24), and 8.1% in Mozafari et al. (
8). The study of Mohajeri et al. (
25) showed that the rate of Beijing genotype in the western provinces of Iran was 10.4%. It seems that the frequency of the Beijing genotype in the western provinces of Iran is higher than other areas of the country.
In the present study, 84 out of 140 cases (60%) were female, and 56 (40%) were male. In the Haarlem family, 39.4% were male and 60.6% were female. CAS family contained 31.6% male and 68.4% female. Unlike other lineages, in the Beijing family, 66.7% were male and 33.3% were female. (Male to female ratio was 2 to 1). But there was no significant relationship between sex and lineage (Chi-square: 5.656, P value: 0.463). Study on Afghan refugees in Iran showed that the predominant genotypes were Haarlem and CAS families (
6). Since Khorasan provinces are located in the Northeast of the country, and share geographic borders with Afghanistan, these family strains may have been transported from Afghanistan to Khorasan, Iran.
The genotyping pattern obtained from the South Khorasan province (Haarlem 14.3%, CAS 42.9%) was different from the North and Razavi Khorasan provinces (Haarlem 48.9%, CAS 12.1%). It was, however, similar to the pattern obtained from Sistan and Baluchistan province (Southeast of Iran) (
20) and Pakistan (
26). The South Khorasan province is in the vicinity of Sistan and Baluchistan province and Pakistan country. It seems that these family strains have been most probably transported from these two regions to South Khorasan. In the present study, there was a significant relationship between city of residence and Beijing and CAS lineages (P value: 0.040 for Beijing lineage and 0.032 for CAS lineage). However, there was no significant relationship between city of residence and other lineages. The same 140 isolates were analyzed using the MIRU-VNTR technique and 77 different profiles were observed. 59.3% (83/140) of the isolates were distributed into 19 clusters with 100% of identity, containing 10 clusters of 2 strains each, 5 clusters of 3 strains each, 2 clusters of 4 strains, 1 cluster of 13 strains, and 1 cluster of 27 strains. Based on this study, MIRU10, MIRU26, and ETRF were highly discriminating, but ETRD was poorly discriminating. Most of the previous studies in Iran similarly found that MIRU10, MIRU26, and ETRF were highly discriminating, and MIRU4 or ETRD was poorly discriminating (
1,
27-
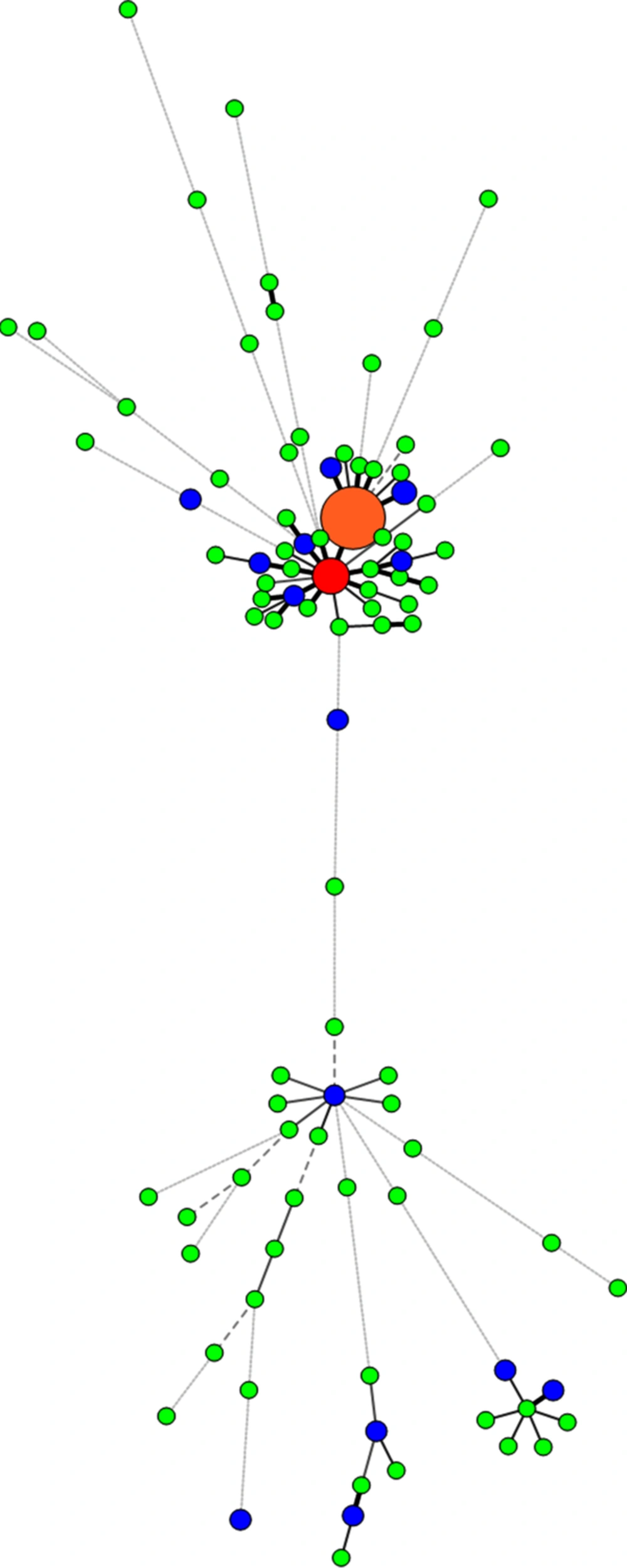
30). By combining the two techniques, 94 distinct patterns comprising 15 clusters containing 61 isolates (43.6%), and 79 unique patterns were identified.
