1. Background
Diarrheagenic Escherichia coli are the most common cause of bacterial diarrhea in infants in developing countries (1). Several studies performed in developed and developing countries worldwide have identifiedbenteroaggregative Escherichia coli (EAEC) strains as the emerging and the most common cause of pediatric diarrhea (2-4). HEp-2 or HeLa cell adherence assay is the gold standard test for identifying EAEC strains. EAEC strains show a special stacked-brick aggregative adherence (AA) pattern (5). The EAEC strains in the HEp-2 cell assay exhibit the AA pattern. However, this technique requires special expertise and facilities and is time consuming. Therefore, it is only performed in a few laboratories worldwide, thus limiting both the diagnosis of and epidemiological studies on EAEC (6).
A recent study described a molecular method called multiplex PCR (mPCR) to identify EAEC strains by detecting 3 virulence plasmid genes, namely, aap, aggR, and aatA (6). Cell adhesion properties of some EAEC strains are attributed to a 60- to 65-MDa plasmid PAA. The plasmid PAA contains genes encoding several virulence factors such as AA fimbriae (AAF/I, AAF/II, and AAF/III), dispersin (AaP), transcriptional activator (AggR), plasmid-encoded toxin, and heat-stable toxin 1 (7). EAEC strains adhere to the mucosal surface of the small and large intestines and stimulate mucus secretion, thus resulting in the formation of a thick, aggregating biofilm (8). Biofilm formation restricts the penetration of antimicrobial agents, decreases the growth rate of EAEC strains, and increases the possibility of the expression of resistance genes. For these reasons, biofilm colonies of EAEC cannot be easily eradicated using bactericidal antibiotics (9).
Several methods such as pulsed-field gel electrophoresis (PFGE), Random amplified polymorphic DNA (RAPD), and Multilocus sequence typing (MLST) have been used for determining the molecular epidemiology of EAEC strains (7, 10, 11). PFGE is a powerful tool for determining the clonal identity of bacteria and for obtaining information to understand and control the spread of diseases (12). PFGE is the gold standard technique for typing many bacterial species, including E. coli, but not for typing some species such as Mycobacterium tuberculosis (13, 14).
2. Objectives
Because the cell adherence assay requires special expertise and is expensive and time consuming, we investigated the utility of mPCR for detecting EAEC strains. Few studies have performed the molecular typing of and have investigated the virulence characteristic of EAEC strains isolated from children in Iran. To our knowledge, this is the first study to use PFGE for determining the clonal relatedness of EAEC strains isolated from Iranian children with diarrhea. Thus, this study aimed to investigate the phenotypic and genotypic characteristics of EAEC strains from isolated from Iranian children with diarrhea who were referred to the Children's Medical Center in Tehran, Iran.
3. Materials and Methods
3.1. Bacterial Strains
This study included 170 EAEC strains obtained from a culture collection center at Molecular Biology Unit, Pasteur Institute of Iran. These strains were originally isolated from children with diarrhea aged below 5 years who were admitted to the children’s medical center hospital, Tehran, Iran, during 2007 - 2008. All the strains had been previously characterized as EAEC by performing the HeLa cell adherence assay. These strains were maintained at -80°C in Luria broth (HiMedia, India) supplemented with 20% glycerol (Merck, Germany). This study was approved by the Research Ethics Committee of the Pasteur Institute of Iran (no. 4312).
3.2. Plasmid DNA Extraction
EAEC strains grown overnight in Luria broth were used for plasmid DNA extraction by using alkaline lysis method (15). Purified plasmid DNA (3 µL) was analyzed by performing gel electrophoresis using 1% agarose gel (SinaClon Co., Iran) stained with ethidium bromide (10 mg/L; SinaClon Co.) and was visualized using a UV transilluminator (BioDoc-It; UVP, USA).
3.3. Multiplex PCR
Sequences of primers used for mPCR were based on those described previously (6). EAEC strains (17-2 and O42) and E. coli K12 were used as positive and negative controls, respectively. Multiplex PCR was performed in a 25-µL reaction mixture containing the extracted plasmid DNA as the template, 200 μmol dNTP’s, 20 pmol of primers (Takapouzist, Iran) against aatA, 15 pmol of primers against aggR, and 10 pmol of primers against aap, 0.75 µL MgCl2, 1 U Taq polymerase (Gibco, UK), and 2.5 µL 10 × PCR buffer (Gibco, UK). PCR was performed in MasterCycler Gradient (Eppendorf, Germany) by using the following conditions: initial denaturation at 94.5°C for 3 minutes; 30 cycles of denaturation at 94.5°C for 1 minute, annealing at 50°C for 1 minute, and extension at 72°C for 1.5 minutes; and final extension at 72°C for 8 minutes. PCR products were electrophoresed on 1% agarose gel, and amplicons of correct size were considered positive.
