1. Background
Human Herpes Virus 4 (HHV-4), also called Epstein-Barr virus (EBV), as a member of Herpesviridae, is one of the most common viruses in humans. Epstein-Barr virus infection frequently causes mononucleosis or glandular fever in teenagers. The infection in infants and children is usually less severe than that of adults. It is commonly associated with non-specific clinical signs and is usually presented by painful sore throat, swollen glands, chills, fever and chronic fatigue syndrome. These symptoms could last for one to six weeks (1, 2).
The primary EBV infection in Africa, Southeast Asia and Latin America occurs in early childhood, while in developed countries the first peak of infection seems to be before the first five years of life while the second peak is during adolescence (3). According to epidemiological studies the average age of EBV primary infection is increasing (4), e.g. in US, it ranges from 50% for 5 year olds to 90% for 25 years old (5). In Brazil the pattern of positive EBV IgG Ab showed a higher prevalence with increasing age, reaching a peak in the second decade of life (3). In Spain, the distribution of primary infection has shown two peaks, one at the age of two to four years and another at the age of 14 - 18 years (6). A recent study of 94 children in the Republic of China demonstrated that 78.6% had EBV-VCA IgG by the end of the first year of life and 80.7% were seropositive by the age of three (7, 8). A similar study in Chile revealed that 50% of children from low and medium socioeconomic level had been primarily infected by the age of two in comparison with 5.9% of high socioeconomic children of the same age (9). In Malaysian children the presence of EBV-IgM antibody, occurred at four to six months and by eight years many children became infected with EBV (10).
