1. Background
Human papillomavirus (HPV) infection of the anogenital tract is the most prevalent viral sexually-transmitted infection in males and females, with a broad range of clinical manifestations and consequences varying from subclinical and self-limited to persistent and related with malignant progression (1, 2). Persistent infection with HPV is a well-established cause of cervical cancer and there has been massive advancements in the characterization of the natural history of cervical HPV infection in females (3-10). However, there is a lack of knowledge with regard to the natural history of HPV infection in males, especially in developing countries (2).
HPV infection is associated with several malignant disorders in males, including penile, anal, and oral cancers (11-14). Moreover, HPV is responsible for the development of condylomata acuminata (genital warts) and other nonmalignant diseases such as recurrent respiratory papillomatosis and oral papillomas. Most HPV-related cancers in males are associated with HPV16/18 and nearly all HPV-positive nonmalignant diseases are caused by HPV6/11 (15-18). Recently, it has been revealed that HPV infection in males has been associated with the increased risk of human immunodeficiency virus (HIV) infection acquisition (19). Taking into account that HPV is a sexually-transmitted virus, HPV infection in males may result in considerable diseases in females (2). Therefore, HPV infection in males is a significant clinical matter. With the advent of effective prophylactic vaccines against oncogenic HPV genotypes, understanding of the burden of HPV infection and its genotype-specific prevalence in males has attracted enormously growing attention (20, 21)
2. Objectives
The aim of this study was to assess the prevalence of genital HPV infection and its genotype distribution among Iranian males which might be useful for designing public health policies and prevention measures, including vaccination.
3. Patients and Methods
3.1. Patients
A total of 483 males, referred to Iran University of Medical Sciences-affiliated sexually transmitted infections (STI) clinics for genital HPV testing and genotyping during March 2009 through April 2014, were enrolled in this cross-sectional study. The inclusion criteria included the presence of a genital lesion, no symptoms and having an HPV-positive partner, no symptoms and a desire to screening for sexually-transmitted infections. This investigation was approved by the ethical committee of Tehran University of Medical Sciences and informed consents were obtained from all the participants. Exfoliated epithelial cells were collected from the urethra, penile shaft, glans, scrotum and anus with sterilized brush or Dacron swab (22). The samples were placed into standard transport medium and stored at -80°C prior to HPV detection and genotyping.
3.2. DNA Extraction and INNO-LiPA HPV Genotyping Assay
Total DNA was extracted using the QIAamp DNA Mini kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions. HPV genotyping was carried out using the INNO-LiPA HPV genotyping extra assay (Innogenetics NV, Ghent, Belgium), according to the manufacturer’s instructions. The INNO-LiPA HPV assay is one of the most widely used HPV genotyping tests based on the concept of reverse hybridization, planned for the identification of 28 different genotypes of HPV including 15 HR genotypes (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73 and 82 ), three probable HR (pHR) genotypes (26, 53 and 66), seven LR genotypes (6, 11, 40, 43, 44, 54 and 70) and three genotypes (69, 71 and 74) which are not categorized as HR, pHR or LR genotypes, based on Munoz et al. (23). A 65 bp region of HPV L1 gene was amplified using consensus SPF10 primers, followed by denaturation and hybridization of the resulting biotinylated amplicons, with specific oligonucleotide probes fixed on membrane strips (24). To control the specimen quality and DNA extraction an additional primer pair targeting, the human HLA-DPB1 gene was included.
After PCR amplification using the INNO-LiPA HPV Genotyping Extra Amp, the amplified biotinylated product was denatured using an alkaline solution. The denatured PCR product was then hybridized to probes immobilized on membrane strips. Thereafter, streptavidin-conjugated alkaline phosphatase was added, which binds to any biotinylated PCR product/probe hybrid previously formed. Finally, the PCR product bound to a specific probe was detected by adding BCIP/NBT chromogen. The results were interpreted visually using the interpretation chart provided.
3.3. Statistical Analysis
Data analysis was performed using SPSS version 16 software (SPSS Inc., Chicago, IL, USA). The association of HPV infection and age was evaluated using the logistic regression test. A P value < 0.05 was considered as statistically significance. 95% CI for prevalence estimations was performed using an online confidence intervals calculator (https://www.mccallum-layton.co.uk).
