1. Backgrounds
Thermophilic Campylobacter species, particularly Campylobacter jejuni and C. coli are zoonotic bacteria frequently associated with human diarrhea in developing and industrialized countries (1). Campylobacter infections are acquired by ingestion of contaminated food, water, or milk, but the sources and transmission routes of campylobacteriosis in humans remain debatable. Numerous vehicles for transmission of Campylobacter infection have been described; however, chicken appears to be the most significant agent for sporadic cases (2-4). To reduce the occurrence of campylobacteriosis in humans, it is essential to improve general understanding of the epidemiology of infection.
The use of conventional bacteriological tests for differentiation and species identification of campylobacters is often hampered by the fact that these bacteria are fastidious and possess few distinguishing biochemical characteristics (5). In the past decades, various methods have been developed for discrimination of Campylobacter at the DNA levels to trace various sources of infection and genotyping replaced the traditional typing methods such as serotype classifications (6-8). Among various genotypic methods, PCR-Restriction Fragment Length Polymorphism analysis of the flagellin-A gene (flaA) is widely used because of its rapidity, ease and cost-effectiveness (9). The existence of extremely conserved and inconsistent regions in flaA gene makes the locus proper for scrutiny of the effects of different restriction enzymes (10).
2. Objectives
The aim of this study was to determine genetic diversity of random collection of C. jejuni and C. coli strains isolated from broiler feces in Shiraz, southern Iran, using PCR-RFLP and investigate the association of Campylobacter species and genetic polymorphism.
3. Materials and Methods
Totally, 42 C. jejuni and 48 C. coli were recovered from fecal samples of the broilers in 10 visits of sampling from Shiraz slaughterhouse in January 2012. The fecal samples were collected in Tryptic Soy Broth (TSB) broth tubes by sterile gloves and brought to the laboratory on ice packs in less than six hours. To eliminate other bacteria, 0.8 μm membrane filter was used and 250 µL of filtered samples was cultured in an enriched broth media [TSB (30 g/L), dextrose (2.5 g/L), sodium thioglycolate (0.5 g/L), Rifampicin (10 mg/L), Trimethoprim (10 mg/L), Vancomycin (10 mg/L), Ceftriaxone (10 mg/L), Amphotericin -B (10 mg/L)], incubated in a microaerophilic atmosphere (Merck, Germany) at 37°C for 4 hours, followed by incubation at 42°C for 44 hours. Thereafter, 50 μL of enriched samples in TSB was cultured on selective agar [brucella agar base (41 g/L), and above antibiotics with identical dose] (11). All culture media were from Merck, Germany. The growth of thermophilic campylobacters was detected by their typical appearance on culture media, i.e. the presence of flat grayish colonies like droplets of water sprayed on the medium. Preliminary identification of Campylobacter species was based on phenotypic characteristics such as colony appearance, Gram staining, microscopic morphology, oxidase and catalase reaction, fermentation of glucose and nitrate reduction (2). The type strains Campylobacter jejuni (ATCC 33291) and Campylobacter coli (RTCC 2541) were included as positive controls for culture identification of isolates.
3.1. DNA Extraction
DNA extraction was performed using the phenol-chloroform extraction technique previously described by Khoshbakht et al. (12). Briefly, a loopful colony of each isolates on agar plate was picked and suspended in 200 µL distilled water. After vortexing, samples were centrifuged at 10000 × g, the supernatants were discarded before adding 250 µL of buffer 1 (resuspension solution contained 100 μg/mL RNase) and 250 µL buffer 2 (Lysis buffer solution contained Tris-HCl and EDTA), a 550 µL saturated phenol was then added, mixed thoroughly and centrifuged at 8000 × g. The supernatant was collected into a new tube; 550 µL of the phenol was then added and centrifuged at 8000 × g again. The supernatant aqueous phase was collected into a fresh tube, previous to adding sodium acetate (2 M, pH 5.2, 0.1 × volume of each aliquot). Then 1.5 mL 100% ethanol were added to the aliquots and mixed, kept at -20°C for 1 hour, centrifuged at 12,000 × g, the supernatant was then discarded and DNA pellet was washed by 80% ethanol, before being dried and suspended in 30 µL of 1 × TE buffer until use.
