1. Background
Probiotics are defined as living microorganisms that confer various health benefits and promote or support a beneficial balance of the autochthonous microbial population of the gastrointestinal tract (1). Most probiotic organisms are lactobacilli or bifidobacteria, which are normal inhabitants of the human colon. These human commensal bacteria are thought to be better at adapting to survive the highly stressful environment of gastrointestinal tracts and can adhere to the human epithelial wall of intestines much better than those from other sources. Therefore, they are extensively studied and preferred as commercial probiotics (2). However, a small number of commensal Lactic Acid Bacteria (LAB) and bifidobacteria have been reported to be potential probiotics. Only a few strains of LAB isolated from the feces of breast-fed infants have shown high resistance to gastric juice and bile as well as high adherence ability (3). The strains reported to be beneficial to health so far, have been confined to a few species of lactobacilli and bifidobacteria. Probiotic properties are mostly strain specific.
Although LAB and bifidobacteria are well recognized as antibacterial agent producers (4-6), the majority cannot always play antagonistic roles under gut conditions. Protective mechanisms against pathogens in the gut are brought about through competition of binding sites and nutrients, immune modulation and antibacterial secretion (7). The adhesion property of probiotics ensures their ability to colonize in the host gut that prolongs enough transit time to prevent any pathogens from adhering to mucin and intestinal epithelial cells (8). Therefore, potential probiotics that produce antibacterial agents must be able to adhere and colonize mucosal surfaces, apart from having the ability to survive extreme gastric acid and bile present in the stomach and intestinal conditions, respectively.
Antagonistic interaction with various food-borne pathogenic bacteria was previously reported to be species and strain specific (9). Borriello et al. (10) suggested that the ideal bacteria applied for the development of novel probiotics should be isolated from the human fecal microflora of healthy volunteers. For ecological reasons, the bacteria selected for probiotic use in humans must be of human origin (11). These bacteria may have a better chance to out-compete intestinal flora to become successfully established in their new host at a significant level. Several probiotic bacteria that are now being exploited commercially are mostly isolated from humans. For example, Lactobacillus rhamnosus GG (ATCC 53103) (12) was originated from human feces. Lactobacillus acidophilus LA-1 (12), and Lactobacillus reuteri strains mm4-1a (ATCCPTA 6475) and fj1 (ATCCPTA 5289) were isolated from human breast milk and human oral cavity (13), respectively.
2. Objectives
The aim of this research was to study the antagonistic interactions of LAB and bifidobacteria isolated from Thai healthy infant feces against various food-borne pathogens through investigating antibacterial secretion, mucin adhesion and competitive mucin adhesion.
3. Materials and Methods
3.1. Ethics Statement
This study was reviewed and approved by the Ethics Committee of the Faculty of Medicine, Prince of Songkla University (EC Number: 51/400 - 020). All infants’ parents provided verbal informed consent on behalf of the participating infant. The verbal consent was granted by the Ethics Committee because this research presented negligible risks to the participants. Besides, they were not identifiable from the data collected. To obtain written consent or record, the isolation process could be delayed leading to a long period of oxygen exposure of fecal samples. This would seriously cause the destruction of the oxygen-sensitive bacteria indigenous to the human colon.
3.2. Isolation of Lactic Acid Bacteria and Bifidobacteria From Infant Feces
Lactic Acid Bacteria and bifidobacteria were isolated from stool samples collected from twenty-five Thai healthy infants who had no history of antibiotic treatment. Ten-fold dilution of fecal samples was then serially performed. The appropriate dilution was then plated with molten modified de Man Rogosa and Sharpe agar (MRS; Himedia, India) containing 0.05 % L-cysteine (Sigma, Japan) and 0.004% bromocresol purple (Labchem, Australia). These plates were incubated at 37°C for 48 - 72 hours in an anaerobic jar with a disposable BBL gas pack (USA). Presumptive colonies of LAB and bifidobacteria were picked. Colonies exhibiting the presumptive characteristics of LAB (rod or short rod or cocci) and bifidobacteria (bifid), which were Gram-positive and catalase negative, were subcultured in L-cysteine containing de Man Rogosa and Sharpe (MRS) broth. They were then stored with 30% glycerol in liquid nitrogen (-196°C).