4. Results
A total of 483 males were enrolled in this investigation. The mean age of the participants was 34.4 ± 8.5 (range: 15 - 76). Table 1 shows the prevalence of HPV genotypes. Totally, HPV DNA was detected in 269 (55.7%, 95% CI: 51.2 - 60.1%) of the subjects. HPV6 was the predominant HPV genotype detected overall (46.2%). HPV11 was confirmed as the second most common genotype (8.1%).
| HPV Genotypes | No. | Percent (95% CI) |
|---|---|---|
| Any HPV genotypes | 269 | 55.7 (51.2 - 60.1) |
| High-risk | 46 | 9.5 (7.2 - 12.5) |
| 16 | 11 | 2.3 (1.2 - 4) |
| 18 | 9 | 1.9 (1 - 3.5) |
| 31 | 3 | 0.6 (0.2 - 1.8) |
| 33 | 3 | 0.6 (0.2 - 1.8) |
| 39 | 3 | 0.6 (0.2 - 1.8) |
| 45 | 1 | 0.2 (0.04 - 1.2) |
| 51 | 4 | 0.8 (0.3 - 2.1) |
| 52 | 9 | 1.9 (1 - 3.5) |
| 58 | 2 | 0.4 (0.1 - 1.5) |
| 82 | 1 | 0.2 (0.04 - 1.2) |
| Probable high-risk | 4 | 0.8 (0.3 - 2.1) |
| 53 | 1 | 0.2 (0.04 - 1.2) |
| 66 | 3 | 0.6 (0.2 - 1.8) |
| Low-risk | 268 | 55.5 (51.1 - 59.9) |
| 6 | 223 | 46.2 (41.8 - 50.1) |
| 11 | 39 | 8.1 (6 - 10.8) |
| 43 | 1 | 0.2 (0.04 - 1.2) |
| 44 | 4 | 0.8 (0.3 - 2.1) |
| 54 | 1 | 0.2 (0.04 - 1.2) |
aAbbreviation: HPV, human papillomavirus.
Forty six (9.5%, 95% CI: 7.2 - 12.5%) males were positive for HR HPV genotypes. The most prevalent HR genotype was HPV16 (2.3%), followed by HPV18 (1.9%) and HPV52 (1.9%). LR HPV genotypes were detected in 268 (55.5%, 95% CI: 51.1 - 59.9%) subjects. The most common LR genotypes were HPV6 and HPV11. The prevalence of infection with multiple HPV genotypes or coinfection determined by two or more HPV genotypes is shown is Table 2. Infection with multiple HPV genotypes was observed in 38 (7.9%, 95% CI: 5.8 - 10.6%) of the study participants (14.1% (95% CI: 10.5 - 18.8%) of HPV-positive cases). Of those with multiple HPV genotypes infection, 31 (6.4%) had coinfection with two genotypes, 4 (0.8%) had coinfection with three genotypes, 2 (0.4%) had coinfection with four genotypes, and only 1 (0.2%) had coinfection with five genotypes.
| No. | Percent (95% CI) | |
|---|---|---|
| Multiple HPV genotypes infection | 38 | 7.9 (5.8 - 10.6) |
| Multiple HR HPV genotypes infection | 4 | 0.8 (0.3 - 2.1) |
| Multiple LR HPV genotypes infection | 9 | 1.9 (0.1 - 3.5) |
| Infection with both HPV16 and HPV18 genotypes | None | 0 (0) |
| Infection with both HPV6 and HPV11 genotypes | 6 | 1.2 (0.6 - 2.7) |
| Infection with HPV6, HPV11, HPV16 and HPV18 genotypes | None | 0 (0) |
| Number of HPV genotypes | ||
| 1 | 231 | 47.8 (43.4 - 52.3) |
| 2 | 31 | 6.4 (4.6 - 9) |
| 3 | 4 | 0.8 (0.3 - 2.1) |
| 4 | 2 | 0.4 (0.1 - 1.5) |
| 5 | 1 | 0.2 (0.04 - 1.2) |
aAbbreviations: HPV, human papillomavirus; HR, high-risk; LR, low-risk.
