1. Background
Helicobacter pylori is a Gram-negative, microaerophilic bacterium that infects the gastrointestinal epithelium and affects nearly half of the global population (1). It is a common cause of chronic gastritis, which often remains asymptomatic (2). Additionally, it has been linked to various health conditions, including the aggravation of gastritis, duodenal ulcers, iron deficiency anemia, lymphoma, and reactive adenocarcinoma (3-5). As the only bacterium classified as a class 1 carcinogen (6), H. pylori is the strongest known risk factor for gastric adenocarcinoma, which accounts for nearly two-thirds of gastric cancer cases worldwide (7). Moreover, the expression of the cytotoxin-associated gene A (cagA) in H. pylori has been considered a risk marker for mucosal atrophy and carcinogenesis. The encoded protein induces morphological and malignant transformations in gastric epithelial cells by disrupting intracellular signaling pathways (5, 8, 9). The bacterium can evade the host’s immune system by entering the cytoplasm of gastric epithelial cells, where neoplastic transformation occurs through the oncogenic protein encoded by the cagA gene (5, 9). Notably, gastric inflammation works in tandem with compensatory genetic and epigenetic alterations, leading to self-sustaining carcinogenesis (5, 10).
Helicobacter pylori infection commonly occurs within the first decade of life (11). In children, it is often transient and self-limiting (12), with symptoms typically manifesting later in life (13). The presence of the bacterium in the oral cavity supports both fecal-oral and oral-oral transmission routes (14-17). Additionally, poor personal and food hygiene practices in densely populated areas significantly contribute to intrafamilial transmission (18). Interestingly, the eradication of H. pylori from the oral cavity has been shown to improve the success rate of gastric infection treatment (19). Consequently, several previous studies have suggested that oral H. pylori may indicate actual infection rather than mere transient colonization (16, 20, 21).
The established relationship between H. pylori infection and gastric cancer (7), along with its poor prognosis as a leading cause of cancer-related mortality (22, 23), underscores the importance of early detection (24). This is particularly relevant since the prevalence of H. pylori in adulthood has been suggested to depend on childhood infection (25). Previous studies on the Iranian population have reported H. pylori infection prevalence ranging from 30.6% to 82% (26). However, given the close relationship between oral H. pylori and actual infection (27), and the fact that H. pylori infection typically occurs before the age of five (28), the prevalence of the bacterium in young Iranian children has yet to be investigated.
2. Objectives
The present study aimed to detect H. pylori and the cagA gene using polymerase chain reaction (PCR) in oral samples from asymptomatic children under the age of five residing in Tehran, Iran. Additionally, the study explored associations with age, sex, family history of gastritis, and the decayed, missing, and filled teeth (dmft) score.
3. Methods
3.1. Study Design and Participants
Ten kindergartens across districts #1 and #2 of Tehran were selected for this cross-sectional study. A total of 160 asymptomatic, healthy children under five years of age participated. Participants were chosen using simple random sampling from a list of enrolled children provided by the kindergartens. The sample size was determined based on feasibility considerations and a previous study by Castro-Muñoz et al. on 162 asymptomatic young Mexican children, which reported a prevalence rate of 13% for oral H. pylori, with an expected precision of 5.21% (29). Only asymptomatic children whose parents provided written informed consent were included in the study. Confidentiality and anonymity of the participants were ensured throughout the research process. Children or parents/guardians unwilling to participate, as well as those with systemic diseases or gastrointestinal symptoms, were excluded from the study.
3.2. Data Collection
Data collection took place between January and March 2020. For each participant, information on age, sex, and family history of gastritis was obtained from parents or legal guardians using questionnaires specifically designed for this study. A trained dentist then conducted an individual oral examination for each participant through visual inspection, using a dental explorer and mirror under adequate lighting conditions, to record the dmft score (30).
3.3. Sample Collection
Oral samples were collected by swabbing the buccal mucosa and sublingual areas of each participant using sterile cotton swabs (Arta Teb, Iran) (29). The swabs were immediately placed in sterile test tubes containing Luria-Bertani (LB) broth (Thermo Fisher Scientific, USA) and transferred to an independent laboratory for further analysis. To minimize potential detection bias, PCR analysis was conducted by trained personnel who were blinded to the participants’ information (31).