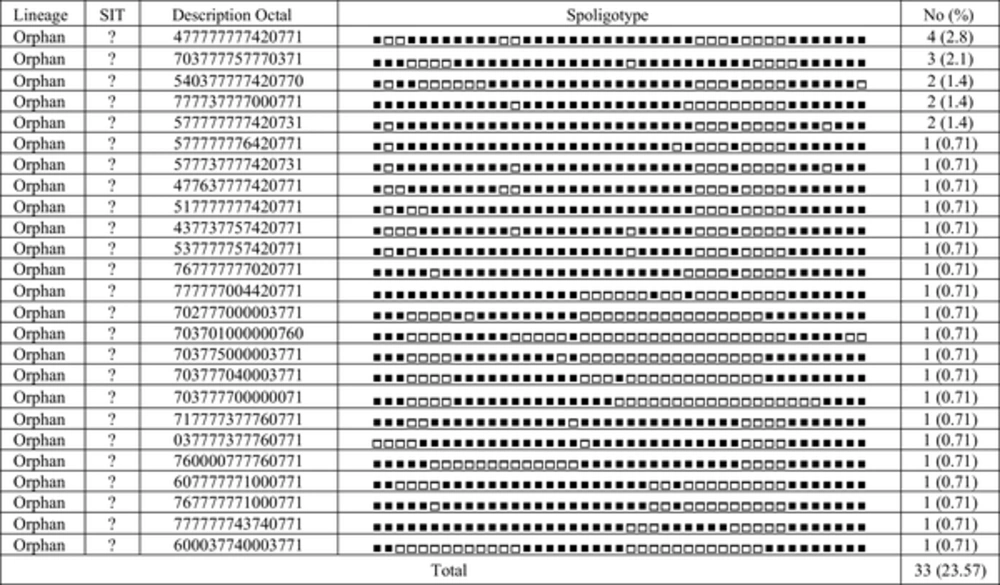
In 10 clusters, some strains exhibited exactly the same profile typing for two methods and were classified as real clusters. Some strains (20 isolates) in the other 9 MIRU-VNTR clusters, however, exhibited different Spoligotype profiles. These different types are named homoplasy. Interestingly, one of the cases of homoplasy was about the two samples from two patients, one of them isolated from the youngest patient of the study (15 years old), and the other one from the oldest patient (91 years old), both living in Mashhad, the center of Razavi Khorasan province. The strains of both of them had the same MIRU-VNTR profile but different types of Spoligotype profiles. Because the half-life of MIRU-VNTR profile is shorter than that of Spoligotyping profile (
31), different observed MIRU-VNTR profiles are more reliable and natural in comparison with Spoligotyping patterns. Similar MIRU-VNTR profiles with different Spoligotype profiles are named homoplasy. In this phenomenon, an independent mutational event in a particular spacer leads to the loss of the spacer and makes an invalid Spoligotype pattern (
32).
All strains with MDR and combined (INH + RIF) resistance, in the present study, were related to the Beijing strains, confirming its tendency to resist antibiotics. Also, all mono drug resistant strains were relevant to the Haarlem family, but other strains were susceptible to the first-line anti-TB drugs. However, there was no significant relationship between drug resistance and lineage (Fisher’s Exact Test: 218.725, P value: 0.133). Also, the relationship between age and lineage was not significant (Fisher’s Exact Test: 13.040, P value: 0.588). In addition, the discriminatory power (HGDI) of Spoligotyping method was 0.832, the discriminatory power (HGDI) of MIRU-VNTR method was 0.951, and the discriminatory power (HGDI) of the combined two techniques was 0.962, which was higher than that of each technique alone. Therefore, the results showed that in molecular typing of M. tuberculosis, the simultaneous use of the two methods of “Spoligotyping” and “MIRU-VNTR” could increase the genetic patterns of M.tuberculosis strains much effectively compared to the use of only one method.
5.1. Conclusions
MIRU-VNTR is a reproducible method, potentially applicable in tracking epidemiological events such as transmission or relapse. It also allows direct comparison of results between laboratories. In MIRU-VNTR typing method, according to the genotype of each area, the loci with high discriminatory power (such as miru10, miru26, and ETRF in Iran) are recommended to be used and the loci with poor discrimination (such as ETRD in Iran) are not recommended. This typing technique is also accepted as a first line method for molecular epidemiology of M. tuberculosis.
To reduce the amount of homoplasy with these methods, it is recommended to use sequencing-based methods simultaneously.