3.2. Simple and Multiplex PCR Assay
Simple and multiplex PCR reactions were performed for identification of Campylobacter genus, C. jejuni and C. coli species, respectively. PCR amplifications were performed in a final volume of 25 µL. The reaction mixtures consisted of 2 µL DNA template, 2.5 µL 10 × PCR buffer [75 mM Tris-HCl, pH 9.0, 2 mM MgCl2, 50 mM KCl, 20 mM (NH4)2SO4], (CinnaGen, Iran), 1 µL dNTPs (50 µM), (CinnaGen, Iran), 1 µL (1U Ampli Taq DNA polymerase), (CinnaGen, Iran), 1 µL (25 pmol) from the forward and reverse primers (CinnaGen, Iran), shown in Table 1 and the volumes of the reaction mixtures were reached to 25 µL using distilled deionized water. The thermal cycler (MJ mini, BioRad, USA) was adjusted under the following conditions: initial denaturation at 94°C for 5 minutes, followed by 35 cycles of denaturation at 94°C for 1 minute, annealing as shown in Table 1 for 1 minute and extension at 72°C for 1 minute. Final extension was performed at 72°C for 10 minute and the PCR products were remained in the thermal cycler at 4°C until they were collected. Amplified products were separated by electrophoresis in 1.5% agarose gel stained with ethidium bromide. 100 bp DNA ladder was used as molecular size markers. Visualization was undertaken using a UV transilluminator (BTS-20, Japan). The type strains C. jejuni (ATCC 33291) and C. coli (RTCC 2541) were included as positive controls for PCR identification of the isolates and the master mix without sample DNA used as negative control.
3.3. FlaA PCR-Restriction Fragment Length Polymorphism
Isolates identified as C. jejuni and C. coli by m-PCR were typed by PCR-RFLP for the flaA gene. A fragment of 1725 bp (Table 1) of the flaA gene was amplified in a PCR reaction using a pair of specific primers listed in Table 1 previously described by Nachamkin et al. (13). Seven microliters of all the amplicons were restricted with 4 U DdeI (Thermo scientific, Germany) in 1.5 µL 10 × recommended restriction buffer in the final volume of 20 µL and then were incubated at 37°C for 15 hours. The digested PCR products (15 µL) were immediately separated on 2.5% agarose gel stained with ethidium bromide. Bands were photographed under UV transilluminator and the results were evaluated manually. The 50 bp DNA ladder (CinnaGen, Iran) was used as molecular marker to estimate the size of bands. Dendrogram was constructed based on the obtained DNA fragment patterns using Phoretix 1D Version 10 software.
3.4. Statistical Analysis
Statistical analysis was performed using SPSS version 12.0.1. Discrete variables were expressed as percentages and proportions were compared using Chi-squared test with the significance level defined at P < 0.05.
| Name of Primer | Sequence (5´ - 3´) | Target Gene | Annealing Temperature, °C | Product Size, bp | Reference |
|---|---|---|---|---|---|
| MapA | mapA (C. jejuni) | 52 | 589 | (14) | |
| F | CTATTTTATTTTTGAGTGCTTGTG | ||||
| R | GCTTTATTTGCCATTTGTTTTATTA | ||||
| Coli | ceuE (C. coli) | 52 | 462 | (14) | |
| F | AATTGAAAATTGCTCCAACTATG | ||||
| R | TGATTTTATTATTTGTAGCAGCG | ||||
| PLO6 | GGTTAAGTCCCGCAACGAGCCGC | 16SrRNA (genus specific) | 50 | 283 | (15) |
| CAMPC5 | GGCTGATCTACGATTACTAGCGAT | 16SrRNA (genus specific) | 50 | 283 | (15) |
| Fla | flaA (RFLP) | 45 | 1725 | (13) | |
| 1 | GGATlTCGTATTAACACAAATGGTGC | ||||
| 2 | CTGTAGTAATCTTAAAACATTTG |
a Abbreviations: F, forward; R, reverse; RFLP, restriction fragment length polymorphism.
