1. Background
Epiphytic or endophytic fungi spend a part of their lifecycle outside or inside leaf tissues, with no negative impact on the host (1). Endophytic fungi, which have been reported in numerous plant species, are important components of the forest community and significantly contribute to the diversity of natural ecosystems (2). It has also been shown that some fungal endophytes can produce various bioactive chemicals and have potential applications in biocontrol and resistance (3). They play important roles in recycling nutrients in natural ecosystems (4, 5). Medicinal plants represent an important health and economic component of biodiversity; therefore, it is essential to make a complete inventory of the medicinal components of each country’s flora, in order to facilitate conservation and sustainable use (6). According to Mossa et al. (7), the Kingdom of Saudi Arabia possesses a wide range of flora, consisting of a large number of medicinal herbs, shrubs, and trees; folk medicine has been practiced since time immemorial.
Previous studies have reported the isolation and identification of endophytic fungi and bacteria from medicinal plants such as Cressa cretica, Achillea fragrantissima, and Artemisia species (8-10). A few other studies have evaluated the antimicrobial activity of the fungal endophytes against plant pathogenic fungi such as Neurospora sp. (11), Magnaporthe grisea, Corticium sasakii, Botrytis cinerea, Phytophthora infestans, Puccinia recondita, Blumeria graminis (12), Alternaria alternata, Fusarium oxysporum, B. cinerea, and Pythium ultimum (13).
2. Objectives
In the present investigation, we evaluated the diversity of endophytic fungi colonizing seven species of medicinal plants (Alhagi graecorum, C. cretica, Citrullus colocynthis, Tamarix nilotica, A. fragrantissima, Artemisia sieberi, and Neurospora retusa), collected from salt marshes of the Al-Gouf governorate in North Saudi Arabia. The molecular characterization and antimicrobial activity of these isolates were investigated.
3. Materials and Methods
3.1. Collection of Plant Samples
A total of 70 stem and roots samples of seven disease-free medicinal plants, namely A. graecorum, C. cretica, C. colocynthis, T. nilotica, A. fragrantissima, A. sieberi, and N. retusa, were randomly collected from the salt marshes of Al-Gouf Governorate, Kingdom of Saudi Arabia, during the period from July to September 2013. The selected plants that belonged to different families are listed in Table 1.
| S/NO | Scientific Name | Family | Common Name | Part of Plant Used | Collection Site |
|---|---|---|---|---|---|
| 1 | Achillea fragrantissima | Asteraceae | Lavender cotton | Stem and roots | Al-Gouf Governorate |
| 2 | Alhagi graecorum | Fabaceae | Camel thorn | Stem and roots | Al-Gouf Governorate |
| 3 | Artemisia sieberi | Asteraceae | Desert wormwood | Stem and roots | Al-Gouf Governorate |
| 4 | Citrullus colocynthis | Cucurbitaceae | Bitter apple | Stem and roots | Al-Gouf Governorate |
| 5 | Cressa cretica | Convolvulaceae | Rudravanti | Stem and roots | Al-Gouf Governorate |
| 6 | Neurospora retusa | Nitrariaceae | Salt tree | Stem and roots | Al-Gouf Governorate |
| 7 | Tamarix nilotica | Tamaricaceae | Nile tamarisk | Stem and roots | Al-Gouf Governorate |
3.2. Isolation of Fungal Endophytes
The samples were rinsed gently in running tap water to remove dust and debris. The stem and roots samples were cut into three segments measuring 0.5 to 1 cm. The samples were surface sterilized according to the modified method of Dobranic et al. (14), immersed in 70% ethanol for five seconds, in 4% sodium hypochlorite for 90 seconds, and then rinsed in sterile distilled water for 10 seconds. Excess moisture was blotted using sterile filter paper. The sterilized segments were placed in Petri dishes containing PDA medium (HiMedia, Mumbai, India); these were sealed with parafilm, and incubated at 26 ± 1°C in 12-hours light/12-hours dark cycles for two to four weeks. The petri dishes were monitored daily to check the growth of fungal endophytic colonies from the segments. Fungi growing out of the plant segments were isolated and identified using morphological characteristics, according to the established procedure (15).
The colonization frequency (CF) and the percentage of the dominant endophytic fungi were calculated (13).