3.3. Bacterial Strains and Cultivation Conditions
Lactic acid bacteria were cultivated routinely in MRS broth (Himedia, India) at 37°C for 24 - 48 hours. Bifidobacteria were anaerobically cultivated in modified MRS broth containing 0.05% L-cysteine and 1 mg l-1 resazurin (Sigma, US) in an injection vial. Escherichia coli TISTR 780 and Staphylococcus aureus TISTR 1466 were obtained from the Microbiological Resources Centre (MIRCEN), Thailand. All patient isolates of Shigella sonnei, S. flexneri, Salmonella enterica subsp. enteric serovar typhimurium SA2093 and S. Paratyphi A were obtained directly from Microbiological Laboratory of Songklanakarind Hospital, Prince of Songkla University, Hat Yai (Songkhla, Thailand). These pathogens were cultivated in Muller Hinton broth (MHB; Himedia, India) at 37°C for 24 hours.
3.4. Survival of Probiotic Bacteria in the Upper Gastrointestinal Conditions
3.4.1. Tolerance to Bile
Lactic acid bacteria (313 strains) and bifidobacteria (17 strains) were cultivated in MRS broth as mentioned above. A bile tolerance assay was performed in accordance with the method of Vinderola and Reinheimer (14) with some modifications. Overnight LAB or bifidobacterial culture was serially diluted in a ten-fold manner. One milliliter of the appropriate dilution was plated onto a sterilized plate, onto which MRS agar with or without 0.3% (w/v) bile salt was poured. The plates were incubated at 37°C for 24 - 72 hours. The ability of the bacteria to grow in 0.3% bile salt was expressed as percentage survival, which was calculated using the equation below:



3.4.2. Tolerance to Simulated Gastric Juice
The overnight cultures of LAB and bifidobacteria showing high tolerance to bile salt were cultivated in MRS broth as mentioned above. One milliliter of the culture broth was centrifuged at 4°C, 10000 rpm for 10 minutes. The cell pellet was washed twice in PBS buffer and then resuspended in 1 mL of gastric juice (2 g l-1 NaCl containing 3.2 g l-1 pepsin (Sigma-Aldrich, USA) adjusted to pH 2.0 with 0.5 M HCl) (15). The mixture was incubated at 37°C for three hours with gentle agitation. Total viable counts were performed before (log N0) and after (log N1) acid exposure by the pour plate method with 0.02% bromocresol purple containing MRS agar for LAB and by using the Live/Dead BaclightTH Bacterial Viability Kits (Eugene, Oregon, USA). The percentage of acid survival was calculated according to the following equation:

3.4.3. Sequential Exposures of Lactic Acid Bacteria and Bifidobacteria to Gastric Acid and Bile
The strains that presented > 80% survival in acidic conditions were selected for sequential exposure to the simulated gastro-intestinal (GI) tract passage test. They were firstly exposed to gastric acid for three hours as described above and were then resuspended in 0.3% bile salt containing 3 mg mL-1 pancreatin. The test tubes were then incubated at 37°C for six hours with gentle agitation. After that, the aliquot was then serially diluted and the appropriate samples were plated in MRS agar or Live/Dead BaclightTH Bacterial Viability kits in order to count the survived cells (N1) of LAB or bifidobacteria, respectively. The results were expressed as percentage survival by comparison with the total viable count before exposure to the gastrointestinal transit conditions (N0). The survival percentage was calculated according to the equation displayed above. The experiments were carried out in triplicates.
3.5. Analysis of 16S rDNA Sequences for Identification of Lactic Acid Bacteria and Bifidobacteria
Bacterial DNA was extracted, and the 16S rDNA gene was amplified by Polymerase Chain Reaction (PCR) carried out in a thermal cycler (PerkinElmer 2400, USA). The primers of 27-f and 1492-r were used to generate 1465 bp of PCR product, corresponding to 16S rDNA nucleotide (position 27 - 1492) of LAB. For bifidobacterial DNA, Im26-f and Im3-r were used as the primers for amplification of 1417 bp 16S rDNA fragment (16). The PCR products were purified using the NucleoSpin® Extract II (Germany), according to the manufacturer’s instructions. The purified PCR products were sequenced by BioDesign Co., Ltd. (Bangkok, Thailand). The nucleotide sequences of the selected strains were compared with sequence data deposited on GenBank, using the BLAST search program (available at http://www.ncbi.nlm.nih.gov/). Sequence alignment between the selected probiotic bacteria and the reference strains was done using the Clustal X program (2.1.12). A phylogenetic tree was constructed using the Mega 5 program.