4. Results
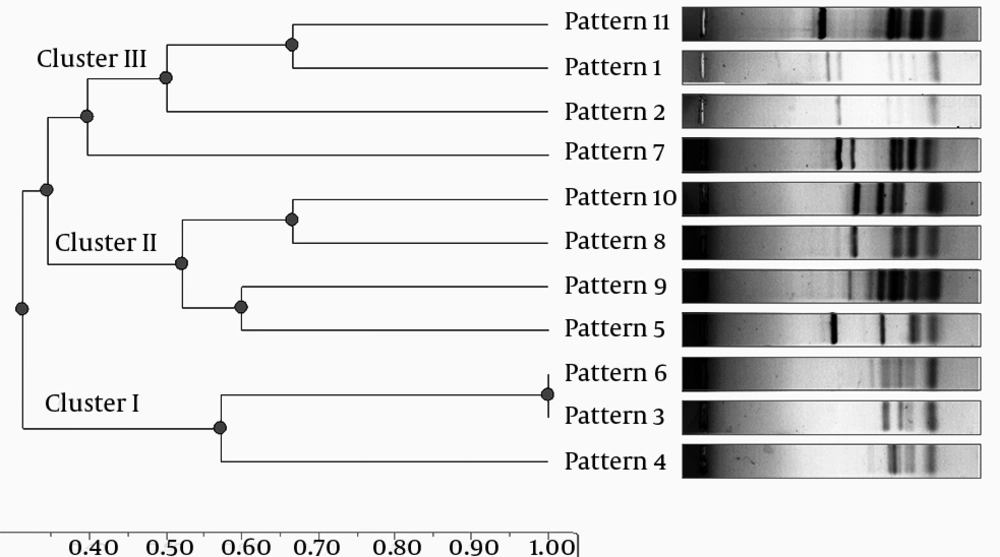
In total, 90 Campylobacter isolates including 48 C. coli and 42 C. jejuni were recovered from fecal samples and identified based on simple and multiplex PCR reactions. All 90 isolates were typed successfully and 11 different genotypes (Table 2) were defined by flaA-PCR-RFLP method. The most prevalent fla-typing pattern of the isolates was F3 (26.6%). Among 42 C. jejuni originating from broiler fecal samples, 10 different types (all types except F4) were defined (Table 2) and the most common type was F1 (19%). Among 48 C. coli isolates, seven types (F1, F3, F4, F5, F6, F8 and F10) were defined and the most common type was type F3 (41.6%). Between 11 different types, four patterns (F2, F7, F9 and F11) were specific for C. jejuni and one pattern (F4) was specific for C. coli isolates. Statistical analysis of flaA-typing data showed that RFLP patterns F2 and F10 were significantly (P < 0.05) predominant among C. jejuni and RFLP patterns F3 and F6 were significantly (P < 0.05) predominant among C. coli isolates. The phylogenetic analysis based on the dendrogram generated from RFLP-PCR demonstrated that C. jejuni and C. coli isolates belong to three clusters (I, II and III in Figure 1). 45.5% (41/90) of isolates were found in cluster I and 27.7% (25/90) and 26.6% (24/90) of isolates belonged to clusters II and III, respectively. The dominant cluster of C. jejuni and C. coli isolates was significantly (P < 0.05) difficult, so that 72.9% (35/48) of C. coli belonged to cluster I, while 50% (21/42) and 35.7% (15/42) of C. jejuni belonged to clusters III and II, respectively (Figure 1).
| Species | No. | Occurrence of Different flaA-RFLP Patterns | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 | F11 | ||
| C. jejuni | 42 | 8 (19) | 4 (9.5) | 4 9.5) | 0 (0) | 3 (7.1) | 2 (4.7) | 7 (16.6) | 2 (4.7) | 4 (9.5) | 6 (14.2) | 2 (4.7) |
| C. coli | 48 | 3 (6.2) | 0 (0) | 20 (41.6) | 3 (6.2) | 2 (4.1) | 12 (25) | 0 (0) | 6 (12.5) | 0 (0) | 2 (4.1) | 0 (0) |
| Total | 90 | 11 (12.2) | 4 (4.4) | 24 (26.6) | 3 (3.3) | 5 (5.5) | 14 (15.5) | 7 (7.7) | 8 (8.8) | 4 (4.4) | 8 (8.8) | 2 (2.2) |
a Data are presented as No. (%)
5. Discussion
Typing of Campylobacter isolates from different sources provides epidemiological information needed for infection control and contributes to risk evaluation of transmission. Genotype-based identification methods have been developed to avoid the problems caused by biochemical inertness of Campylobacter species. These methods are now more frequently used, but have yet to be standardized. The application of molecular typing methods can offer a constant and highly discriminatory investigation of bacterial isolates. The present study considered the heterogeneity among C. jejuni and C. coli isolates obtained from broiler feces by molecular methods for the first time in Iran.