3.3. Antimicrobial Activity of Endophytic Fungi
Three plant pathogenic fungi obtained from the college of agriculture, King Saud University (F. oxysporum, F. solani, and Alternaria) and five human pathogenic bacteria obtained from the military hospital in Riyadh City (Enterococcus faecalis, Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, and Klebsiella pneumoniae) were used as target fungal and bacterial pathogens in this study. A dual culture technique was adopted to investigate the antimicrobial activity of fungal endophytes isolated from A. graecorum, C. cretica, C. colocynthis, T. nilotica, A. fragrantissima, A. sieberi, and N. retusa against the selected fungal and bacterial pathogens. The conidial suspension and assay plates of plant pathogenic fungi were prepared according to the method described by Gong et al. (16). The inocula and assay plates for bacterial strains were prepared as described by Pelaez et al. (17).
Endophytic fungi were grown on PDA plates. Five-day-old discs (5 mm in diameter) of endophytes were placed on three points in petri plates containing PDA medium. The target pathogens were inoculated in the center of the PDA plates. All petri dishes were incubated in the dark, and were randomly distributed. After incubation of the fungi at 25°C for 10 days, and of the bacteria at 37°C for 48 hours, the diameters (in mm) of the inhibition zones were measured. The level of inhibition was calculated by subtracting the distance (mm) of fungal and bacterial growth in the direction of an antagonist colony from the fungal growth radius. The widths of inhibition zones between the pathogen and the endophytes were grouped as follows: 10 mm (+++, strong inhibition), 2 to 10 mm (++, moderate inhibition), 1 mm (+, weak inhibition), and less than 1 mm (-, no activity determined) (18).
3.4. DNA Extraction, Amplification, and Sequencing
Two microliters of potato dextrose broth (PDB) (HiMedia, Mumbai, India) were poured into PDA tubes and vortexed to disperse the spores, and the spores-PDB mixtures were poured into flasks containing 100 mL PDB. The flasks were kept undisturbed at room temperature for two to three days. The mycelium was harvested by filtration, frozen at -80°C for 30 minutes, lyophilized, and stored at -80°C. The mycelium was ground in liquid nitrogen with a sterile mortar to obtain mycelium powder. DNA was extracted from 20 mg of mycelium powder using a DNeasy plant mini kit. The DNA quantity and quality were checked by electrophoresis on a 0.8% agarose gel, and visualized with ethidium bromide under UV trans-illumination (13).
The ITS region of the ribosomal DNA was amplified by PCR with the primers ITS1-F (5’-CTTGGTCATTTAGAGGAAGTAA-3’) and ITS4 (5’-TCCTCCGCTTATTGATATGC-3’) (19). PCR amplifications were carried out in a final volume of 50 µL, containing 2 µL of DNA, 0.5 mM of each primer, 150 mM of dNTP, 1 U of Taq DNA polymerase (Promega), and PCR reaction buffer. Amplification was carried out in a thermal cycler with an initial denaturation of three minutes at 94°C, followed by 35 cycles of one minute at 94°C, one minute at 50°C, one minute at 72°C, and a final extension of 10 minutes at 72°C. The amplified products were checked by electrophoresis on a 1% agarose gel, and visualized with ethidium bromide under UV trans-illumination. The PCR products were purified using an ExoSAPIT kit (USB Corporation, Amersham Place, UK, under license from GE Healthcare), based on the manufacturer’s instructions. The purified products were sequenced in an automated DNA sequencer (ABI PRISM 3700) using the BigDye Deoxy Terminator cycle-sequencing kit (Applied Biosystems, Darmstadt, Germany), following the manufacturer’s instructions. Sequences were submitted to GenBank, NCBI (http://www.ncbi.nl m.nih.gov). Sequences obtained in this study were compared with the previously deposited sequences in the GenBank database, using BLAST, on the NCBI website (http://www. ncbi.nlm.nih.gov/BLAST/).
3.5. ITS Sequence and Phylogenetic Analysis
DNA sequences were initially aligned with Clustal X 1.81 (20). TREECON (21) for Windows (version 1.3b, 1998) was used to construct a neighbor-joining tree using the Jukes-Cantor model.