3.6. Antibacterial Activity Assay
Antibacterial activity against food-borne pathogens was performed using a broth microdilution assay in a 96-well plate according to Kongnum and Hongpattarakere (17). The cell-free culture supernatant of LAB or bifidobacteria was treated as follows: (i) the supernatant without any treatment; (ii) the supernatant neutralized to pH 6.5 - 7.0 using 1 M NaOH; and (iii) the supernatant adjusted to pH 6.5 - 7.0 and treated with 200 unit mL-1 of catalase (Sigma-Aldrich, USA) (18). The activity was expressed as an Arbitrary Unit (AU) per mL estimated from the reciprocal of the highest dilution of the culture supernatant that did not give visible growth of bacteria. Arbitrary Unit mL-1 was calculated according to (1000/100) D, whereas D was the dilution factor.
3.7. Adhesion Assay
The partially purified porcine gastric mucin type III (Sigma, USA) was coated on a sterilized polystyrene microtiter plate well (Maxisorp Nunc, Denmark) before the adhesion assay was performed according to Tallon et al. (19). A minimum of four replicates was used to estimate the adhesion of the tested strain. Lactobacillus plantarum 299V, a well-known adhesive strain, was used as a positive control. Briefly, 100 µL of probiotic suspension (107 CFU mL-1) was added to each mucin coated well before incubation at 37°C for one hour. The wells were washed twice with sterile Phosphate Buffered Saline (PBS) (200 µL per well) to remove unbound bacteria and then 200 µL of a 0.05% (v/v) Triton X-100 solution (Sigma-Aldrich, Sigapore) was added. The plates were then incubated for two hours at room temperature under gentle agitation to dislodge the bound bacteria (NAdhere). The viable cells before (NInitial) and after (NAdhere) mucin absorption were enumerated by plating the appropriate dilution in MRS agar. The percentage of adhesion was calculated according to the following equation:

3.8. Determination of Competitive Exclusion of Food-Borne Pathogens
Fifty microliters of pathogen suspension (107 CFU mL-1) was mixed with 50 µL of probiotics (107 CFU mL-1) and added to the mucin coated plates to perform the adhesion assay described above. The treatment of pathogen alone was performed in parallel (50 µL of pathogen suspension + 50 µL of PBS). Viable counts of pathogens before and after adhesion were serially diluted and counted on Mueller-Hinton Agar (MHA) plates. The adhesion percentages of both treatments (with and without probiotic) were calculated, as mentioned above. The probiotic inhibition of the adhesion of pathogen to mucin was expressed as percentage competitive exclusion, which was calculated from the difference between the adhesion percentage of the pathogens in the absence and presence of the probiotic strains.
3.9. Statistical Analysis
Statistical significances of data obtained from the assays for acid and bile resistances, adhesion and competitive exclusion were evaluated using one-way analysis of variance and Duncan’s multiple range tests. Significant differences were indicated by P < 0.05. The data were analyzed using the SPSS software version 15 for Windows.
4. Results
4.1. Survivals of Lactic Acid Bacteria and Bifidobacteria Through the Upper Part of the Gastrointestinal Tract
Among 313 strains of LAB, one hundred and eighty six strains showed survival in the presence of bile. Meanwhile, no growth was observed in the other strains. However, only fifty strains showed a high survival rate of > 80 %. Eight out of 17 strains of bifidobacteria showed survival in MRS broth containing 0.3% ox-gall bile, whereas the rest completely lost their viability (data not shown). Among the fifty bile tolerant LAB strains and eight strains of bifidobacteria, the majority showed high tolerance to gastric acid. Only two strains of LAB and three strains of bifidobacteria did not survive gastric acid exposure. Therefore, only 11 strains of LAB and five strains of bifidobacteria with high survival rates were chosen to test survival after sequential exposure to the simulated upper part of the human gastrointestinal tract.
These LAB and bifidobacteria showed high viability after exposure to the simulated gastric conditions for three hours with survival rates ranging from 81.52 - 111.11% (Table 1). However, significant survival reduction was observed in LAB strains CIF1A1, CIF1A2 and CIF1A9 after sequential exposure to simulated gastric acid and bile for three and six hours, respectively. Strain CIF1A10 could not survive such sequential exposure. Only seven LAB strains showed survival above 70% with the log reduction ranging between 1.84 and 2.52 log CFU mL-1. The strain CIF17AN8 presented the highest tolerance with 1.84 log reduction. All strains of bifidobacteria displayed higher survival rates, from sequential exposure, compared to LAB. The strain NIF7AN2 was the most tolerant strain with only 0.33 log reduction.
