1. Background
Pterygium is a common benign lesion of corneo-conjunctival limbus of unknown origin (1) and the pathogenesis might be vision-threatening (2). Pterygium is found worldwide, most frequently occurring in sun-exposed areas, in particular, in tropical countries (3). It is characterized by a fleshy and wing-shaped overgrowth of the conjunctiva, which is histologically followed by degenerative and hyperplastic changes in the conjunctival epithelium (2).
Although ultraviolet irradiation, wind and dust (4), genetic predisposition, chronic inflammation (5), viral infections such as human papillomavirus (HPV), Epstein-Barr virus (EBV), herpes simplex virus (HSV) and sometimes, simultaneous detection of these viruses have been suggested as the causative factor in the development of pterygium, experiments have been inconclusive (5, 6).
Detorakis et al. proposed a “two-hit” process of pterygium formation. The first hit is preexisting ultraviolet (UV)-related genetic damage and the second hit is an oncogenic event mediated by a viral agent (5). Several studies proposed potential contribution of HPV in pterygium formation; however, the presence of HPV in pterygia varies widely from 0% to 100% in different geographic regions (7-11). Few studies reported the correlation between other viral infections and pterygia, due to low-level presence of these virus genomes in pterygia (2, 5, 12, 13).
2. Objectives
Expression of specific adenovirus genes such as E1A and E1B, which potentially have many functions, may contribute to their oncogenic activity as well as relevance to cellular immortalization. We investigated, for the first time, the presence of adenoviruses in pterygia in Iran, a country with a high prevalence of pterygium.
3. Patients and Methods
3.1. Patient Samples
This cross-sectional study was performed in the Department of Ophthalmology and Virology in Golestan University of Medical Sciences, Gorgan, Iran, between February 2011 and May 2013. The study and sampling were approved by the Ethics Approval Committee of the Faculty of Medical Sciences, Golestan University of Medical Sciences with code of practice 262790101806, in January 2011, and informed consent was obtained from patients. The study group included 50 cases of surgically excised pterygium and the group of 50 conjunctival swab samples of the same patients and 10 conjunctival biopsy specimens of individuals who were undergoing cataract surgery but exhibited no characteristics of pterygium, as controls, in 5th Azar Hospital of Gorgan, Iran. The samples stored at –70°C until processing.
3.2. Deoxyribonucleic Acid Extraction
Briefly, all sections were used for DNA extraction using a QIAamp DNA Mini kit (Qiagen, Valencia, CA). The experiments were performed according to the manufacturer’s instructions.
3.3. Polymerase Chain Reaction
First, β-globin primers were used to access the quality of extracted DNA and exclude false negative results. Then PCR amplification for the presence of adenovirus DNA was performed with primers ADHEX1F (5’-CAA CAC CTA YGA STA CAT GAA-3’; NCBI Reference Sequence: KF268198.1) and ADHEX1R (5’-KAT GGG GTA RAG CAT GTT-3’; NCBI Reference Sequence: KF268200.1; 475 bp) from the moderately conserved region (amino acids 540 to 662) of the hexon gene that are sufficient to allow HAdV speciation and in most cases, serotype identification (14, 15), in a 50 μL reaction volume, containing 5 μL extracted DNA sample, 20 pmol of each of the universal forward and reverse primers, 0.5 mM of each deoxynucleotide (dNTP; Genet Bio [A type], Nonsan, Republic of Korea), 2.5 U of Taq DNA polymerase (Genet Bio [A type]), 2 mM MgCl2 (Genet Bio [A type]) and 5 μL 10X PCR reaction buffer (Genet Bio [A type]). All standard precautions were followed to prevent PCR contamination and positive and negative controls were included with each run. Reactions were performed in a Peq Lab thermal cycler (Primus Advanced 96 thermal cycler), programmed as follows: 95°C for 5 minutes, 30 cycles of 94°C for 1 minute, 50°C for 1 minute and 68°C for 1 minute; and 72°C for 3 minutes.
3.4. Detection
PCR amplicons were visualized with 2% agarose gel electrophoresis.