4. Results
This is probably the first report to describe the endophytic fungi that colonize the medicinal plants A. graecorum, C. cretica, C. colocynthis, and N. retusa in the northern desert of Saudi Arabia. A total of 70 stem and root samples were derived from seven medicinal plants, and were screened for the presence of endophytic fungi. 275 isolates belonging to 23 species and 14 genera were obtained from stem and root segments of A. graecorum, C. cretica, C. colocynthis, T. nilotica, A. fragrantissima, A. sieberi, and N. retusa. The isolates were identified as follows: one species of Alternaria from 23 isolates, seven species of Aspergillus from 68 isolates, one species of Chaetomium from two isolates, one species of Drechslera from two isolates, one species of Emericella from one isolate, one species of Eupenicillium from three isolates, four species of Fusarium from 14 isolates, one species of Gibberella from four isolates, one species of Monilia from one isolate, one species of Mucor from four isolates, one species of Mycelia from 65 isolates, one species of Penicillium from 81 isolates, one species of Scopulariopsis from three isolates, and one species of Ulocladium from four isolates. The most commonly isolated species were P. chrysogenum with an overall colonization frequency of 98.57%, followed by Mycelia sterilia with a colonization frequency of 92.86% (Table 2).
| Fungal Endophyte | Isolate Numbers | CFa | Dominant Fungia |
|---|---|---|---|
| Alternaria alternata | 23 | 32.86 | 8.4 |
| Aspergillus candidus | 2 | 2.86 | 0.73 |
| A. flavus | 1 | 1.43 | 0.36 |
| A. niger | 19 | 27.14 | 6.91 |
| A. ochraceous | 30 | 42.86 | 10.91 |
| A. oryzae | 1 | 1.43 | 0.36 |
| A. sydowii | 4 | 5.71 | 1.45 |
| A. terreus | 11 | 15.71 | 4 |
| Chaetomium subaffine | 2 | 2.86 | 0.73 |
| Drechsleria indica | 2 | 2.86 | 0.73 |
| Emericella quadrilineata | 1 | 1.43 | 0.36 |
| E. crustaceum | 3 | 4.3 | 1.1 |
| Fusarium brachygibbosum | 1 | 1.43 | 0.36 |
| F. nygamai | 1 | 1.43 | 0.36 |
| F. oxysporum | 11 | 15.71 | 4 |
| F. cf. solani | 1 | 1.43 | 0.36 |
| Gibberella moniliformis | 4 | 5.71 | 1.45 |
| Monniliasp. | 1 | 1.43 | 0.36 |
| Mucorsp. | 4 | 5.71 | 1.45 |
| Mycelia sterilia | 65 | 92.86 | 23.64 |
| Penicillium chrysogenum | 69 | 98.57 | 29.45 |
| Scopulariopsissp. | 3 | 4.3 | 1.1 |
| Ulocladium atrum | 4 | 5.71 | 1.45 |
| Total | 263 | NA | NA |
aValue’s unit is %.
A total of 263 endophytic fungal isolates were obtained from 70 stem and root fragments. Thirty five isolates were selected based on their antimicrobial activity, and identified based on the sequencing of the ITS region of rDNA (Table 3).