3.4. Drug Susceptibility Test
Antibacterial susceptibility testing was performed using disk diffusion method according to the CLSI guideline (16). Disks (bioMerieux, UK) of the following antibiotics were used: ciprofloxacin (5 µg), streptomycin (10 µg), chloramphenicol (30 µg), tetracycline (30 µg), nalidixic acid (30 µg), kanamycin (30 µg), gentamicin (10 µg), cephalothin (30 µg), ampicillin (10 µg), ceftizoxime (30 µg) and trimethoprim/sulfamethoxazole (1. 25/23. 75 µg).
3.5. Biofilm Assay
Biofilm formation test was performed according to a method described by Wakimoto et al. (17) with some modifications. Briefly, 200 µL Mueller–Hinton broth (Merck, Germany) supplemented with 0.45% glucose was added to 96-well flat-bottom microtiter polystyrene plates (Greiner, Germany) and was inoculated with 5 µL of EAEC culture grown overnight in Luria broth at 37°C with shaking. The samples were incubated overnight (18 hours) at 37°C and were visualized by staining with 0.5% crystal violet (Sigma-Aldrich, Germany) for 5 minutes after washing with water. Biofilm formation was quantified in duplicate by adding 200 µl of 95% ethanol and by using an enzyme-linked immunosorbent assay plate reader (BioTek Instruments, Winooski, VT) at 570 nm. EAEC strain 042 was used as a positive control, and E. coli HB101 was used as a negative control. Strains with OD at 570 nm of more than 0.2 were regarded as biofilm producers (biofilm-positive strains) according to a previous study (17).
3.6. Pulsed-Field Gel Electrophoresis
Thirty-one EAEC strains carrying the virulence plasmid genes were selected as representative strains and were typed using PFGE to investigate their clonal relationships according to a protocol described by Zhao et al. (18). The resulting fragments from PFGE were resolved by performing contour-clamped homogeneous electric field electrophoresis with CHEF Mapper system (Bio-Rad, USA) in an autoalgorithm mode and 1% PFGE-grade agarose gel (Bio-Rad) in 0.5 × TBE (44.5 mM Tris-HCl, 44.5 mM boric acid, and 1.0 mM EDTA [pH 8.0]) at 6 V/cm for 18 hours at 14°C. The gels were stained with ethidium bromide (30 mg/L) and were digitized for computer-aided analysis.
Chromosomal DNA (225-2,200 kb; Bio-Rad) of Saccharomyces cerevisiae was used as a DNA marker. The DNA fragments were digested with XbaI and were separated on 1% agarose gel. Images of PFGE patterns were clustered using GelCompar II software (Applied Maths, Belgium). Similarity percentage was determined using Dice coefficient. Strains were considered to be clonally related if their Dice coefficient of correlation was ≥80% (19, 20).
4. Results
4.1. Multiplex PCR
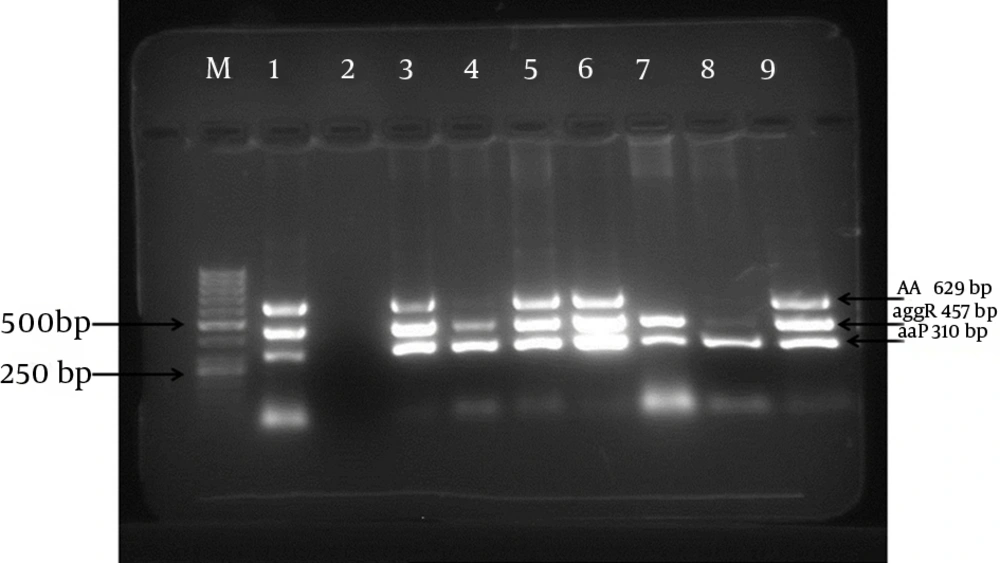
The mPCR assay detected the 3 virulence plasmid genes, namely, aap, aggR, and aatA, in the examined EAEC strains. A representative agarose gel of the mPCR assay for control strains and some EAEC strains is shown in Figure 1. Of the 170 EAEC strains that were previously characterized by determining AA to HeLa cells, 114 (67%) strains yielded positive results in the mPCR assay and had at least one virulence plasmid gene. The remaining 56 (33%) strains yielded negative results in the mPCR assay. The frequency of aap, aggR, and aatA was 67%, 64.7%, and 47%, respectively.