3.4. DNA Extraction
Bacterial genomic DNA was extracted from the collected samples using the HiPurA™ Bacterial Genomic DNA Purification Kit (HiMedia, India) following the manufacturer’s instructions. The quantity and purity of the extracted DNA were evaluated using a NanoDrop spectrophotometer (Thermo Scientific, USA).
3.5. Polymerase Chain Reaction Analysis
The presence of H. pylori and the cagA gene in oral samples was evaluated using PCR with specific primers (Table 1). Each PCR reaction was carried out in a total volume of 25 µL, consisting of 12.5 µL of master mix (Amplicon, Denmark), 1 µL of each primer (10 pmol/µL), 2 µL of template DNA, and 8.5 µL of nuclease-free water. The thermal cycling conditions were as follows: Initial denaturation at 95°C for 5 minutes; denaturation at 95°C for 30 seconds (35 cycles); annealing at 55°C for 30 seconds; extension at 72°C for 1 minute; and a final extension at 72°C for 7 minutes (32). Genomic DNA from the standard H. pylori strain (ATCC 49503) was used as a positive control (33-35).
3.6. Gel Electrophoresis
The PCR products were analyzed by electrophoresis on a 1.5% agarose gel stained with ethidium bromide. The gel was run at 100 volts for 45 minutes in 1X TAE buffer and visualized under ultraviolet light using a gel documentation system (Bio-Rad, USA). The presence of specific bands corresponding to H. pylori and the cagA gene indicated positive samples (32).
3.7. Statistical Analysis
Statistical analysis was performed using SPSS software version 22.0 (IBM Corp., Armonk, NY, USA). Fisher’s exact test was used to examine associations between H. pylori presence and variables such as age, sex, family history of gastritis, and dmft scores. A P-value of less than 0.05 was considered statistically significant.
4. Results
4.1. Participants’ Demographics and Dental Health Status
The mean age of the children was 3.15 ± 1.30 years. No pathological soft-tissue oral lesions were observed during intraoral visual examinations. The dmft scores ranged from 0 to 3, with a mean of 1.56 ± 0.49. A positive family history of gastritis was reported in 25 children (15.6%).
4.2. Prevalence of Helicobacter pylori
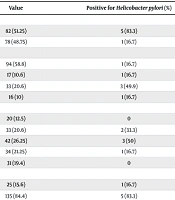
The PCR identified bacterial genomic DNA in 6 children (3.75%), all of whom were also positive for the cagA gene. Statistical analysis did not reveal any significant association with the participants' age, sex, family history of gastritis, or dental health (P > 0.05). Table 2 provides a breakdown of participants by age group, sex, dmft score, and family history of gastritis.
| Variables | Value | Positive for Helicobacter pylori (%) | P-Value b |
|---|---|---|---|
| Gender | 0.109 | ||
| Male | 82 (51.25) | 5 (83.3) | |
| Female | 78 (48.75) | 1 (16.7) | |
| dmft Score | 0.173 | ||
| 0 | 94 (58.8) | 1 (16.7) | |
| 1 | 17 (10.6) | 1 (16.7) | |
| 2 | 33 (20.6) | 3 (49.9) | |
| 3 | 16 (10) | 1 (16.7) | |
| Age | 0.567 | ||
| < 1 | 20 (12.5) | 0 | |
| 1 - 2 | 33 (20.6) | 2 (33.3) | |
| 2 - 3 | 42 (26.25) | 3 (50) | |
| 3 - 4 | 34 (21.25) | 1 (16.7) | |
| 4 - 5 | 31 (19.4) | 0 | |
| Family history of gastritis | 0.913 | ||
| Present | 25 (15.6) | 1 (16.7) | |
| Absent | 135 (84.4) | 5 (83.3) |
Demographic Information, Dental Health Status, and Family History of Gastritis a
5. Discussion
To the best of the authors' knowledge, no previous study has investigated the prevalence of H. pylori in young children in Tehran. Therefore, this study aimed to explore the oral prevalence of H. pylori and the cagA gene in symptom-free young children in this city. The PCR was used in this study due to its ability to exclude other phylogenetically close bacteria, such as Campylobacter species (37), as well as its high reliability and efficiency in detecting H. pylori in children (38). The results showed a detection rate of 3.75% among children, which is considerably lower than that reported for Iranians under 15 years old (42% prevalence, 95% CI: 41 - 44%) or for the general healthy Iranian population (30.6% to 82% overall prevalence, with an age range of four months to 83 years) (26, 39).
