1. Background
Plasmid-mediated extended-spectrum beta-lactamase (ESBL) enzymes-producing microorganisms are most common in both hospital and community settings. The extended-spectrum CTX-M β-lactamases are the most prevalent ESBLs worldwide, that first defined in Japan in 1986 (the enzyme was firstly named TOHO-1), and have been mainly associated with Enterobacteriasea, most prevalently in Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis and Salmonella typhimurium (1). These enzymes are member of Ambler class A and classified in group 2be of the Bush, Jacoby and Medeiros classification and generally most active against cefotaxime and show little activity against ceftazidime and are inhibited by clavulanic acid and tazobactam (2, 3).
Phylogenetically, these enzymes have been classified into five major groups according to the amino acid similarities: the CTX-M-1cluster (CTX-M-1, -3, -10, -11, -12, -15, -28 and FEC-1), the CTX-M-2 cluster (CTX-M-2, -4, -5, -6, -7, -20, and TOHO-1), the CTX-M-8 cluster (CTX-M-8), the CTX-M-25 cluster (CTXM- 25 and -26), and the CTX-M-9 cluster (CTX-M-9, -13, -14,-16, -17, -19, -21, -24, -27, and TOHO-2) (4, 5). The rapid dissemination of CTX-M enzymes is facilitated by plasmids, transposons, and integrons gene cassettes (6). Recently, prevalence of CTX-M-type extended-spectrum - β lactamases have dramatically increased among Asian, African, European, and American E. coli clinical isolates (1, 2, 7). Therefore, rapid and discriminatory typing methods are essential for epidemiological analysis of CTX-M-producing isolates.
Little data on the type of CTX-M ESBL transmission is available at the regional level, and the genetic diversity of resistant isolates has not been reported until now. Rep-PCR DNA fingerprinting, is a rapid and simple typing method that relies on PCR amplification of the DNA between adjacent repetitive extragenic palindromic (rep) elements interspersed throughout the genome (8, 9).
2. Objectives
This study aims to determine the antimicrobial resistance and E. coli CTX-M ESBL-producing isolates and also to determine their genetic diversity using PCR based fingerprinting techniques (rep-PCR).
3. Materials and Methods
3.1. Clinical isolates
During 2009 - 2010, 200 E. coli isolates were collected from 5 different teaching hospitals in Tehran. These isolates were obtained from blood cultures (n = 15), urine samples (n = 63), soft tissue specimens (n = 63), stool specimens (n = 38), and exudates samples (n = 21). The isolates were identified by conventional methods, and were stored in skimmed milk at –70°C until required.
3.2. Susceptibility Tests and Confirmation of ESBL Production
Susceptibility testing was conducted by disk diffusion according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI) (10). Fourteen Antimicrobial disks such as Cefoxitin, Ceftazidime, Cefotaxime, Cefepime, Aztreonam, Erythromycin, Gentamicin, Tetracycline, Co-trimoxazole, Amoxicillin, Amoxicillin-clavulanic acid, Imipenem, Amikacin, and Ciprofloxacin were used in this study. E. coli ATCC 25922 was used for quality control purposes in susceptibility testing. The ESBL phenotype was detected by combined disk methods using discs of Ceftazidime and Cefotaxime with and without clavulanic acid. The MIC of antibiotics were determined by agar dilution methods using Ceftazidime and Cefotaxime with and without clavulanic acid (10).
3.3. Detection of the blaCTX-M Gene
PCR template DNA was prepared using a boiling lysis method as previously described (11). The specific primers (Table 1) for diverse CTX-M groups (CTX-M-1, CTX-M-2, and CTX-M-9 groups) were used, as previously described (12 , 13). Amplification reactions were performed in a total volume of 25 µL of reaction mixture containing 5µL of 10× PCR buffer, 2.5 mM Mgcl2, 200 µM dNTPs, 1.25 units of Taq polymerase, 10 pmol of each primer and 1µL of sample DNA. Amplification reactions were carried out in an Eppendorf thermal cycler (Eppendorf AG, Hamburg, Germany), with an initial denaturation (4 min at 94°C) followed by 30 cycles of denaturation (60 s at 94°C), annealing (30 s at 55°C), and extension (1 min at 72°C), with a single final extension of 5 min at 72°C. DNA template from control clinical strains with well characterized CTX-M beta-lactamases (groups 1,2 ,9) were used as the positive controls for PCR (14). The PCR products were analyzed on 1% agarose gels stained with ethidium bromide.
