Previous studies in Iran have reported a high rate of MRSA infections (
9,
23). The results of the current study showed an MRSA infection rate of 20.4% in the samples collected from different wards. In 2 studies performed in Tabriz (2013) and Kermanshah (2013), Iran, methicillin resistance rate was found to be 34.4% and 37.2%, respectively (
24,
25). Also, in a study by Leandro et al. in 2008,
S. aureus MIC was examined. MIC above 8 mg/mL was considered resistant, and MIC below 4 mg/mL indicated methicillin susceptibility (
26).
In the present study, the majority of
mecA-positive strains belonged to type IV, followed by types III and I, respectively. In a study by Hallin et al. in Belgium in 2006, a total of 511 MRSA isolates were tested for Scc
mec. The results revealed that most strains belonged to type IV, while a small number were of types II and I; also, Scc
mec type III was not reported in any of the strains (
27). In this regard, in a study by Soge et al., all MRSA isolates belonged to Scc
mec type I, and
mecA-positive,
coa-negative
S. aureus isolates belonged to a variety of groups (I, II, III, and IV) (
28).
Typing is of utmost importance for controlling resistant strains, understanding their epidemiology, and determining whether they proliferate from a single clone or multiple clones. In our previous study (
1), all the strains (MRSA and MSSA) were subjected to MLST. Most MRSA isolates were epidemic MRSA and belonged to type ST239. It is noteworthy that 2 MRSA isolates indicated 2 novel STs, ie, ST1939 (isolated from sputum) and ST1942 (isolated from an abscess); these strains were of similar Scc
mec types (group IV). It should be noted that STs were more diverse in MSSA strains.
In addition, novel STs were found in the mentioned isolates, which are as follows: ST1938, ST1947, ST1939, ST1940, ST1937, ST1941, and ST1936. ST8 was the predominant ST in MSSA isolates, mainly collected from wound samples. Regarding the number of novel STs found in this study, it should be noted that no studies on MLST have been conducted in Iran, and it is possible to find more novel STs in extended geographical regions.
In a study by Kim et al. in 2007, the prevalence of MRSA strains was reported to be 58.4%, of which 5.9% were community-acquired. On the other hand, the present results showed an MRSA rate of 20.4%; based on the findings, 5.9% of MRSA strains were community-acquired. In the present study, the prevalent ST was ST72, while other main STs, including ST239 and ST5, were the minority (
29). In addition, MRSA isolates were community-acquired and epidemic. Community-acquired isolates are also prevalent in Korea, although the STs are not epidemic.
In the mentioned study performed in Korea, community-acquired MRSA isolates were mainly from wound exudate, eye and ear secretions, and blood, whereas in the present study, community-acquired MRSA isolates, mainly belonging to ST239-Sccmec IV, were collected from wound, sputum, urine, abscess, peritonea, and blood samples. This is atypical of community-acquired MRSA isolates, since they are usually collected from more superficial infections (usually related to the skin). Therefore, these isolates are growing stronger to produce deeper and more severe infections; this in fact underlines the importance of public hygiene.
It is well established that
agr locus in
S. aureus is a virulence regulatory system, with hypervariable regions making it a suitable target for typing. Similar to most previous studies, the present research found
agr group I to be of the highest prevalence (90.8%), followed by group III (3.7%). It should be noted that groups II and IV were not found, and a number of isolates (5.5%) were nontypable. Basically, the prevalence of
agr-specific groups in our isolates (type I being the most prevalent) was similar to the patterns found in other parts of the world, as well as the patterns previously reported in Iran (
21).