| Strains | Sequential Acid (3 h) and Bile (6 h) Exposures | ||||
|---|---|---|---|---|---|
| Initial (Log CFU mL-1) or (Log Cell mL-1) b | Acid Exposure (Log CFU mL-1) or (Log Cell mL-1) b | % Survival b,c | Bile Exposure (Log CFU mL-1) or (Log Cell mL-1) b | Survival, % b,c | |
| Lactic acid bacteria | |||||
| L. rhamnosus CIF1A1 | 8.88 ± 0.10 | 8.30 ± 0.11 | 93.47 ± 1.22 | 1.31 ± 0.04 | 14.80 ± 0.48 |
| L. rhamnosus CIF1A2 | 8.73 ± 0.08 | 8.58 ± 0.19 | 98.33 ± 2.16 | 1.61 ± 0.03 | 18.39 ± 0.31 |
| L. rhamnosus CIF1A9 | 8.54 ± 0.55 | 6.84 ± 0.11 | 80.08 ± 1.26 | 3.77 ± 0.06 | 44.17 ± 0.64 |
| L. rhamnosus CIF1A10 | 8.78 ± 0.10 | 8.42 ± 0.07 | 95.91 ± 0.78 | - | - |
| L. casei NIF1A7 | 9.24 ± 0.14 | 7.88 ± 0.59 | 85.22 ± 6.34 | 6.74 ± 0.04 | 72.94 ± 0.40 |
| L. casei NIF1AN12 | 8.36 ± 0.51 | 8.25 ± 0.03 | 98.67 ± 0.39 | 6.09 ± 0.20 | 72.79 ± 2.43 |
| L. plantarum CIF17A2 | 9.19 ± 0.01 | 8.18 ± 0.14 | 89.03 ± 1.48 | 6.86 ± 0.18 | 74.62 ± 1.95 |
| L. plantarum CIF17A4 | 9.17 ± 0.07 | 8.52 ± 0.02 | 92.89 ± 0.18 | 6.90 ± 0.14 | 75.22 ± 1.52 |
| L. plantarum CIF17A5 | 9.29 ± 0.08 | 8.10 ± 0.03 | 87.26 ± 0.30 | 6.77 ± 0.39 | 72.85 ± 4.21 |
| L. plantarum CIF17AN2 | 9.17 ± 0.12 | 8.31 ± 0.06 | 90.54 ± 0.65 | 7.14 ± 0.08 | 77.87 ± 0.85 |
| L. plantarum CIF17AN8 | 9.21 ± 0.03 | 8.31 ± 0.02 | 90.27 ± 0.22 | 7.37 ± 0.02 | 80.03 ± 0.24 |
| Bifidobacteria | |||||
| B. longum NIF3AN3 | 7.68 ± 0.05 | 7.29 ± 0.07 | 94.98 ± 0.22 | 6.79 ± 0.09 | 88.45 ± 2.22 |
| B. longum NIF7AN2 | 8.01 ± 0.01 | 7.45 ± 0.03 | 93.04 ± 01.20 | 7.68 ± 0.20 | 95.84 ± 2.14 |
| B. bifidum NIF7AN3 | 7.63 ± 0.013 | 6.67 ± 0.028 | 87.36 ± 2.10 | 7.09 ± 0.11 | 92.94 ± 0.82 |
| B. bifidum NIF7AN5 | 8.14 ± 0.03 | 6.60 ± 0.05 | 81.15 ± 0.82 | 7.08 ± 0.09 | 86.97 ± 0.88 |
| B. bifidum NIF7AN10 | 7.70 ± 0.12 | 6.33 ± 0.25 | 82.27 ± 0.42 | 6.89 ± 0.16 | 89.52 ± 0.64 |
a Abbreviation: CFU, colony forming unit.
b Results are mean values of the triple determinations ± Standard Deviation (SD).
c Different letters within the same column indicate significant difference (P < 0.05).