| Target | Primer Sequence | Amplicon size, bp |
|---|---|---|
| CTX-M1 (F) | F-GGT TAAAAAATC ACTGCG TC | 850 |
| R- TTGGTGACGATTTTAGCCGC | ||
| CTX-M2 | ATG ATG ACT CAG AGC ATT CG | 850 |
| TGG GTT ACG ATT TTC GCC GC | ||
| CTX-M9 | ATG GTG ACA AAG AGA GTG CA | 850 |
| CCC TTC GGC GAT GAT TCT C |
3.4. rep-PCR
DNA was extracted by using the Cinagen Mini kit (Cinagen Co. Iran). Repetitive extragenic palindromic sequence PCR (rep-PCR) have been described in detail in previous publications (8, 9). The primers REP1 (5-IIIGCGCCGICATCAGGC-3) and REP2 (5-ACGTCTTATCAGGCCTAC-3) was used to amplify putative REP-like elements in the genomic bacterial DNA. Amplification reactions were performed in a final volume of 25 μL of 40 pmol of each primers, 1.25 mmol/L of each four dNTPs, 3 U Taq DNA polymerase, 1.5 mmol/L MgCl2, 10µL of 10× PCR buffer and 10 ng of genomic DNA. PCR were carried out in an Eppendorf thermal cycler, with an initial denaturation (10 min at 94°C) followed by 30 cycles of denaturation (1 min at 94°C), annealing (1 min at 40°C), extension (6 min at 65°C), and with a single final extension of 16 min at 65°C. Aliquots (10 µL) of each sample were subjected to electrophoresis on 2.0% agarose gel in Tris acetate–EDTA buffer at 50 V for 3 h, stained with ethidium bromide, and visualized under UV light. In each run, rep- PCR was repeated for E. coli ATCC 25922, as internal control for the standardization of the PCR parameters.
Phoretix 1D and NTSYSpc software was used to make complete linkage dendrograms patterns. Bands Identification performed manually. The Dice similarity coefficient was used with position tolerance settings of 2.0%. Rolling ball method was used to remove the background.
4. Results
4.1. Antimicrobial Susceptibility and Detection of ESBL Production
The results for the antibiotic resistance are shown in Table 2. Among the β-lactams tested, maximal resistance was noticed against amoxicillin (91%) and amoxicillin-clavulanic acid (85%), while 39.5% and 72% of isolates were resistant to ceftazidime (CAZ) and cefotaxime (CTX) respectively. Resistance rate to other agents were 49% for Cefoxitin, 36% for Cefepime, 39% for Aztreonam, 94% for Erythromycin, 36% for Gentamicin, 58% for Tetracycline,¬ 57% for Co-trimoxazole , 15% for Amikacin, and 39% for Ciprofloxacin. None of the isolates shows resistance to Imipenem.
A total of 140 (70%) non-duplicate clinical isolates of ESBL positive E. coli were detected based on their phenotype using cefotaxime/clavulanate and ceftazidime/clavulanate. The difference in inhibition zones of β-lactam disks with and without clavulanic acid was 5 - 10 mm.
The MICs of all isolate were determined for CTX and CTZ with and without inhibitor (Table 3). A total of 144 (72%) isolates were resistant (MIC ≥ 4mg/L) to CTX, and 42 (29%) of resistant isolates expressed high MIC(256mg/L ≤). The MIC of CTZ for 78 (48%) of isolates were resistant (MIC ≥ 16mg/L), and 11 (14%) of resistant isolates expressed high MIC (256 ≤).
The phenotypic ESBL agar dilution confirmatory test was positive, and the rate of CTX and CTZ resistant isolates dramatically decreased from 72% to 8.5% and 39% to 4.5% respectively, in the presence of clavulanic acid (Table 3).
4.2. Detection of blaCTX-M Gene
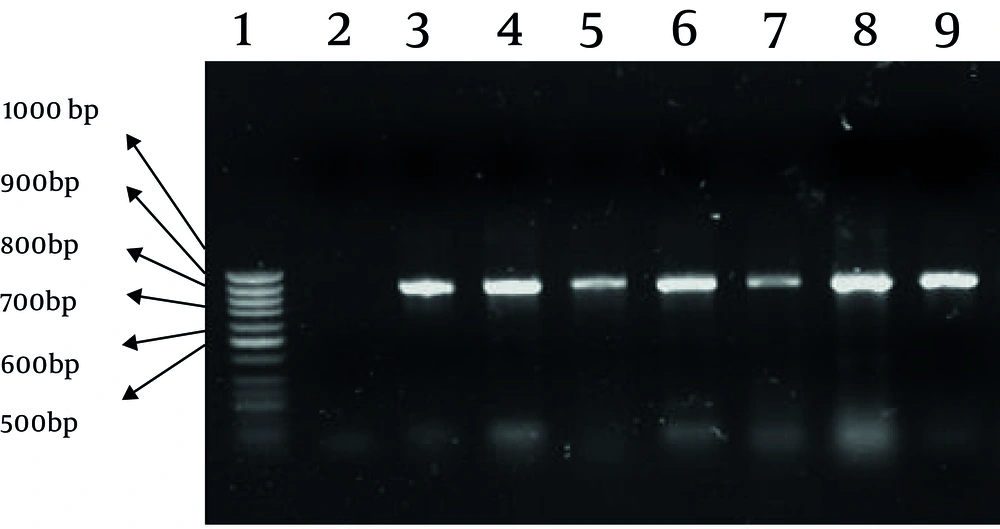
PCR was performed for all CTX resistant isolates according to results obtained by phenotypic tests. Eighty nine (61.8%) of the 144 CTX resistant E. coli isolates carried the blaCTX-M-1 group alleles (Figure 1). None of the isolates were positive for CTX-M groups 2 and 9.