4. Results
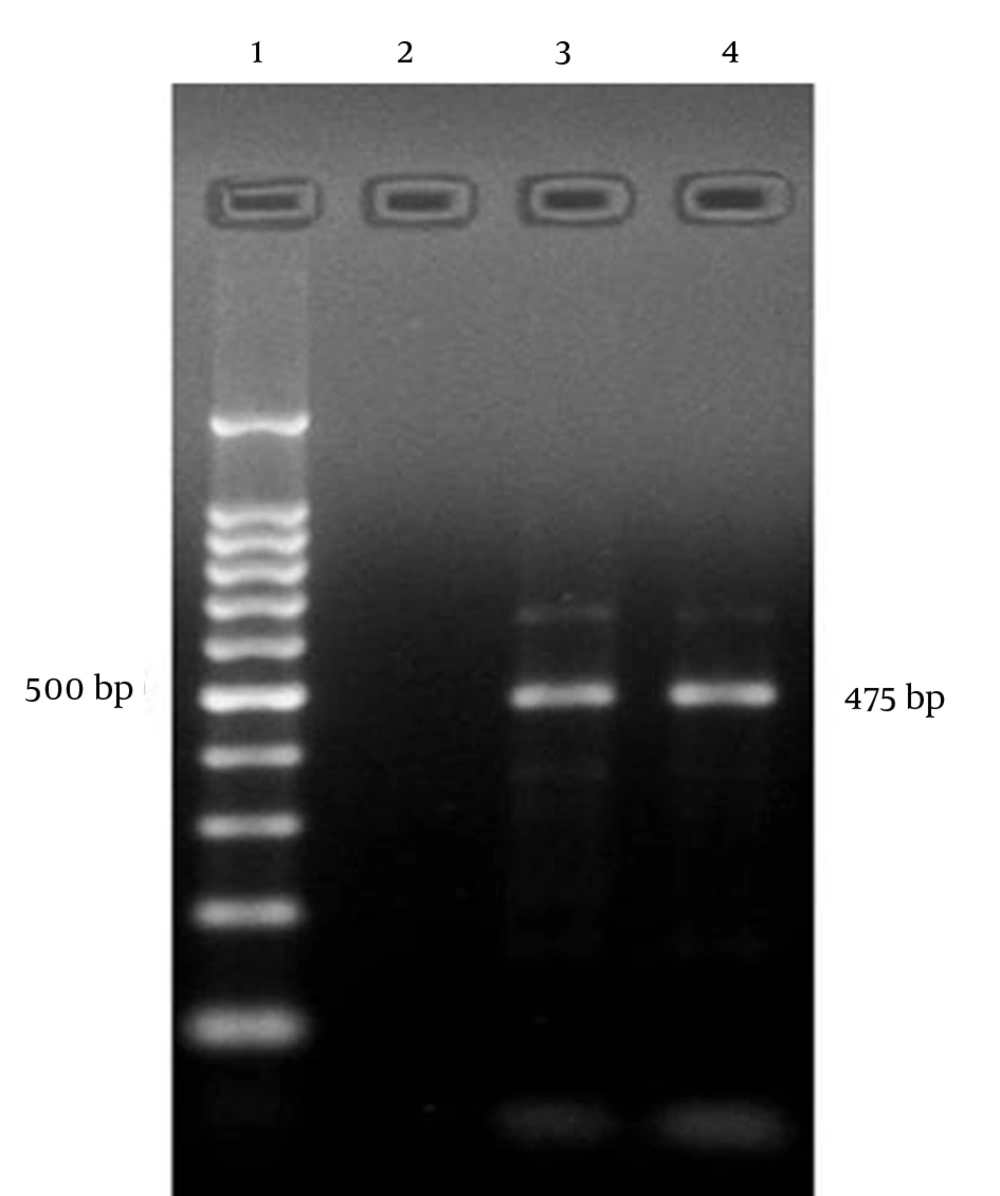
The study groups included 50 cases of surgically excised pterygium (20 men, 30 women, mean age: 61.1 ± 16.9 years, age range 22 to 85 years) (as cases) and 10 conjunctival biopsy specimens of individuals who underwent cataract surgery but exhibited no characteristics of pterygium (6 males, 4 females, mean age: 58.9 ± 15.5 years, age range: 19 to 70 years) (as control). As well, to search for persistent or latent infections with adenoviruses, 50 conjunctival swab samples from the first group were analyzed with polymerase chain reaction. All patients with pterygium had positive results for adenovirus DNA with PCR, but none of the negative control groups displayed adenoviruses (Figure 1). The pterygium groups and the control groups were β-globin positive. Direct sequencing of PCR products confirmed Adenovirus infection. Thirteen patients had bilateral pterygia. Forty-two pterygia were primary lesions and eight were postoperative recurrent. The patients were clinically evaluated to grade the severity of their pterygia; 14 (28%) were atrophic/grade 1, 17 (34%) were intermediate/grade 2 and 19 (38%) were fleshy/grade 3. A family history of pterygium was found in two cases (4%). Five positive cases (10%) had a history of outdoor activities. Smoking history was found in seven (14%) patients with pterygium. Forty-six (92%) patients with pterygium and nine (90%) individuals without pterygium (control group) had a history of red eye of unknown origin.