This disparity may stem from differences in detection methods—such as enzyme-linked immunosorbent assay or stool antigen—, age-related inclusion criteria, or socioeconomic factors. In fact, social class, parental educational level, living conditions, household size, and drinking water sources have all been shown to influence the prevalence of H. pylori, even within different regions of the same country (17, 18). Moreover, the low prevalence in this study can be explained by our sampling method, which did not include dental plaque. Indeed, dental plaque has the highest detection rate of H. pylori in the oral cavity owing to its biofilm characteristics (21, 40). The lower prevalence of H. pylori in younger children, compared to older children or adults, in other countries also help explain the low detection rate in this study (15, 26, 41).
In this study, the virulence gene cagA was detected in all H. pylori-positive oral samples. This finding is consistent with a previous study that reported similar results using saliva samples from Iranian adults with gastroduodenal disease, who were referred to an endoscopy center in a different city (34). Additionally, another study investigating H. pylori genotyping and host antibody response identified the cagA gene in 91% of Iranian H. pylori strains isolated from gastric biopsies (35). These findings suggest that the cagA gene may exhibit low regional and possibly age-related variability within Iran. However, further studies are needed to validate this hypothesis.
In the present study, none of the H. pylori-positive subjects reported experiencing gastrointestinal symptoms at the time of investigation. This lack of symptoms may be attributed to a diminished immune response and reduced cell infiltration in children following bacterial colonization, leading to less gastric inflammation compared to adults (42, 43). However, even in the absence of symptoms, when H. pylori infection is suspected, it is important to consider the increased risk of gastrointestinal ulceration, particularly from nonsteroidal anti-inflammatory drugs and chronic immune thrombocytopenic purpura (44).
Although H. pylori-positive participants in this study were predominantly male, the difference between the two sexes did not reach statistical significance. This finding aligns with a previous study evaluating anti-H. pylori IgG in the serum of 2,561 healthy adults living in rural and urban areas of Tehran province (45). A meta-analysis also confirmed the absence of a clear gender-based predisposition for H. pylori infection in children, possibly due to differential antibiotic exposure or gender-specific protective immunity rather than solely differential exposure (46). Interestingly, while sex was found to be of no significance in H. pylori prevalence among children in South Korea, being male was reported as a risk factor in adults (41). Thus, although no significant association was found in the present study, a higher bacterial detection rate in males may suggest a possible trend in young Iranian children that warrants further investigation.
The present study did not show any association between H. pylori detection and the dmft score, which aligns with a previous report by Mehdipour et al. (mean age: 7.97 ± 1.83 years) (30). In contrast, Sruthi et al. reported significantly higher mean scores in Indian H. pylori-positive children aged 3 - 6 years, demonstrating that higher caries status and caries severity were associated with a greater prevalence of H. pylori (47). This discrepancy may be attributed to their small sample size (n = 20), as well as differences in geographical location, age range, and sampling method.
The present study had several limitations. First, a larger sample size was required to detect small effect sizes. Second, the cross-sectional nature of the study was unable to provide data on incidence rates, sources of infection, or lifelong trends, all of which are essential for devising effective future prevention strategies. Third, while PCR is a highly specific method, it does not differentiate between transient colonization and persistent infection. Fourth, participants were recruited from only two districts, which may limit the generalizability of the findings. Consequently, future studies should include larger, more diverse samples and incorporate additional diagnostic techniques.
5.1. Conclusions
The prevalence of oral H. pylori was low in the study population. However, the cagA gene was detected in all H. pylori-positive participants. No associations were found with age, sex, family history, or dental health status.