Dear Editor,
Free living amoebae (FLA) are frequently distributed in environment, such as air, water, dust and soil (1). Some strains of Acanthamoeba spp. are non-pathogen, while others are pathogenic in immunocompromised patients or even healthy persons (2-4). Acanthamoeba encephalitis is rare and less likely to be diagnosed with successful medical intervention. These amebas can carry and transmit important and pathogenic bacteria such as Vibrio cholera and Legionella (5). Since there was not any information about distribution of Acanthamoeba in Mashhad City, the main objective of the present research was investigating the presence of potentially pathogenic Acanthamoeba genus by morphological criteria and temperature tolerance assays in water sources of Mashhad, Iran.
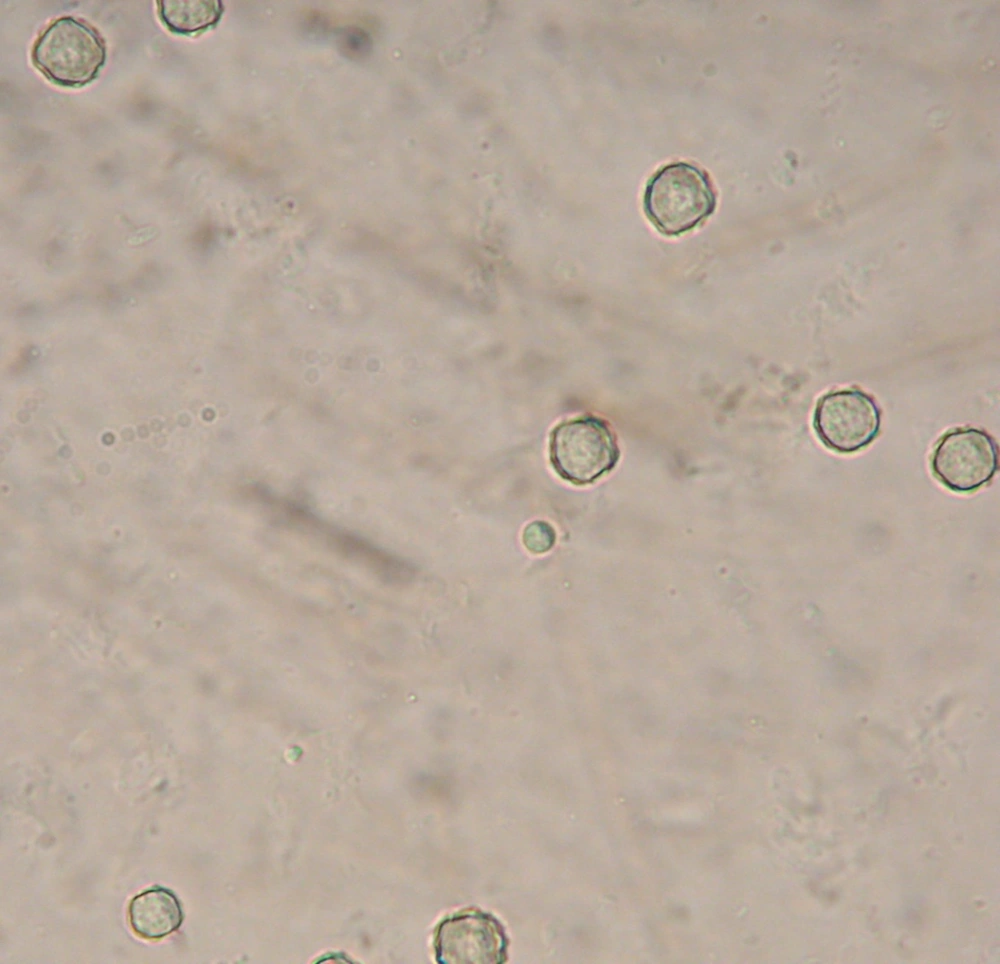
Between December 2012 to June 2013, 61 water samples were collected randomly from squares and ponds of parks of 13 regions of Mashhad, Iran. For each sample, approximately 500 mL of water was filtered through 0.45 µm pore-size cellulose nitrate membranes. Then the filter was cultured on Non-nutrient agar enriched with Escherichia coli (NNA) prepared with Amoeba Page Saline (6). Then the plate was sealed with parafilm and incubated at the respective temperatures. Two plates from each of the sampling point and subculture were incubated at room temperature (25 - 28°C) and 37°C, respectively. Identification of the obtained Acanthamoeba spp. was achieved according to morphological characteristics of both trophic and cyst stages (Figure 1).
In the present study, in 15 samples, free-living amoebae grown at room temperature and 6/61 (9.8%) of the total examined water samples had positive results at 37°C. In this study, some of these free living amebas were potentially pathogenic according to morphological criteria and thermotolerant assay. Moreover, Acanthamoeba is introduced as a vehicle for circulation of different pathogens viruses and bacteria such as Legionella, Pseudomonas and Helicobacter between human and environments (7-9). Human activity was seen in all of the localities from which samples were taken. Therefore it is necessary to prevent potential contamination of water resources with free-living amebas and infectious agents using new disinfectant and treatment methods. Since the holy city of Mashhad receives millions of pilgrims annually, it is necessary to provide sufficient information for preventing contamination, especially in high risk groups by public health organizations. Recently, molecular characteristics are more accurate for genotyping these organisms. Therefore, further study is needed to confirm pathogen and non-pathogen amoebas by molecular and sequencing methods in Mashhad.