| NO. | Isolate Codes | Accession Numbers | The Closet Genbank Taxa | Similarity, % |
|---|---|---|---|---|
| 1 | TUEF1 | HF546360 | Penicillium chrysogenum FJ613114 | 99 |
| 2 | TUEF2 | HF546361 | P. chrysogenum EF200090 | 99 |
| 3 | TUEF3 | HF546362 | P. chrysogenum FJ613114 | 98 |
| 4 | TUEF4 | HF546363 | Scopulariopsis flava AB627114 | 93 |
| 5 | TUEF5 | HF546364 | Gibberella moniliformis AB587011 | 100 |
| 6 | TUEF6 | HF546365 | Aspergillus sydowii EF652473 | 99 |
| 7 | TUEF7 | HF546366 | Emericella quadrilineata EF652433 | 100 |
| 8 | TUEF8 | HF546367 | A. sydowii EF652473 | 99 |
| 9 | TUEF9 | HF546368 | Gibberella moniliformis AB587011 | 99 |
| 10 | TUEF10 | HF546369 | Eupenicillium crustaceum AF033466 | 99 |
| 11 | TUEF11 | HF546370 | P. chrysogenum AF034451 | 99 |
| 12 | TUEF12 | HF546371 | Scopulariopsis sp. EU821474 | 87 |
| 13 | TUEF13 | HF546372 | G. moniliformis AB587011 | 99 |
| 14 | TUEF14 | HF546373 | A. sydowii EF652473 | 99 |
| 15 | TUEF15 | HF546374 | Chaetomium subaffine HM365247 | 100 |
| 16 | TUEF16 | HF546375 | A. terreus EF669586 | 100 |
| 17 | TUEF17 | HF546376 | E. crustaceum AF033466 | 99 |
| 18 | TUEF18 | HF546377 | Fusarium cf. solani JN235174 | 99 |
| 19 | TUEF19 | HF546378 | Scopulariopsis sp. EU821474 | 86 |
| 20 | TUEF20 | HF546379 | P. chrysogenum AF033465 | 100 |
| 21 | TUEF21 | HF546380 | C. subaffine HM365247 | 99 |
| 22 | TUEF22 | HF546381 | F. nygamai X94174 | 100 |
| 23 | TUEF23 | HF546382 | P. chrysogenum FJ613114 | 99 |
| 24 | TUEF24 | HF546383 | A. sydowii EF652450 | 99 |
| 25 | TUEF25 | HF546384 | P. chrysogenum GQ241341 | 99 |
| 26 | TUEF26 | HF546385 | F. brachygibbosum GQ505450 | 99 |
| 27 | TUEF27 | HF546386 | P. chrysogenum EF200090 | 99 |
| 28 | TUEF28 | HF546387 | G. moniliformis AB587011 | 99 |
| 29 | TUEF29 | HF546388 | P. chrysogenum AF033465 | 95 |
| 30 | TUEF30 | HF546389 | P. chrysogenum FJ613114 | 99 |
| 31 | TUEF31 | HF546390 | P. chrysogenum AF034451 | 99 |
| 32 | TUEF32 | HF546391 | E. crustaceum AF033466 | 99 |
| 33 | TUEF33 | HF546392 | A. oryzae EF591304 | 100 |
| 34 | TUEF34 | HF546393 | F. oxysporum EU214567 | 99 |
| 35 | TUEF35 | HF546394 | F. oxysporum EU214567 | 99 |
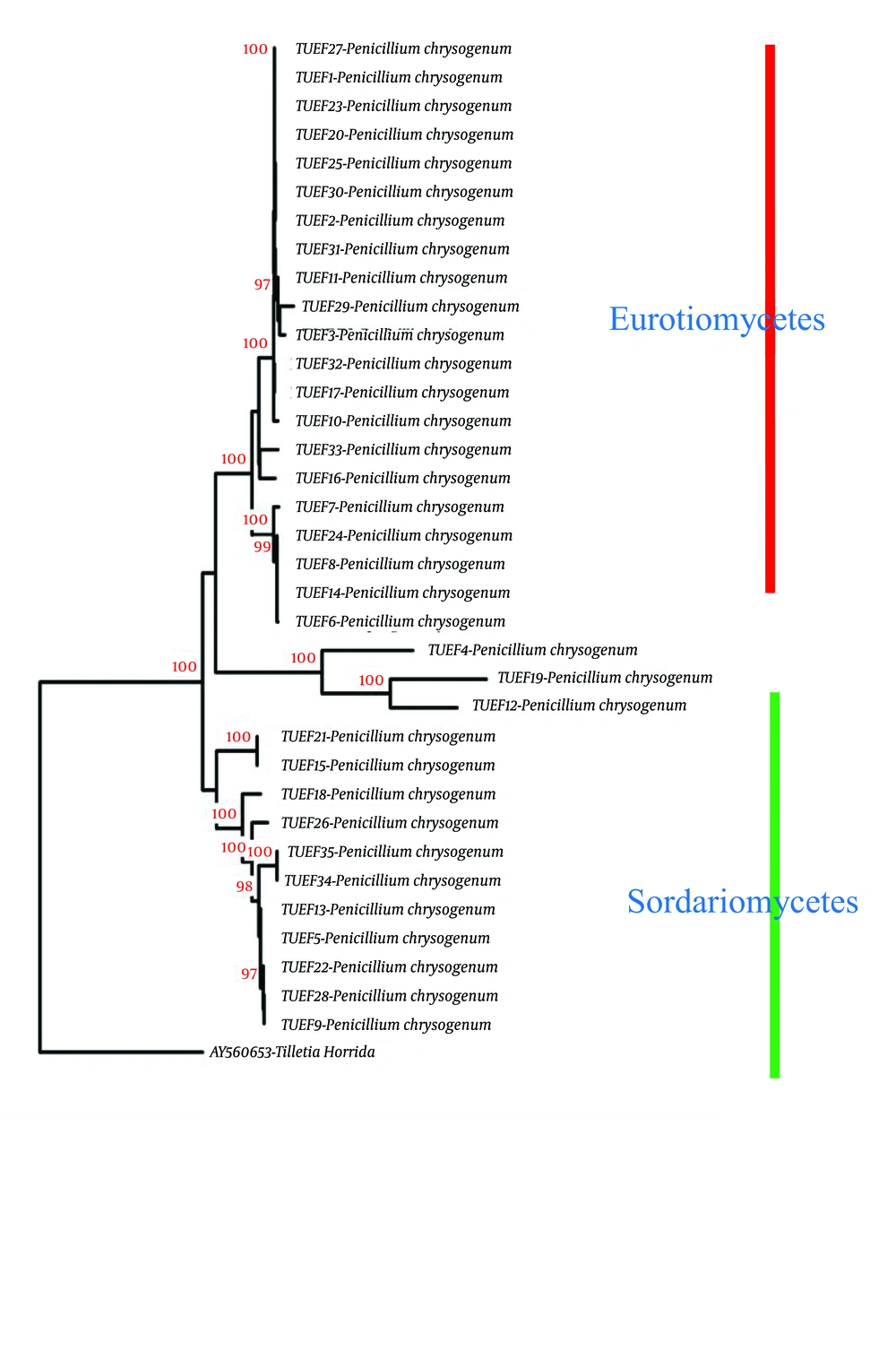
