1. Background
Although urinary tract infection (UTI) is one of the most common bacterial diseases in children, its diagnosis is often delayed due to obscure clinical findings. In pediatric patients, acute pyelonephritis may be associated with high morbidity and long-term complications like renal scarring, hypertension, and chronic renal failure (1-3). Gram-negative enteric bacilli, especially Escherichia coli and Klebsiella spp., are the leading pathogens, though Enterococcus spp., yeasts, and Staphylococcus spp. have recently emerged as prominent agents.
2. Objectives
Resistance to aminoglycosides, cephalosporins, and fluoroquinolones - the most preferred agents for empiric therapy of UTIs-has increased markedly in these recent years (4-6). Therefore, knowledge of the most common uropathogens and their antibiotic susceptibility patterns in a specific geographic area can be of great help in the selection of the most appropriate agents. The objective of this study was to determine the common uropathogens in admitted patients with UTI and to assess their antibiotic susceptibility patterns.
3. Patients and Methods
This retrospective study was conducted on all urinary tract cultures performed during October 2008 to May 2011 at the Department of Pediatrics, Abuzar Children's Hospital, Ahvaz (In south west of Iran). We included all admitted pediatric patients presenting with symptomatic community acquired urinary tract infection.
3.1. Antimicrobial Susceptibility Testing
Urine samples with a colony count of > 105 (by midstream collection), > 104 (by catheter collection), and > 103 (by suprapubic collection) were included in the study (7). Cultures with growth of more than 1 organism or fungal infection and samples from patients who received prophylactic antibiotics were excluded from the study. All the samples were cultured for 24 hours at 37°C on standard media of blood agar or EMB from Merck and Himedia companies in the autoclave and standard techniques were used for culture and identification of pathogens (8).
After bacteria were identified by standard methods, antimicrobial susceptibility testing was performed by the Kirby-Baut method using a panel of antimicrobial agents, including amikacin, gentamicin, ceftriaxone, cefotaxime, cefixime, co-trimoxazole (TMP/SMX), nalidixic acid, and nitrofurantoin. After 24 h, the bacterial isolates were divided into resistant and sensitive groups according to the standard NCCLS table accompanying each disc and we excluded the intermediate reactions (9).
A questionnaire was designed to include data such as age, gender, clinical findings, lab data, and antibiotic susceptibility of bacterial isolates. All the variables were compared using Student’s t-test, Mann-Whitney U-test, and chi-square test (for quantitative and qualitative variables, respectively). Quantitative variables are provided as median (with IQR), and P values of < 0.05 were considered statistically significant.
4. Results
A total of 288 urinary specimens of different patients were included in the study. There were 62 boys (21.5%) and 226 girls (78.5%) with a median age of 13 months (range, 1 month to 14.5 years; IQR=9-40). About 72% of males with UTI were aged less than one year. In infants under the age of one year, the number of females with UTI was approximately two times more than that of males (67.1% versus 32.9%). In children above the age of one year, the female: male ratio increased to 8:1 (89.4% girls and 10.6% boys).
Fever was present in 46.9% of cases, and dysuria, vomiting, and abdominal pain were present in 44.2%, 24.8%, and 23.4%, respectively. Abnormal findings on sonography (including hydronephrosis, hydroureter, and bladder hypertrophy) were present in 19% of patients; vesicoureteral reflux (VUR) and a history of UTI were present in 26.9% and 19.8% of patients, respectively ( Table 1 ).