4.2. Probiotic Identification and Phylogenetic Analysis
According to the 16S rDNA nucleotide sequence analysis, high acid and bile tolerant LAB strains were: L. rhamnosus (CIF1A1, CIF1A2, CIF1A9 and CIF1A10); L. casei (NIF1A7 and NIF1AN12) and L. plantarum (CIF17A2, CIF17A4, CIF17A5, CIF17AN2 and CIF17AN8). They exhibited 99% homology with the type trains of L. rhamnosus JCM1136T, L. rhamnosus ATCC8530, L. casei LC2W, and L. plantarum WCFS1, respectively. The bifidobacterial strains NIF3AN3 and NIF7AN2 showed 99% homology with the type strain B. longum subsp. longum JCM1207. The strains NIF7AN3, NIF7AN5 and NIF7AN19 showed 99% homology with type strain B. bifidum YIT4039. The phylogenetic trees of the 16S rRNA genes from the selected probiotic LAB and bifidobacteria were constructed using the neighbor-joining method (Figure 1A, 1B). Phylogenetic tree analysis revealed that the bacterial strains within each species had a close relationship with the type strains from a human origin.
4.3. Antimicrobial Secretions of the Probiotic Lactic Acid Bacteria and Bifidobacteria
All eleven strains of LAB showed antimicrobial activity against all tested pathogens with activity values of 20 - 80 AU mL-1 (Table 2). However, the majority lost such activity against either Gram-positive or Gram-negative food-borne pathogens after neutralization of supernatants (data not shown). This indicated that the antimicrobial activities of these strains were influenced by acid. Contrarily, the neutralized supernatants from L. plantarum CIF17AN2 and CIF17AN8 retained inhibitory activity against all tested strains. A loss of inhibitory activity was also observed in the 200-unit mg-1 catalase-treated supernatant. This indicated that production of both acid and H2O2 played an important role in pathogenic inhibition of strains CIF17AN2 and CIF17AN8. Bifidobacteria showed a lower bacteriostatic effect (Minimum Inhibitory Concentration (MIC) of 10 - 20 AU mL-1) than LAB against most pathogens (Table 2).
| Test Strains | Inhibition Activity (AU mL-1) | |||||
|---|---|---|---|---|---|---|
| E. coli TISTR 780 | S. sonnei | S. flexneri | Sal. Paratyphi A | Sal. Typhimurium SA2093 | S. aureus TISTR 1466 | |
| Lactic acid bacteria | ||||||
| L. rhamnosus CIF1A1 (pH4.04) | 40 | 40 | 40 | 40 | 40 | 40 |
| L. rhamnosus CIF1A2 (pH4.05) CFCS | 40 | 40 | 40 | 40 | 40 | 40 |