PCR-RFLP genotyping method was used to explain genetic diversity among these isolates. All 90 isolates confirmed based on m-PCR were typed successfully using fla typing method. Harrington et al. (16) in their study compared three different methods of flaA-RFLP technique and showed that, full flaA gene and DdeI digestion are proper for fla-typing of campylobacters. Other similar studies indicated the benefits of full length flaA gene and DdeI (17, 18), which we used in this study. In the current study, 11 distinct flaA-types were obtained, using DdeI, which did not show high diversity among isolates compared with other studies on Campylobacter isolates from some other sources (19-22). These differences in genetic variation among thermophilic Campylobacter strains can be explained by geographical diversity, different number of isolates compared with other studies and difference in origin. Compared with other poultry-originated Campylobacter isolates in some studies (9), the isolates in the present study showed lower diversity, but some other investigations showed lower diversity (1).
Results showed that among 11 RFLP patterns, six (54.5%) patterns were similar between C. jejuni and C. coli strains. Ertas et al. (1) showed seven different fla-types among C. jejuni and C. coli isolates, which all were similar among two species. Nevertheless, Oporto et al. (22) in analysis of thermophilic campylobacters from cattle, sheep and swine farms obtained 46 different patterns, which only five (10.8%) were similar between C. jejuni and C. coli isolates. These data indicate low genetic diversity among our study broiler originated thermophilic campylobacters. More studies are necessary to evaluate genetic diversity differences between two thermophilic species. Isolation of Campylobacter from each cloacal swab, without resorting to pooling, made it possible to perform an epidemiological study using a molecular method such as flaA-typing to evaluate the relatedness of Campylobacter isolates of each farm.
Our isolates were not recovered from a single commercially reared flock and samples were collected randomly from separate flocks in slaughtering line at the end of the rearing period. Genetic differences between poultry-originated isolates may be due to variety in age of chickens, site of sampling and category of rearing (23). According to the results, C. jejuni isolates showed significantly (P < 0.05) higher diversity (10 patterns) than C. coli isolates (7 patterns). In addition, predominant flaA-typing patterns of C. jejuni strains were significantly (P < 0.05) different from C. coli isolates. Acik and Cetinkaya (21) showed that among 209 isolates, 28 different flaA types were found. Twenty-three flaA types were identified among 179 C. jejuni isolates and the remaining five from C. coli isolates. Using RFLP image analysis, dendrogram was drawn and we examined the genomic association between the Campylobacter isolates tested in the study. C. jejuni and C. coli were randomly distributed in three clusters, but most isolates were found in cluster I (Figure 1). The results showed that C. jejuni and C. coli isolates significantly (P < 0.05) belonged to different clusters in phylogenic dendrogram drawn according to flaA-RFLP patterns. Clusters I and III had significant (P < 0.05) relatedness with C. coli and C. jejuni, respectively. This can show the differences between fla-typing features of these two species, which can be observed by sequencing and comparison of all flaA PCR products. Three of four patterns, which were not present in C. coli isolates, were in cluster III.
In conclusion, the presence of campylobacters in broiler feces can contaminate the environmental and human food chain. Therefore, detection of Campylobacter spp. in broiler originated samples is important to identify possible sources of infection and to have a better understanding of the epidemiology of infection subtyping of isolates is considered essential. Results indicated that Fla-typing is a simple and low-cost typing method characterized by a high level of discrimination power and it is an efficient tool for identifying any outbreak-related strains in short-term investigations. The results of this study revealed a comparatively low heterogeneity among C. jejuni and C. coli isolated from the broilers in southern Iran. In addition, results showed that the predominant RFLP patterns of C. coli and C. jejuni isolates can be significantly different and belong to various distinct phylogenic clusters. These molecular epidemiologic findings should be taken into account in the investigations towards developing effective control strategies against C. jejuni and C. coli infections.