| Findings | Overall (n=288) | Afebrile (53.2%) | Febrile (46.8%) | P value |
|---|---|---|---|---|
| Dysuria | 56.1% | 57.2% | 54.9% | 0.69 |
| Abdominal pain | 23.4% | 23% | 25.2% | 0.68 |
| Vomiting | 24.8% | 24% | 28.4% | 0.4 |
| History of UTI | 19.8% | 20.9% | 18.5% | 0.61 |
| Leukocytosis a | 37.5% | 24.5% | 52.2% | < 0.001 |
| Anemia b | 57.9% | 51% | 65.7% | 0.012 |
| Nitrite positive | 66.7% | 71.6% | 62.8% | 0.22 |
| Decreased specific gravity c | 8% | `7.9% | 8.1% | 0.93 |
| VUR | 26.9% | 26.3% | 27.5% | 0.85 |
| Proteinuriad | 21% | 14.6% | 28.1% | 0.005 |
| Elevated ESR e | 50.5% | 27.1% | 69.2% | < 0.001 |
| Positive CRP | 60.3% | 40% | 78% | < 0.001 |
Clinical Characteristics in Patients With Urinary Tract Infection
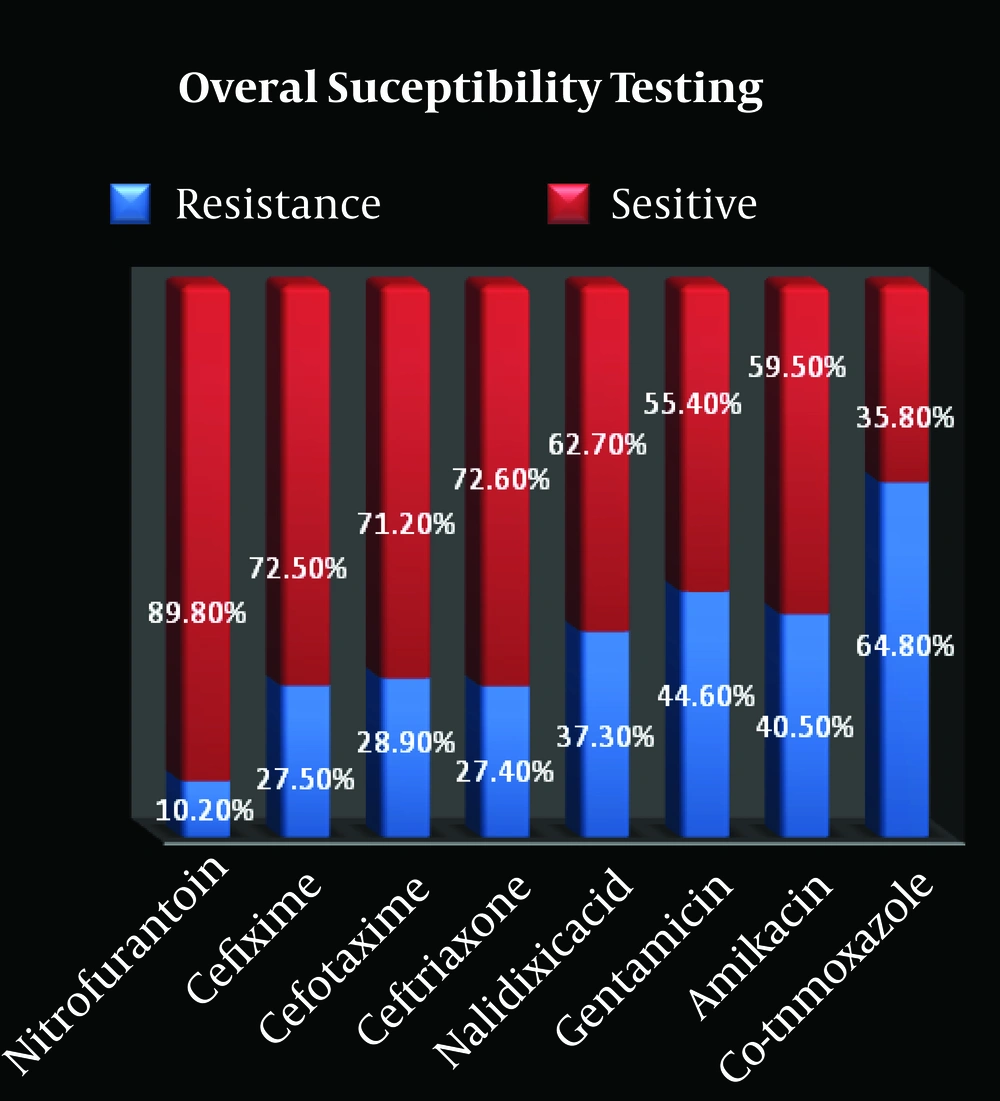
The most common pathogens were E. coli (84%), followed by Klebsiella spp. (10.1%), Enterococcus spp. (2.4%), Proteus spp. (1.7%), and Pseudomonas spp. (1.7%). Overall bacterial resistance spectrum was highest for TMP/SMX (64%) and lowest for nitrofurantoin (NF) (10.2%) (Figure). In 60 patients (21%), there was no resistance to all our antibiotics, but multidrug-resistant cultures (resistant to more than 3 antibiotics) were seen in 43 cases (15%).