4.2. Drug Susceptibility Test
Highest resistance rates were observed against ampicillin (100%), tetracycline (65.7%), streptomycin (58.7%), chloramphenicol (52.6%), and trimethoprim/sulfamethoxazole (51.7%). The frequency of resistance to cephalothin, kanamycin, ceftizoxime and gentamicin were 40.3%, 21%, 15.7% and 7% respectively. All the strains were susceptible to ciprofloxacin and nalidixic acid. Of the 114 EAEC strains examined, 47 showed different antibiotic resistance profiles. The most common antibiotic resistance profiles were ampicillin, tetracycline, streptomycin, chloramphenicol, and trimethoprim/sulfamethoxazole (in 7 strains); ampicillin, tetracycline, streptomycin, chloramphenicol, trimethoprim/sulfamethoxazole, and cephalothin (in 5 strains); and ampicillin, tetracycline, streptomycin, and chloramphenicol (in 4 strains). Very few strains showed identical resistance profiles (data not shown).
4.3. Biofilm Formation
Biofilm formation is more common among EAEC strains than among other E. coli pathotypes (17). Therefore, we compared mean biofilm formation between EAEC and EPEC strains. Biofilm formation test was performed for all EAEC strains that yielded positive results in the mPCR assay and for 40 EPEC strains. In all, 73 (64%) EAEC strains produced biofilm, with a mean biofilm production of 0.857 and standard deviation of ± 0.763. Of the 40 EPEC strains, 15 strains (37.5%) produced biofilm, with a mean biofilm production of 0.698 and standard deviation of ± 0 926.
4.4. Pulsed Field Gel Electrophoresis (PFGE)
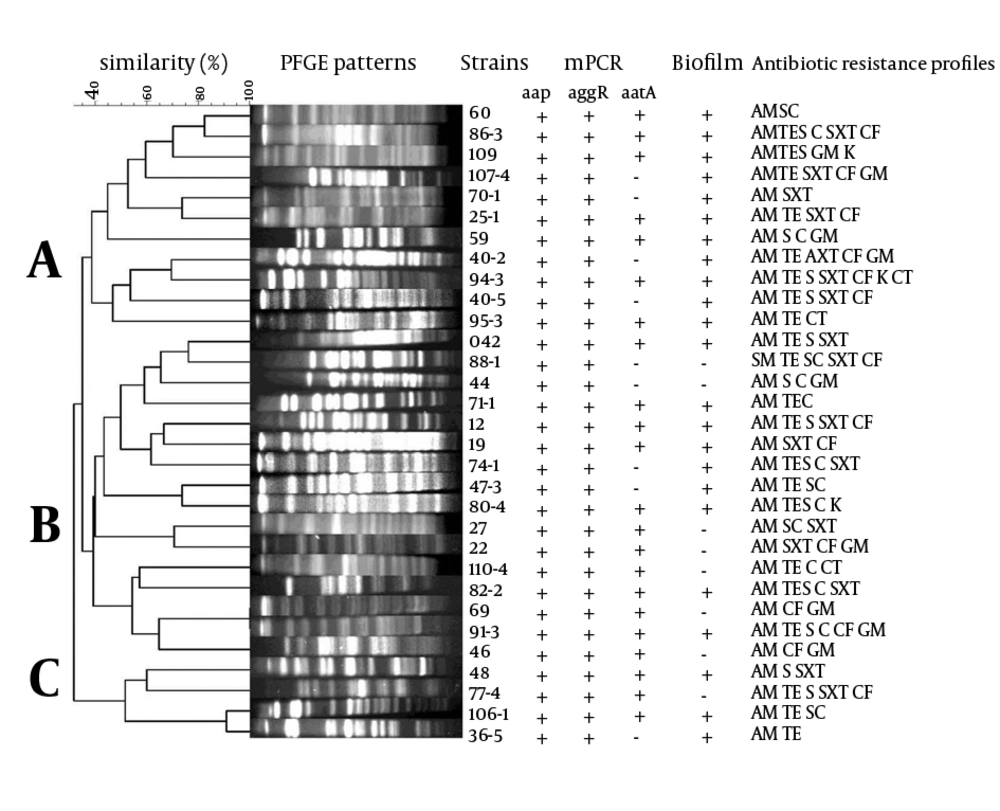
PFGE dendrogram of the EAEC strains examined showed 26 different PFGE patterns for 31 isolates. Most of these patterns (88.5%) had 1 isolate and 3 patterns (11.5%) consisted of two strains (strains 60 and 86-3 in one pattern; strains 69 and 91-3 in one pattern; and strains 106-1 and 36-5 in one pattern) (Figure 2). EAEC strains 60 and 86-3 carried the 3 virulence plasmid genes and produced biofilm. EAEC strains 69 and 91-3 had identical PFGE patterns and carried the 3 virulence plasmid genes; however, strain 69 did not produce biofilm. EAEC strains 106-1 and 36-5 produced biofilm; however, strain 36-5 lacked aatA.