The majority of blaCTX-M-1 -carrying isolates were recovered from urine (33.7%) and tissue (32.5%), followed by 18 (20.2%) from stool, and 9 (10.1%) from exudates and 3 (3.3%) from blood. The resistance of blaCTX-M-1 -carrying E. coli isolates to antibiotics were 93.2% for amoxicillin and amoxicillin-clavulanic acid, 70.7% to ceftazidime, 68.5% cefoxitin, 65.1% to cefepime, 86.5% to Aztreonam, 94.3% to Erythromycin, 38.2% to Gentamicin, 75.2% to Tetracycline, 67.4% to Co-trimoxazole, 14.6% to Amikacin, and 43.8% to Ciprofloxacin. No imipenem resistance was detected.
The MIC of CTX for 38 (42%) blaCTX-M-1 -carrying E. coli isolates was high (256≤). While the MIC of 7 (7.8%) of blaCTX-M-1 -carrying isolates were 4 - 8µg/mL. The MIC of CTX for all blaCTX-M-1 -carrying isolates was reduced by 4 - 7 doubling dilutions in the presence of clavulanic acid. The blaCTX-M-1 -carrying isolates were also resistant to CAZ, and only 11 isolates were susceptible.
4.3. rep-PCR
Molecular typing with rep-PCR was used to define the clonal patterns of 89 CTX-M1 isolates. There were no common rep-PCR fingerprint patterns that provided an average to differentiate CTX-M1 isolates. However, these isolates included 4 different clusters (Figure 2). The genetic fingerprints of isolates obtained different sources such as blood, urine, and stool were similar, and differences were found between isolates obtained from the same samples.Figure 2.
Phoretix 1D and NTSYSpc software was used to make Complete Linkage dendrograms of fragment patterns. The blaCTX-M1 isolates classified into four clusters.
There was also considerable genetic diversity among isolates collected from the same hospital. The levels of similarity of the rep-PCR fingerprints of isolates ranged from 40 to 90%.
5. Discussion
Multidrug resistant isolates of E. coli have been increasing worldwide, and is associated with the acquisition of mobile element-encoded β-lactamases. The production of CTX-M β-lactamases is an important mechanism of resistance to oxyimino-cephalosporins in this microorganism. This family of plasmid-mediated ESBLs of Ambler class A that has been detected mainly in Asian countries, Europe, and America, (2, 12-14). In this study, 140 isolates (70%) showed the ESBL phenotype by combined disk and Agar dilution methods. In a study conducted by Mirzaee, 56% of E. coli isolates from three university hospitals of Tehran revealed ESBL phenotype, which is less than our findings (15). Obviously, our results were analyzed based on the new 2010 CLSI interpretative criteria (10). A high incidence of ESBL-producers amongst E. coli was reported from India (79%), Pakistan (64%) and Kuwait (77.9%) (16-18). These studies indicate a high incidence of ESBL-producers amongst E. coli isolates in Asian countries.
In this study, a total of 63.5% of ESBL producers isolates carried blaCTX-M-1, this subgroup of blaCTX-M is prevalent in our country (15, 19). The blaCTX-M-1 has detected as the most common type of blaCTX-M from Asia, Europe, North and South America among multidrug-resistant E. coli (17, 20-22). A high level resistance to β-lactam, as well as Erythromycin, Tetracycline, and Co-trimoxazole, was observed among our blaCTX-M-1 carrying isolates. However, aminoglycosides such as Amikacin and to some extent, Gentamicin was showed the active antibacterial effects. Imipenem was the most effective antibiotic against all ESBL- producing E. coli.
Analysis of the rep-PCR typing of the bla CTX-M-1 -carrying isolates showed that there is no evidence of any clonality within a single hospital or clonal spread across hospitals. The heterogeneous natures of these enzymes were reported by Al Hashem et al. (16), and Khanna et al. (23). The rapid dissemination of non-clonal multi-resistant CTX-M-1 producing E. coli isolates indicates the need for further investigations in medical centers.