Table 2 shows the resistance patterns of E. coli and Klebsiella spp. against antimicrobial agents. In patients with febrile UTI (46.8%), E. coli and Klebsiella spp. were the most common pathogens (86.3% and 8.1%, respectively). Resistance was the highest to co-trimoxazole and the lowest to nitrofurantoin. Febrile UTI was significantly correlated with anemia (P = 0.012), leukocytosis (P < 0.001), proteinuria (P = 0.005), increased erythrocyte sedimentation rate (P < 0.001) and C-reactive protein (P < 0.001), but the antibiotic resistance pattern in this group was not significantly different from that in afebrile children.
Among children infected with E. coli and Klebsiella spp. resistance to antibiotics was not correlated with age, gender, and past history of UTI (P > 0.05); however, VUR and abnormal sonography were associated with more resistance to cefotaxime (P = 0.017), ceftriaxone (P = 0.004), nitrofurantoin (P = 0.014), and nalidixic acid (P < 0.001). In boys, the mean age was 11.8 mon; 64% of them were not circumcised, and VUR was found in 37.5% (vs. 24.2% in girls). The pattern of resistance to antibiotics in boys was not significantly different from that in girls, but infection with Klebsiella spp. was more common (16.1% vs. 8.4%).
| Overall Resistance, % (n=288) | Escherichia coli (n=242) | Klebsiella spp.(n=29) | |
|---|---|---|---|
| Co-trimoxazole | 64.8 | 66 | 56 |
| Amikacin | 40.5 | 39.3 | 42.1 |
| Gentamicin | 44.6 | 44.4 | 40.7 |
| Nalidixicacid | 37.3 | 35.7 | 30.8 |
| Ceftriaxone | 27.4 | 25 | 38.1 |
| Cefotaxime | 28.9 | 25 | 44.4 |
| Cefixime | 27.5 | 22.7 | 71.4 |
| Nitrofurantoin | 10.2 | 4.9 | 24 |
Resistance patterns of Escherichia coli and Klebsiella spp
5. Discussion
UTI is always treated empirically before results of bacteriological cultures are available. The choice of antibiotics depends upon the causative organism and its local expected antibiotic susceptibility pattern. We analyzed the spectrum and resistance pattern of uropathogens to common antimicrobial agents and compared them with the results of other recent similar studies.
About 44% of all strains in our study were resistant to aminoglycosides, making these drugs unsuitable for single therapy in empirical treatment of febrile UTI. There were also high resistance rates to alternative agents such as ceftriaxone (almost 28%) and oral cefixime (almost 28%), which is frequently used for community-acquired UTI in children. The increased resistance rate to aminoglycosides and cephalosporins was presumably due to widespread use of these antibiotics and the unrestricted prescription policy within the last decade.
Resistance rates to TMP/SMX were remarkably high (almost 65%). This high resistance rate was also observed in many other studies (10-12) and may reflect a possible widespread use of this low-cost antimicrobial agent for treatment and prophylaxis of UTI or as a prophylactic agent against Pneumocystis carinii infections in recipients of immunosuppressive drugs and in the HIV-infected population. As expected, the most common pathogen was E. coli, followed by gram-negative bacteria of the Enterobacteriaceae family, which is in accordance with other studies (10, 13, 14).