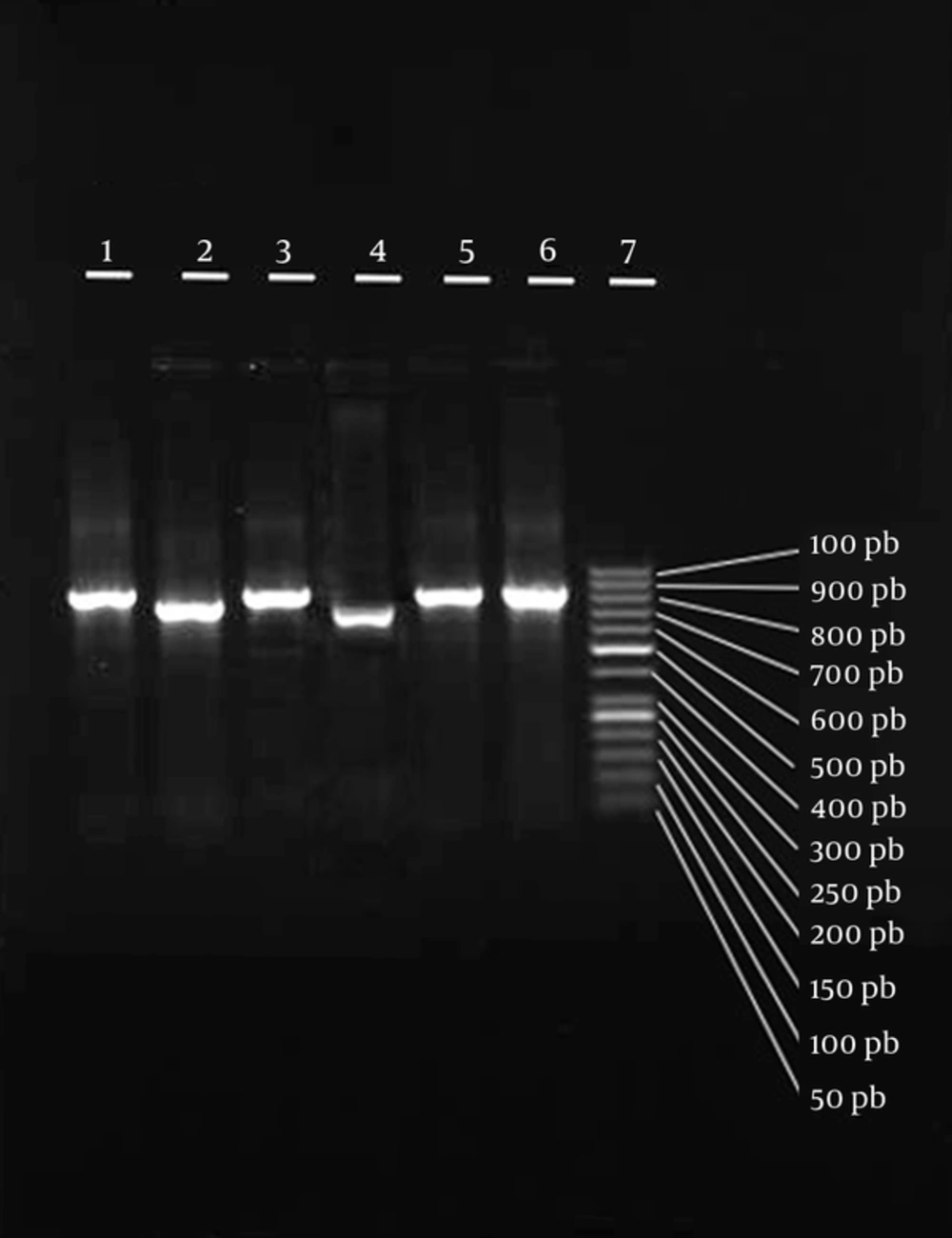
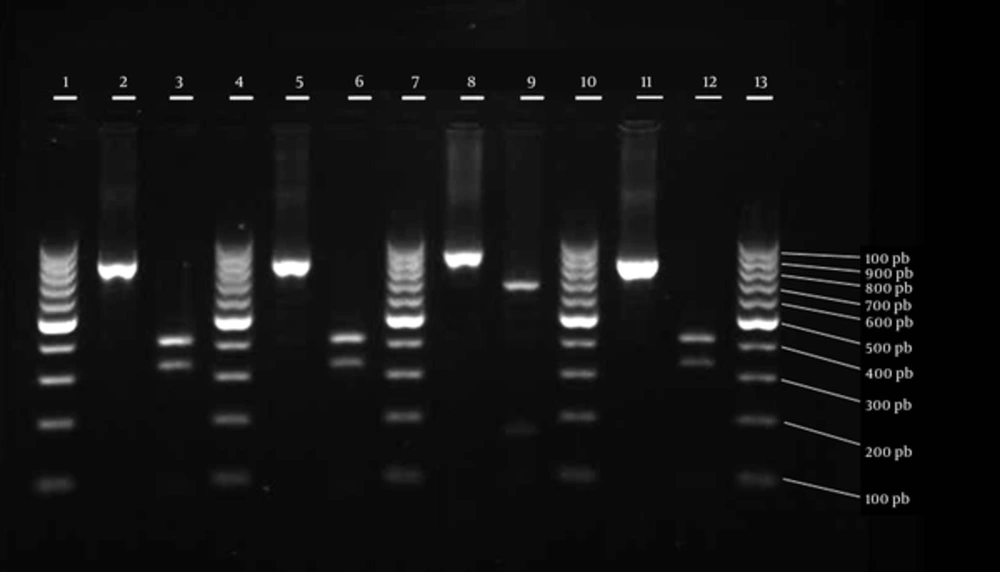
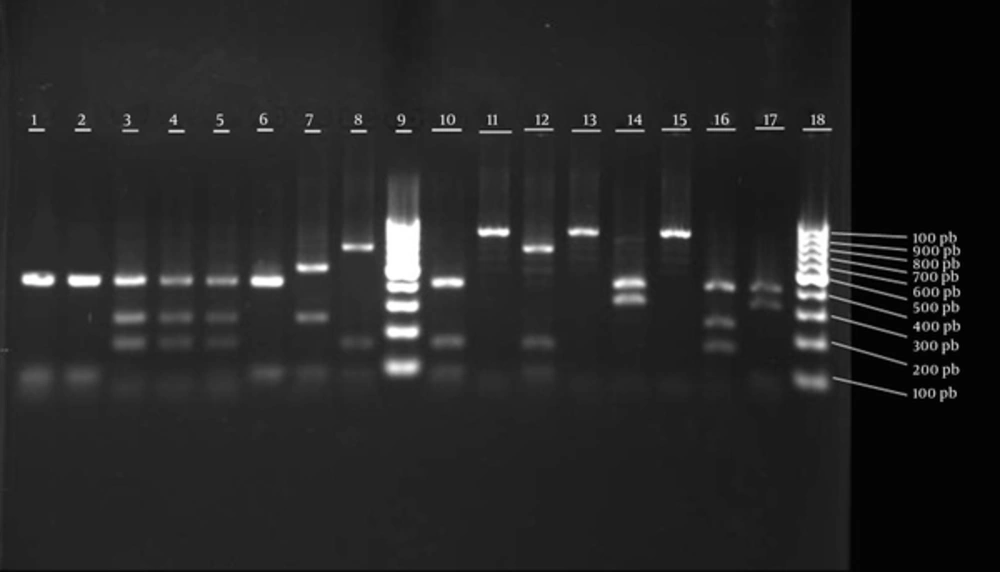
In addition to other typing methods, PCR-RFLP of coa gene was used in this study for typing MRSA and MSSA strains. The results indicated 1 sample collected from a person in a different hospital. Moreover, 15 MSSA isolates showed 8 RFLP patterns, which were different from the identified MRSA isolates. In the current study, MRSA isolates were not variable in terms of restriction patterns, whereas MSSA isolates had a variety of patterns (8 types).
Himabinda et al. in 2009 reported that the majority of MRSA isolates belonged to the A10 pattern (
30). In the present study, the majority of MRSA strains belonged to A2 pattern. Also, most of MSSA strains had A11, A7, and A10 patterns, which were more variable, compared to MRSA strains. In another study by Hata et al. in 2010 on
S. aureus isolates, the differentiation power of various typing methods was analyzed. They reported MLST to have greater differentiation power, compared to RFLP for
coa gene. The results showed high concordance between PCR-RFLP and MLST results (
31).
5.1. Conclusion
The present study indicated that the results of MLST and PCR-RFLP assay for coa gene were consistent. A total of 54 isolates were categorized into 13 STs and 12 groups by MLST and PCR-RFLP methods, respectively. No significant correlation was found between the source of bacterial collection, ST, and PCR-RFLP pattern. It can be concluded that there is striking similarity in the differentiation power of MLST and PCR-RFLP for coa gene in the samples tested in this study.