Since the clinical picture of EBV primary infection could be challenging, and because this infection usually causes no symptoms and can be indistinguishable from other mild, brief infections of children such as streptococcal throat infection, it is important to use the best clinical means for diagnosis (6, 10). The routine diagnosis of EBV primary infection is based on several serological tests such as indirect fluorescent antibody (IFA), rapid monospot tests (for heterophile antibodies) and enzyme immune assay (EIA) for detection of early antigens (EA), the viral capsid antigens (VCA) or the EBV nuclear antigen (EBNA) (11-13).
2. Objectives
To the best of our knowledge, there is no study on the incidence of EBV primary infection in Iran. The aim of this study was to determine the incidence of EBV primary infection, among Iranian suspected patients who had referred to Namazi Hospital of Fars province (south of Iran) by the ELISA method.
3. Patients and Methods
3.1. Patients
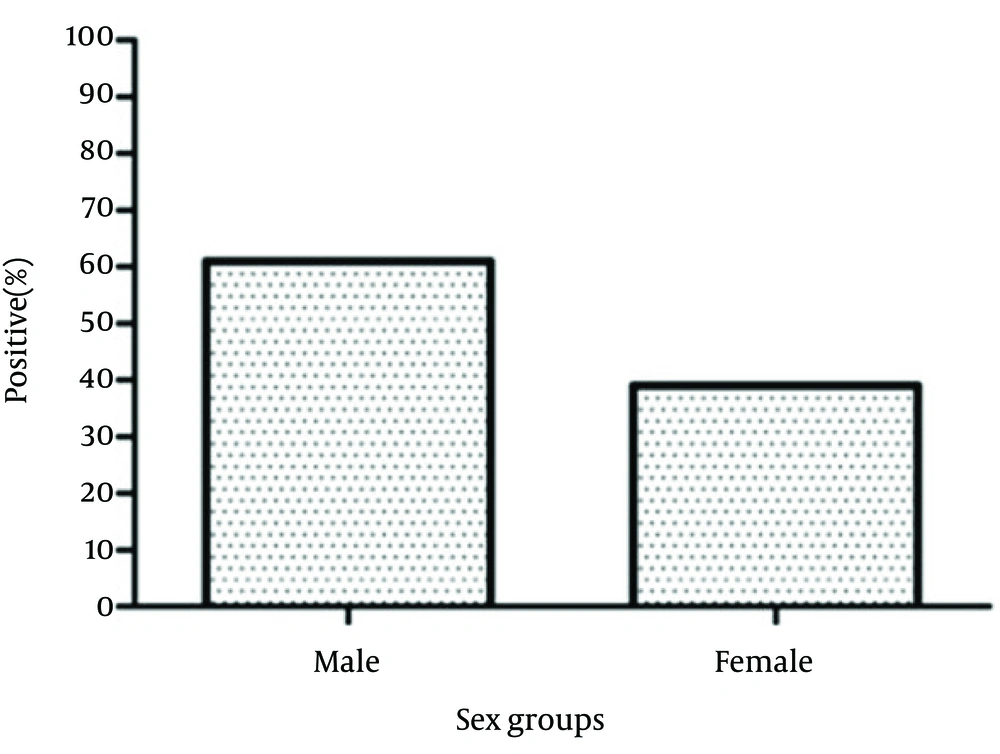
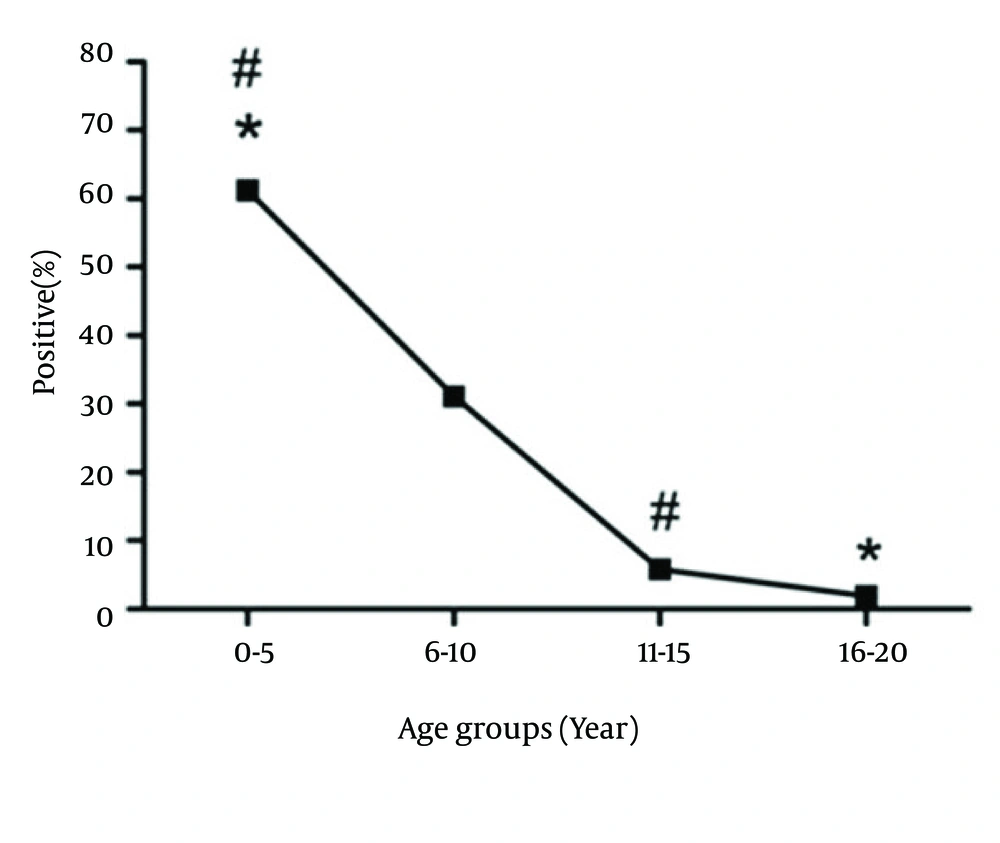
The studied population consisted of 346 suspected patients who had referred to the Professor Alborzi Clinical Microbiology Research center, Namazi Hospital, Shiraz, southern Iran, during March 2007 to March 2011. The major clinical manifestations of these patients included painful sore throat, swollen glands, chills, fever and tiredness. There were 211 males (61%) and 135 females (39%) with an age range of 0 to 20 years (6.31 ± 4.66: 10.97 years). The study population was distributed to four age groups, as follow: group I (0 - 5 years, n = 192), group II (6 - 10 years, n = 96), group III (11 - 15 years, n = 38), and group IV (16 - 20 years, n = 20).