Our study detected much higher resistance rates than those detected by other recent studies (15-19). The Bahadin et al. study in Singapore recently reported lower resistance rates to ceftriaxone (8.1%), nalidixic acid (35%), gentamicin (8.5%), and TMP/SMX (37.8%) among E. coli strains from Bedok hospital (15). In another Korean study performed in 2009 to evaluate antimicrobial resistance in UTI, E.coli resistance to TMP/SMX and ceftriaxone was found to be 32.7% and 5.3%, respectively. (19). The current study found that E. coli resistance to TMP/SMX and ceftriaxone was 64.8% and 27.4%, respectively; both rates are higher than those reported by the previous studies.
The resistance rate to cephalosporins was significantly higher than other new studies (15, 16, 18). The worldwide spread of extended spectrum B-lactamases (ESBLs) produced by enteric pathogens is now the most alarming problem and could explain this widespread increase in resistance against β-lactams. Antibiotic utilization patterns, including widespread cephalosporin use, have been associated with the emergence of ESBLs. More than 200 types of ESBLs have been described in various species of the Enterobacteriaceae family and other non-enteric organisms, including P. aeruginosa and Acinetobacter sp.
ESBLs hydrolyze third-generation cephalosporins and aztreonam but not carbapenems and are inhibited by clavulanic acid and tazobactam. Furthermore, many ESBL-producing pathogens also express plasmid-encoded multidrug resistance (20). Many studies worldwide have reported a noticeable increase in resistance to ciprofloxacin and other fluoroquinolones. Ciprofloxacin is one of the most frequently prescribed fluoroquinolones for UTIs in adults because it has shown excellent activity against pathogens commonly encountered in complicated UTIs (21).
A study from Iran reported that E. coli has a resistance rate of 32% to ciprofloxacin (18); other recent studies from Singapore and Korea have reported resistance rates of about 25% (15, 19). The resistance rate against nitrofurantoin was significantly low in several recent studies (15, 16). It was almost 10.2% in our study, indicating that nitrofurantoin is a suitable choice for the treatment and prophylaxis of cystitis. However, the high frequency of gastrointestinal side effects limits use of this medicine in children. In our study, urinary tract abnormalities were associated with higher frequency of drug-resistant uropathogen infections; this finding is similar to those of other studies (13, 22).
Prophylactic use of antibiotics and a history of antibiotic usage have been mentioned as other risk factors associated with antibiotic resistance (13, 14, 23). A study from the state of Wisconsin showed a high rate of resistance to third-generation cephalosporins in children receiving prophylactic antibiotics (13); in another study from Turkey, resistance rates for gentamicin and ceftriaxone were higher in patients who received antibiotic prophylaxis (14). This may be due to changes in the intestinal flora and indwelling gram-negative rods with the ability of ESBL production, which is especially important in K. pneumoniae and E. coli (12).
In our study, females constituted the majority of children with UTI (77%), with an 8:1 female to male ratio that is in agreement with previous studies (15, 24). The highest frequency of UTI in boys was observed during the first year of life (72%), but at this age, UTI was 2 times more common in girls; however, this finding may be related to the high frequency of circumcision of boys in our region.
According to our study, increasing resistance to third-generation cephalosporins changed our opinion for using them as single empiric intravenous therapy in hospitalized patients with acute pyelonephritis and in very ill patients; better success will be achieved by concomitant use of an aminoglycoside or using other potent antibiotics. On the other hand, patients with their afebrile community-acquired UTI can be treated more conventionally with oral antibiotics such as nitrofurantoin or an oral cephalosporin, particularly in view of the very low resistance of the most common pathogen E. coli to nitrofurantoin and cefixime in this study. However, restricted use of antibiotics and combination therapy may limit the increasing pattern of antibiotic resistance.