5. Discussion
Many theories have been advanced to explain how pterygia develops, but despite various studies, the definitive causative mechanism is still unknown (16). Uncontrolled cell proliferation, recurrence after surgical excision (up to 46% after seven years) (2, 8), abnormal levels of p53 protein (9), detection of loss of heterozygosity and presence of oncogenic viruses support the possible neoplastic nature of the lesion (1, 6). Epidemiological studies around the world have shown that pterygium prevalence rates range from 0.3% to 37.46% (17, 18) and vary according to age (5), geographic location (3, 4), inherited factors, sunlight exposure and activities with high UV exposure (19, 20).
However, stabilization, in particular, overexpression of p53 protein and disruption of the normal process of apoptosis can occur because of pterygium formation (2, 9). The tumor suppressor protein p53 has a short half-life and is normally present in low or undetectable levels within unstressed cells. p53 is essential for maintaining genomic stability after DNA damage. Because of DNA damage, p53 levels increase (21, 22). p53 induces mitotic cell cycle arrest, thus providing time for a cell to repair its DNA before the next round of replication takes place (21). Mutations in the p53 gene are believed to lead to abnormal expression.
UV radiation and oncogenic viruses have been shown to be probable mutagenic factors for the p53 gene that lead to abnormal expression in the limbal basal stem of all pterygia and limbal tumors (23). Pterygia also develops in patients without direct exposure to sunshine; thus, other factors may also participate in pterygium pathogenesis (2, 4). Our finding implies the above explanation and is in contrast with the results of most previous studies that postulate occupations or activities with high UV exposure are associated with a risk of pterygium (20, 24).
No specific evidence for a pathogenic role of the reported virus infection in pterygium has been introduced. Although recent evidence suggests that potentially oncogenic viruses, such as HPV, HSV and EBV may be involved in the pathogenesis of pterygia, the results are still inconclusive (5, 6).
Dushku et al. (11) detected increased p53 in pterygium without evidence of HPV infection, leading the authors to postulate that overexpression of p53 protein in pterygium is affected by either hereditary factors or ultraviolet radiation (2, 3, 9, 11). In contrast, a study in Taiwan found that p53 inactivation might be linked with HPV infection in pterygium (22).
However, some authors suggested that UV-B radiation may activate viruses such as herpes and human papillomaviruses (9, 25). Transforming proteins produced by adenovirus propose that the virus may act as a contributing pathogenic factor. At present, there is no way to be certain how adenoviruses are involved in the development of pterygia, but the following possible explanations might suggest the role of this virus in the occurrence of lesion:
1) Multiple adenovirus early proteins (Ad E) such as E1A, E1B and E4orf6 proteins have been shown to affect the properties and half-life of p53 protein via different mechanisms (21), thus altering the normal biologic function of the p53 protein, which leads to increased changes in the genome (21). Why adenoviruses produce such several different proteins all causing repression of the transcriptional activity of p53 that might contribute to tumorigenesis is unclear. 2) The E1A and large E1B proteins inhibit transcription stimulation and repression of transcription by p53 (21). 3) The E1A protein causes stabilization of the p53 protein. 4) The E1A protein promotes cell proliferation and inhibits differentiation (25). 5) The small E1B protein inhibits p53-mediated apoptosis (21). 6) The E3 transcription unit encodes proteins that modulate the host immune response and all appear to involve immune evasion, resulting in their long-term survival (26, 27).
Epidemiological studies indicated that clinical course of adenoviral conjunctivitis varies from unapparent infection to severe conjunctivitis. Thus, we should consider the possibility of persistent or latent infections with adenoviruses in pterygium tissues (28).
In conclusion, because our entire sample of pterygia had positive result for adenoviruses, adenoviruses are a possible factor responsible for inducing pterygium and other factors might play a synergistic role in the development process. It may help appropriate treatment of pterygia and management. Larger studies in different geographic populations are needed to clarify the role of adenoviruses in pterygium formation.