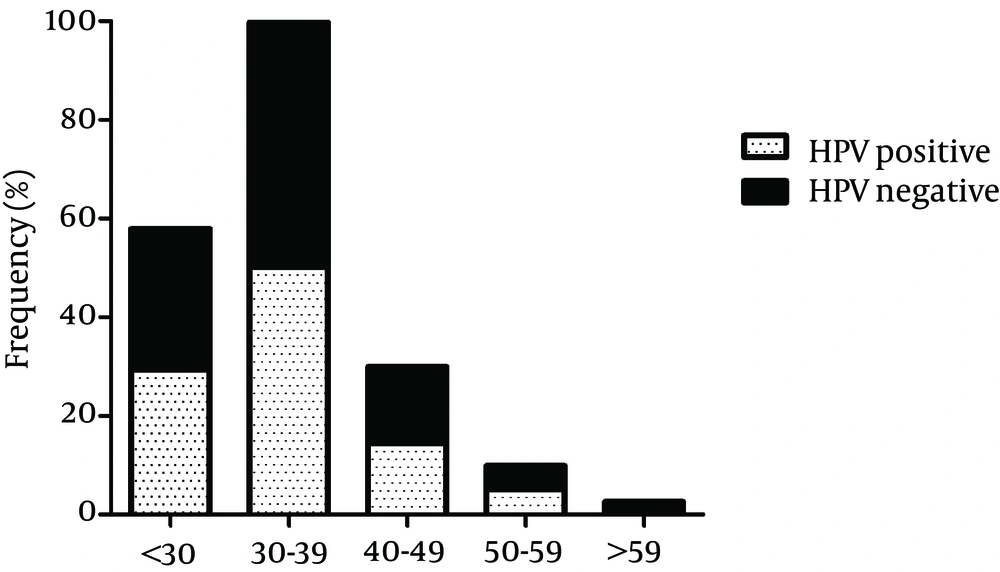
Overall, 42 cases (8.7%, 95% CI: 6.5 - 11.5%) were infected with at least one HR HPV genotype and at least one LR HPV genotype was detected in 258 (53.4%) of the participants. Multiple HR HPV genotypes and multiple LR HPV genotypes infections were found in 4 (0.8%, 95% CI: 0.3 - 2.1%) and 9 (1.9%, 95% CI: 0.1 - 3.5%) of the study samples, respectively. In particular, 20 (4.1%, 95% CI: 2.7 - 6.3%) cases were infected by HPV16 or HPV18. However, concomitant infection with both HPV16 and HPV18 was not observed. In 250 (51.8%, 95% CI: 47.3 - 56.2%) cases, infections with HPV6 or HPV11 were found, while coinfection with both HPV6 and HPV11 was observed in only 6 (1.2%, 95% CI: 0.6 - 2.7%) subjects. pHR genotypes were detected in 4 (0.8%, 95% CI: 0.3 - 2.1%) cases and not-categorized genotypes were not found in any of the cases. Figure 1 shows the age-specific prevalence of HPV infection. The highest prevalence of HPV was detected in males aged between 30 - 39 years and the lowest prevalence was seen in those more than 59 years old. However, using logistic regression, there was no statistical association between HPV infection and age (P = 0.469). In addition, there was no significant correlation between age and HR HPV infection (P = 0.330) or LR HPV infection (P = 0.346).
5. Discussion
The identification of HPV infection and its genotyping description in males is a serious clinical issue due to the strong association of persistent HPV infection and several cancers in males. In addition, males have an important role in the transmission of HPV to females (1). The knowledge of HPV infection in males seems to be necessary for public health policies and males’ vaccination with HPV vaccine. However, data about the epidemiology of HPV infection in Iranian males are considerably low. The present cross-sectional study described the prevalence of genital HPV infection and HPV genotype distribution in Iranian males. To our knowledge, this was one of the largest epidemiological studies reporting the prevalence of HPV infection and its genotype distribution in males.
In this study, the prevalence of HPV infection (any genotype) was relatively high (54.8%). This finding is in agreement with other published reports, which showed the prevalence of about 50% in STI clinic attendees (25, 26). Totally, 17 different HPV genotypes were detected in the study population. The most prevalent HPV genotypes were HPV6 (46.2%) and HPV11 (8.1%), in line with previous reports (15, 27, 28). In agreement with several previous studies investigating the prevalence and genotype distribution of HPV in male genital warts (15, 27, 29-31), HPV16 was confirmed as the most common HR HPV genotype and the third most common HPV genotype detected after HPV6 and HPV11 in this study. Furthermore, these findings are consonant with those of Freire et al. (32) who assessed participants similar with to our study samples. The results also placed HPV18 and HPV52 as the second most prevalent HR genotypes. Therefore, it could be claimed that vaccine-targeted HPV genotypes (HPV6, HPV11, HPV16, and HPV18) were among the commonly detected HPV genotypes in our investigation. Potentially, the availability of an effective vaccine against theses genotypes may allow us to prevent the most common HPV genotypes in Iranian males.
Infection with multiple HPV genotypes which was found to associate with the increased risk of HPV persistence (33) was relatively low in the present study. We detected infection with multiple HPV genotypes in 14.1% of HPV-positive cases. This finding is in disagreement with several reports, showing the rate of infection with multiple HPV genotypes to be 33.8% (15), 56.7% (27) and 59.7% (32). This variation between the studies could be explained by differences in sampling approaches, the HPV detection protocols employed, and geographical variations in HPV genotypes distribution. While the prevalence of HPV infection was the highest among males aged 30 - 39 years, no significant association was identified between HPV infection and age in this investigation (P = 0.469). The absence of association between HPV infection and age was also reported by several studies (14, 32, 34-36).
This study provided beneficial information about the epidemiology of genital HPV infection in Iranian males, which should be applied for evaluating the efficiency of HPV vaccines for the prevention of vaccine-targeted HPV genotypes. However, this report should be interpreted with caution, due to the STI clinic setting of this investigation. Therefore, this data could not be generalizable to the general population of Iranian males. In conclusion, the prevalence of HPV infection was relatively high. Totally, 17 different HPV genotypes were detected and the most frequently detected genotypes were HPV6, HPV11, HPV16, HPV18 and HPV52, respectively.