Excessive and improper use of antimicrobials (antibiotics and biocide) in hospitals and community has led to development and acquisition of bacterial resistance to antimicrobials (
1). These factors can transfer within mobile genetic elements such as conjugative plasmids, transposon, integron and chromosomes (
2,
3). Resistance to a wide range of antimicrobial agents and emergence of MDR, extreme drug resistant (XDR) and pan drug resistant (PDR) phenotypes between some nosocomial infectious agents such
Acinetobacter species in some countries especially in Iran has increased (
4-
6) During the last three decades,
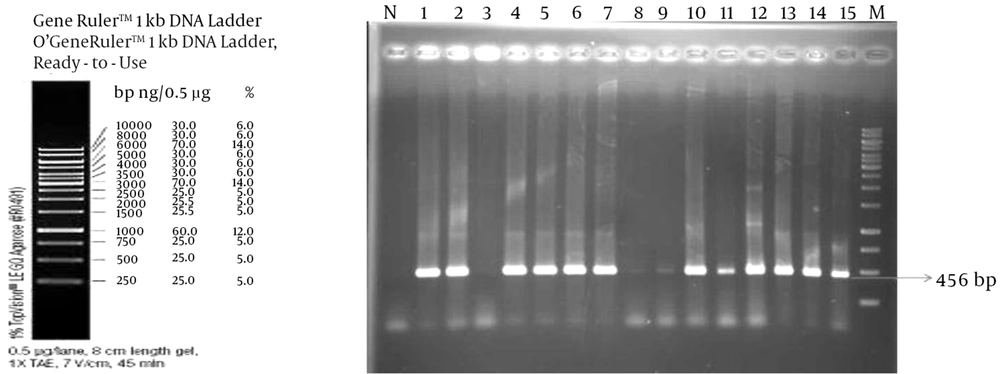
Acinetobacter species are detected accompanied by beta-lactamase encoded enzymes, bla
TEM, bla
CTX and New-Delhe-metallo-B-lactamase-1 from a wide variety of environments (
7).
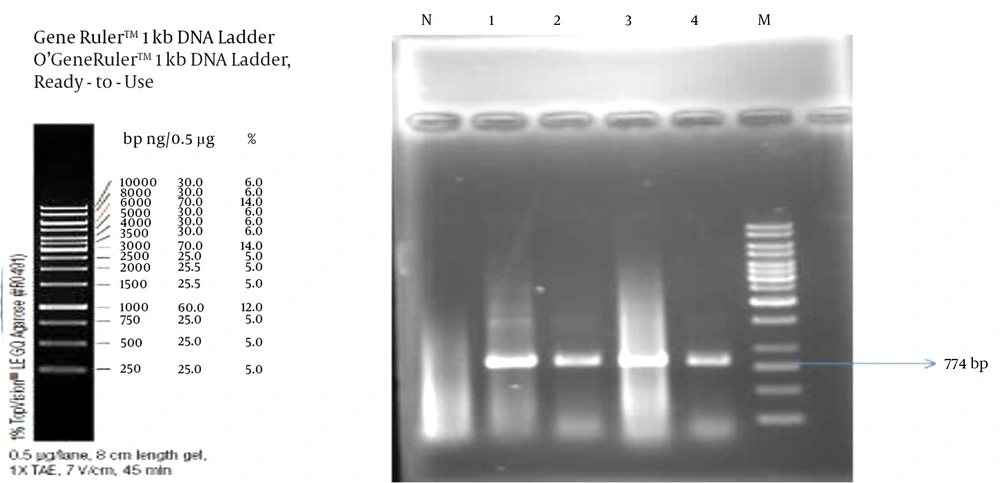
Also the presence of OXA-48, OXA-181, CTX-M-15, SHV, blaoxA-23,24,51, bla
CTX (oxA-10,2), TEM (bla
TEM-1,2,13), methylase, ESBL, qnr and six types of NDM
5 (1-2-3-4-5-6), disinfectant resistance genes, quc (A-E) (
8) and detection of new plasmids such as PNDm-1-DOK01 encoding different types of outer membrane proteins (OMPA), siderophores and iron acquisition systems (
9), presence of pili mediated DNA uptake and biofilm formation by fimbria and exopolysaccharides on biotic and abiotic surfaces convert the
Acinetobacter baumannii to a super bug (
10,
11). Authors were eager to study
A. baumannii isolates collected from hospital ICU environments and medical equipment surfaces in Tehran. The frequency of bla
TEM and bla
CTX resistance genes was evaluated by molecular techniques among the isolates. In the next step, the susceptibility of
A. baumannii isolates to 2% glutaraldehyde solution was determined.