| L. rhamnosus CIF1A9 (pH4.06) CFCS | 40 | 20 | 20 | 20 | 20 | 40 |
| L. rhamnosus CIF1A10 (pH3.9) CFCS | 80 | 40 | 40 | 40 | 40 | 80 |
| L. casei NIF1A7 (pH 3.9) CFCS | 20 | 20 | 20 | 20 | 20 | 20 |
| L. casei NIF1AN12 (pH4.04) CFCS | 20 | 20 | 20 | 20 | 20 | 20 |
| L. plantarum CIF17A2 (pH3.9) CFCS | 20 | 20 | 20 | 20 | 40 | 20 |
| L. plantarum CIF17A4 (pH3.9) CFCS | 20 | 20 | 20 | 20 | 40 | 20 |
| L. plantarum CIF17A5 (pH3.9) CFCS | 20 | 20 | 20 | 20 | 40 | 20 |
| L. plantarum CIF17AN2 (pH3.9) CFCS | 20 | 40 | 40 | 40 | 40 | 20 |
| L. plantarum CIF17AN8 (pH3.9) CFCS | 20 | 40 | 40 | 40 | 40 | 20 |
| Bifidobacteria | ||||||
| B. longum subsp. longumNIF3AN3 (pH4.25) CFCS | 20 | 20 | 20 | 20 | 80 | 20 |
| B. longum subsp. longumNIF7AN1 (pH4.41) CFCS | 10 | 10 | 10 | 10 | 80 | 10 |
| B. bifidum NIF7AN2 (pH4.62) CFCS | 10 | 10 | 10 | 10 | 80 | 10 |
| B. bifidum NIF7AN5 (pH4.65) CFCS | 10 | 10 | 10 | 10 | 80 | 10 |
| B. bifidum NIF7AN10 (pH4.25) | 20 | 20 | 20 | 20 | 80 | 20 |
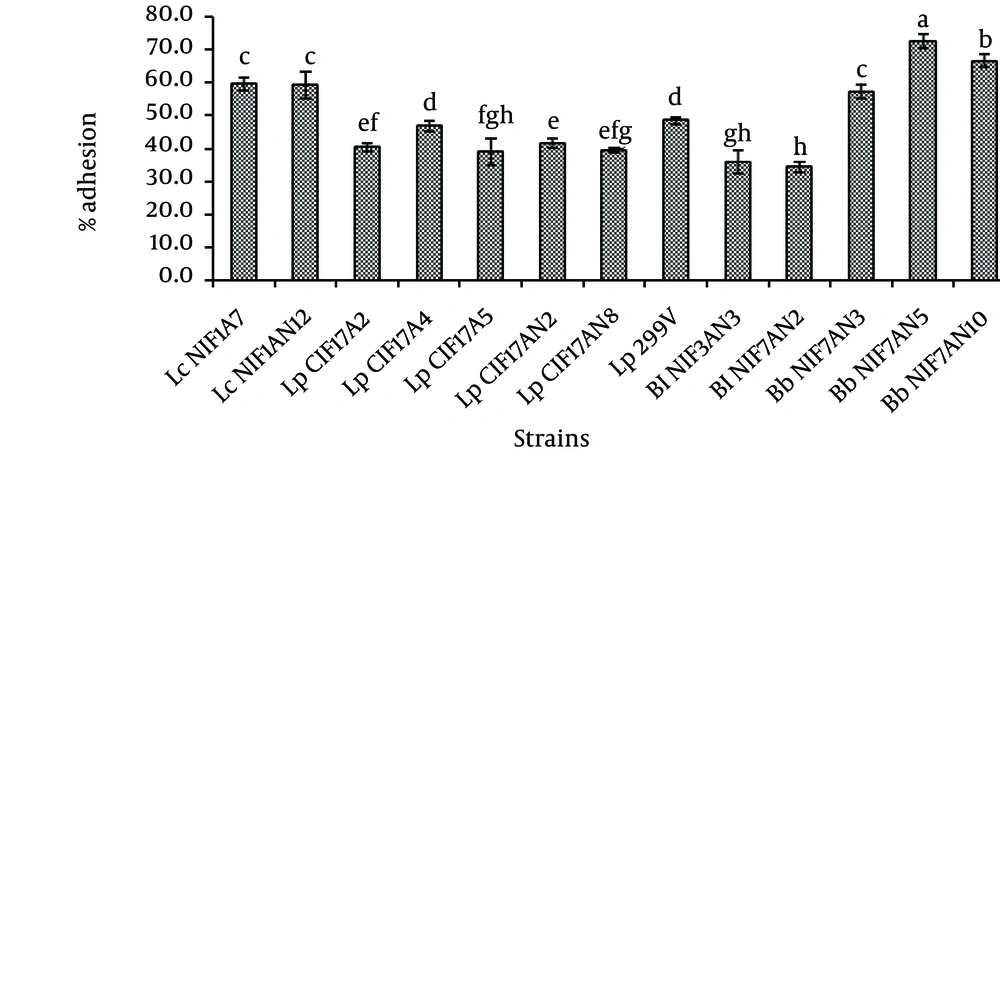
4.4. Adhesion Assay
The tested strains exhibited a much wider range of adhesion ability to mucin (Figure 2). The average adhesion values ranged from 34.31% to 72.54% for the tested strains and 48.49% for the positive control of L. plantarum 299V. In LAB strains, L. casei NIF1A7 (59.66 %) and NIF1AN12 (59.23 %) adhered significantly (P < 0.05) more to mucin than the positive control. Lactobacillus plantarum CIF17A2, CIF17A4, CIF17A5, CIF17AN2 and CIF17AN8 adhered less than the control. On the contrary, all L. rhamnosus (CIF1A1, CIF1A2, CIF1A9 and CIF1A10) lacked adhesion ability to porcine mucin. Bifidobacterium bifidum NIF7AN3, B. bifidum NIF7AN5 and B. bifidum NIF7AN10 possessed higher adhesion ability than the positive control L. plantarum 299V. Lactobacillus casei NIF1A7 showed significantly greater competitive adhesion against Sal. Paratyphi A when compared to L. plantarum 299V (Table 3). Lactobacillus plantarum CIF17AN2 and L. plantarum CIF17AN8 exhibited strong competitive adhesion against S. typhimurium SA2093. Meanwhile, all bifidobacteria were highly competitive in mucin adhesion against all tested pathogens. The human isolated bifidobacteria in this study showed higher adhesion ability and competitive adhesion against food-borne pathogens than LAB.