3.2. Sample Collection and Antibody Detection
A 5 mL blood sample was collected from each individual; sera was separated and stored at -20°C for further examination. The IgM antibodies against EBV VCA were evaluated by a standard commercially available ELISA kit (Euroimmun, Lubeck, Germany). The assay was performed according to the manufacturer’s instructions.
3.3. Statistical Analysis
Descriptive analysis and Chi-square tests were used to explain the results and compare between the incidence of EBV infection in different age and sex groups. The data were analyzed by the SPSS software (SPSS for Windows, version 16, SPSS Inc., Chicago, IL, USA). P values of < 0.05 were considered statistically significant.
4. Results
Out of 346 patients, 104 (30.00%) were positive for EBV VCA-IgM, from whom 62.5% and 37.5% were males and females, respectively. There was no significance difference in the incidence of EBV primary infection between males and females (P > 0.05) (Figure 1). Our results showed that 61.5% of EBV VCA-IgM positive patients belonged to age group I, 30% to age group II, 5.8% to age group III and finally 1.9% to age group IV. Statistical analysis revealed that there were no significant differences between age group I (64/192) and II (32/96), II (32/96) and III (6/38), III (6/38) and IV (2/20) (P > 0.05). However, the incidence of EBV primary infection was significantly different between age groups I and III (P < 0.05) and also between age groups I and IV (P < 0.05) (Figure 2).
5. Discussion
Symptomatic acute EBV primary infection is clinically associated with infectious mononucleosis (IM) accompanied by fever, tonsillopharyngitis, hepatosplenomegaly, lymphadenopathy, and the increase of mononuclear blood cell number (1, 14, 15). Fever, pharyngitis and lymphadenopathy are the classic triad of IM (2, 14). In the present study, the most common symptoms of our cases included fever, temperature reaching 40°C, sore throat with and without exudates and painful cervical lymphadenopathy. However these symptoms were more or less significant between different age groups.
Since the monospot test has an approximate sensitivity and specificity of 85% and 94% for diagnosis of EBV infection, and it may be negative in about 50% of cases under age of 12 (1, 3), definitive diagnosis for EBV infection is based on VCA-IgM and VCA-IgG antibodies detection (8, 14, 16, 17). The presence of VCA-IgM in more than 80% of acute sera is the best indication for confirming EBV primary infection within four to eight weeks since infection, while after this period such indication disappears. Its shortened presence in very young children is related to the lower titer achieved (14, 17-19). In an earlier study in the United Kingdom, 92% of EBV infected patients were anti VCA-IgM positive (14, 16). Our study showed that 30% of 346 cases with typical symptomatic EBV infection were VCA-IgM positive.
Different population-based studies have reported various prevalence rates of EBV infection in children, from 20% to 80% at age two to three years in different regions. In industrialized and developed countries with high standards of living, EBV primary infection occurs mostly in adolescence (20-22). It was shown that in Malaysian children, primary infection indicated by emergence of EBV IgM antibody, occurred at four to six months of age, while all children were seropositive by age of eight (10). In another study in Japan, anti VCA-IgM positive rate in infants with acute primary infection was 25% yet 80% in those four years of age and over (23). In this study, the highest incidence of EBV primary infection belonged to the age group I (0 - 5 years) with a rate of 61.5%. This percentage decreased with age and reached 1.9 % by the age 16 - 20 years.
Previous studies declared that there is no difference in EBV seroprevalence between different genders in children (24-26). In the present study, there was also no significance difference in EBV primary infection incidence between males and females. To sum up, we can conclude that accurate and on time diagnosis of EBV primary infection in both children and adolescents will help prevent unnecessary hospitalization, medication, and incorrect medical decisions. In addition, it will decrease further treatment costs and related medical procedures. Moreover, in most cases, the best means to differentiate EBV primary infection from other infections and malignancies is by detection of IgM specific antibodies against viral capsid antigen (VCA-IgM) in the sera. Among different serological assays, the ELISA test is generally more sensitive and allows rapid diagnosis.