The dendrogram showed 3 PFGE types, namely, A, B, and C. Type A included 11 EAEC strains all of which could form biofilms. Type B was the largest type and included 16 strains, of which 9 produced biofilm. This PFGE type also included EAEC strain 042. Type C was smaller than the other 2 PFGE types and included 4 strains, of which 3 could form biofilms. Thus, only PFGE type A included all biofilm-producing EAEC strains; the other 2 PFGE types included both biofilm-producing and biofilm-nonproducing EAEC strains. Almost all the EAEC strains examined carried aap and aggR. Twenty-two strains in the PFGE dendrogram carried aatA, of which 12 were included in PFGE type B, 7 were included in PFGE type A, and 3 were included in PFGE type C. EAEC strains in the 3 PFGE types showed different antibiotic resistance profiles (Figure 2).
5. Discussion
EAEC cause acute and persistent diarrhea, mainly in children in both developing and developed countries. The pathogenesis of infections caused by EAEC is not well understood. Several studies have shown that EAEC strains exhibit considerable heterogeneity (10, 21-24). Only a few studies have been performed on EAEC strains isolated from Iran (25, 26). In the present study, EAEC strains were characterized using different phenotypic and genotypic methods. To our knowledge, this is the first study in Iran to investigate the PFGE profiles and biofilm production by EAEC strains.
We observed that 67% EAEC strains yielded positive results in the mPCR assay. The frequency of aap, aggR, and aatA was 67%, 64.7%, and 47%, respectively. The remaining 33% strains yielded negative results in the mPCR assay. A study by Cerna et al. (6) and our previous studies showed that 14% and 18.9% EAEC strains, respectively, yielded negative results in the mPCR (27). However, mPCR cannot detect some EAEC strains. The use of mPCR increases both the sensitivity and specificity for EAEC detection, which may help in the early diagnosis of infections caused by these bacteria (6). The HeLa cell adherence assay requires special expertise and is time consuming and expensive. In contrast, the mPCR assay is inexpensive and quick (6). In developing countries such as Iran that have limited resources, the mPCR assay may be useful for monitoring diarrheagenic E. coli.
The results of the present study are consistent with those of several previous studies, which indicated that EAEC strains showed high antibiotic resistance and different antibiotic resistance patterns (7, 10, 11, 25). In the present study, only 3 antibiotic resistance patterns were observed among the EAEC strains examined. These findings are consistent with those of a study by Kahali et al. (7) that showed different antibiotic resistance patterns among EAEC strains. However, high resistance rates of diarrheagenic E. coli, including EAEC, against commonly used antibiotics such as ampicillin, trimethoprim/sulfamethoxazole, and tetracycline are concerning because they may lead to treatment failure (28).
Bangar and Mamatha (29) and Wakimoto et al. (17) reported that all EAEC strains formed biofilms. However, only 64% strains examined in our study produced biofilm. In addition, we observed a significant relation between EAEC strains and biofilm production (P < 0.05). Similar to that observed in previous studies (3, 7, 11), EAEC strains examined in the present study had different PFGE patterns, indicating that they were genetically heterogeneous. Only some strains had identical PFGE patterns, suggesting that these strains were epidemiologically related. The differences in the PFGE patterns of EAEC strains examined in the present study and those reported in previous studies could be because of the different ancestral origins of EAEC strains in each country (28).
Because EAEC strains examined in this study showed different PFGE and antibiotic resistance patterns, no association could be determined between PFGE patterns and antibiotic resistance patterns. These results indicated that the local EAEC strains examined in this study were heterogeneous, which was consistent with that observed in other regions around the world. The PFGE patterns of the strains included in the present study also confirmed this heterogeneity and were consistent with other phenotypic studies performed on these strains. However, these findings did not provide information on the pathogenesis and the role of these strains in pediatric diarrhea.