| Strains | Competitive Exclusion, % | |||||
|---|---|---|---|---|---|---|
| E. coli TISTR 780 | S. sonnei | S. flexneri | Sal. Paratyphi A | Sal. Typhimurium SA2093 | S. aureus TISTR 1466 | |
| Lactic acid bacteria | ||||||
| L. casei NIF1A7 | 1.92 ± 2.59 | -1.49 ± 3.14 | 3.91 ± 1.40 | 13.47 ± 3.08 | 12.74 ± 5.15 | 10.93 ± 2.90 |
| L. casei NIF1AN12 | 1.26 ± 4.93 | 2.09 ± 3.33 | 0.37 ± 0.00 | 4.09 ± 2.63 | 8.20 ± 3.07 | 5.85 ± 2.52 |
| L. plantarum CIF17A2 | -7.98 ± 0.97 | 6.75 ± 0.00 | 2.81 ± 2.53 | 3.17 ± 1.45 | 5.89 ± 2.44 | 4.92 ± 1.24 |
| L. plantarum CIF17A4 | -4.01 ± 3.19 | 3.43 ± 2.87 | 0.37 ± 0.00 | 6.66 ± 0.15 | 9.89 ± 3.99 | 5.70 ± 0.62 |
| L. plantarum CIF17A5 | -3.33 ± 3.66 | 3.43 ± 2.87 | 2.05 ± 2.91 | 5.00 ± 3.03 | 7.96 ± 1.47 | 5.29 ± 0.68 |
| L. plantarum CIF17AN2 | -10.20 ± 3.22 | -3.58 ± 1.58 | 5.41 ± 0.00 | 6.93 ± 3.16 | 16.52 ± 1.99 | 5.78 ± 1.88 |
| L. plantarum CIF17AN8 | -10.45 ± 0.95 | 2.26 ± 4.27 | 2.81 ± 2.53 | 6.66 ± 0.15 | 16.16 ± 2.45 | 5.30 ± 0.93 |
| L. plantarum 299V | 1.88 ± 6.42 | 2.37 ± 2.58 | -1.16 ± 1.85 | 6.92 ± 3.27 | 7.54 ± 1.65 | 6.09 ± 2.79 |
| Bifidobacteria | ||||||
| B. longum subsp. longum NIF3AN3 | 43.81 ± 4.42 | 35.33 ± 1.24 | 34.31 ± 2.40 | 18.49 ± 0.12 | 1.16 ± 0.32 | 14.37 ± 2.82 |
| B. longum subsp. longum NIF7AN2 | 40.74 ± 1.87 | 34.38 ± 1.99 | 34.80 ± 2.94 | 17.78 ± 2.01 | 30.51 ± 8.41 | 37.96 ± 1.78 |
| B. bifidum NIF7AN3 | 36.69 ± 0.64 | 35.39 ± 1.87 | 38.46 ± 0.60 | 13.35 ± 0.55 | 22.86 ± 6.62 | 23.59 ± 0.49 |
| B. bifidum NIF7AN5 | 31.71 ± 1.44 | 33.67 ± 0.37 | 32.49 ± 2.04 | 35.33 ± 2.96 | 2.14 ± 3.44 | 22.51 ± 0.00 |
| B. bifidum NIF7AN10 | 41.60 ± 3.44 | 50.55 ± 2.50 | 34.06 ± 0.38 | 38.69 ± 2.14 | 29.86 ± 10.30 | 27.79 ± 1.25 |
a A higher value represents high inhibition of probiotic strains against adhesion of food borne pathogens, whereas a lower value indicates less inhibitory ability of probiotic strains.
b Different letters within the same column indicate significant difference (P < 0.05).
5. Discussion
In order to survive and colonize in the human gastrointestinal tract, probiotic bacteria should express high resistance to low acid in the stomach and high bile content in the small intestine. To ensure that the bacteria can establish, colonize and persist in the colonic habitat, they must not only tolerate bile exposure but also must be able to multiply in the presence of bile. Therefore, such ability was evaluated and considered as the first important requirement to select probiotic bacteria in this study. Bile salt tolerance is considered as one of the most essential properties required for all probiotics to survive in the small intestine (9, 20, 21). This study isolated 330 strains of LAB and bifidobacteria, which had previously passed through the environmental extremes of the gastrointestinal tracts of infants. Nevertheless, the bile tolerance of fecal LAB and bifidobacteria isolated from infant’s feces varied greatly, and about 43% of the total strains were unable to grow in the presence of 0.3% bile salt. Although many fecal strains lacked the ability to grow in bile-containing media, they could be isolated and recovered from the feces. The results correlated with previous findings regarding the variation of bile tolerance among the same species of enteric L. acidophilus (22). The same author also showed that a significant number of lactobacilli increased in calf jejunum when bile resistant strains were fed compared to bile sensitive ones.
Similarly, some strains of LAB and bifidobacteria were not able to survive the extreme acidic conditions of gastric juice in vitro, even though they previously survived infant gastrointestinal transit. This observation supports the idea that the gastric transit tolerance assayed in vitro may not always provide the absolute prediction about the in vivo behavior of probiotics. Nevertheless, the higher resistant strains evaluated in vitro would have a better chance to survive extreme conditions in the stomach in vivo (23). This is because the environmental conditions in the human gut are heterogeneous and dynamic to a high degree. The fluctuations of acid secretion capacity of the stomach, rates of gastric mobility and emptying gut content, and physical properties of the ingested food could provide protective effects against bacteria (20). The presence of food was reported to raise the pH level of the stomach from 1.5 - 2 to pH 3 (24), at which significant survival improvements of various probiotic LAB and bifidobacteria were observed (15). Charteris et al. (21) demonstrated that the presence of milk protein and gastric mucin could significantly protect the majority of the tested probiotic LAB and bifidobacteria in a simulated gastric transit. This explains why acid sensitive strains could be isolated from feces in this study.
Although bifidobacteria were slightly less acid resistant than LAB, they were much more tolerant to the sequential exposure of gastric acid and bile. According to Dunne et al. (25), bifidobacteria isolated from human ileum were less acid and bile resistant than lactobacilli in general. However, commercial B. bifidum and B. longum were as acid resistant as lactobacilli, yet their bile tolerance was lower (14). In this study, all three strains (NIF7AN3, NIF7AN5 and NIF7AN10) of B. bifidum and B. longum NIF7AN2 even increased in number after bile exposure. Bifidobacteria are known to possess strategies of response to various environmental stresses including acid and bile through a set of mechanisms (26). These adaptation mechanisms facilitate their success and survival in the human gut and transient colonization in this competitive niche.
Antimicrobial activity is one of the important properties of probiotic bacteria. Several Lactobacillus strains from infant feces also produced acid to inhibit the growth of enteropathogens (3-6). Acid production was the major cause of the inhibitory activity of the selected strains in this study. Exceptionally, L. plantarum CIF17AN2 and CIF17AN8 retained their antibacterial activity yet loss of this activity was observed with the treatment of catalase enzyme. Most LABs are lactic acid producers, whereas acetic acid is produced mainly by bifidobacteria. De Keersmaecker et al. (27) confirmed that lactic acid accumulated by L. rhamnosus GG strongly inhibited Sal. Typhimurium. The dissociated forms of these organic acids can permeate the outer membrane of Gram-negative bacteria and lower the local pH to inhibit the growth of acid sensitive bacteria. The acidification of cytoplasm and the collapse of the proton motive force resulted in an inhibition of the transport of nutrients (28).
Bacterial adhesion to host mucin is regarded important in contributing to either the transient or even permanent establishment or colonization of probiotic species in any environmental niche and also enhancement of the ability to stimulate the immune system (29). All strains of L. casei, L. plantarum, B. longum and B. bifidum showed good adhesion ability to mucin, while L. rhamnosus did not do so. The results indicate that the adhesion trait was truly a specific characteristic depending on the individual LAB species as previously noted (29, 30). On the contrary, L. rhamnosus GG was reported to adhere significantly better than other LAB strains to the intestinal mucin (30). Lactobacillus rhamnosus E-800 and Lactobacillus GG adhered well to mucin (29). In addition, the mucin adhesive strains competitively excluded S. flexneri, S. typhimurium SA2093, S. paratyphi A and S. aureus TISTR from adhesion to porcine mucin. The inhibition of pathogen adhesion to mucin was reported to be able to prevent translocation and subsequently infection (31).
In conclusion, the human isolates LAB and bifidobacteria identified as L. rhamnosus, L. casei, L. plantarum, B. longum subsp. longum and B. bifidum exhibited high acid and bile tolerance and antibacterial activity against food-borne pathogens. These strains suggest the great potentiality of probiotics in controlling infection by food-borne pathogens in humans. The inhibition of pathogens by LAB was mainly caused by the secretion of antibacterial substances while bifidobacteria effectively inhibited pathogens through their high competitive exclusion activity of mucin adhesion. The combination of these multiple strains in the presence of gut microflora challenged with food-borne pathogens should be further investigated.