The sequence results corroborated with the morphological identification of the isolated fungal
endophytes (Figure 1). Most of the isolates belonged to the taxa Ascomycota (58.54%), Deuteromycota (39.27%), or Zygomycota (1.45%). All seven medicinal plants were found to host one or more endophytes. Different endophytic taxa showed different relative frequencies on different plants (Table 4). T. nilotica had the highest endophytic diversity (relative frequency 27.27%), followed by C. cretica (relative frequency 19.27%), and C. colocynthis had the lowest endophytic diversity (relative frequency 5.82%) among the seven medicinal plants used in this study.
aValue’s unit is %.
4.1. Antimicrobial Activity of Endophytic Fungi
All endophytic fungi exhibited significant inhibition against a wide range of plant pathogenic fungi and human pathogenic bacteria. The isolates, namely P. chrysogenum (TUEF2), E. crustaceum (TUEF10), A. sydowii (TUEF14), F. brachygibbosum (TUEF26), P. chrysogenum (TUEF30), P. chrysogenum (TUEF31), and E. crustaceum (TUEF32) showed strong inhibition towards plant pathogenic fungi.
thirty one isolates in this work showed promising growth-inhibitory activity against at least one of the test human pathogenic bacteria, but no endophyte had antimicrobial activity against all five pathogenic microbes. A high proportion of fungi (54.3%) had activity against K. pneumonia. The numbers of fungal isolates displaying antimicrobial activity against S. aureus, E. faecalis, E. coli, P. aeruginosa, and K. pneumonia were 10, 13, 6, 18, and 19, respectively. The isolates P. chrysogenum (TUEF27), P. chrysogenum (TUEF23), F. oxysporum (TUEF35), and F. nygamai (TUEF22) displayed the highest inhibition against the human pathogenic bacteria S. aureus, E. coli, P. aeruginosa, and K. pneumonia, respectively. The isolates A. sydowii (TUEF6), P. chrysogenum (TUEF23), and E. crustaceum (TUEF32) showed strong activity against E. faecalis (Table 5).
| Fungal Endophyte | Isolate Number | Plant Pathogenic fungib | Human Pathogenic Bacteriab | ||||||
|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | H | ||
| Penicillium chrysogenum | TUEF1 | +++ | ++ | +++ | - | - | - | ++ | - |
| P. chrysogenum | TUEF2 | +++ | +++ | +++ | - | - | - | - | - |
| P. chrysogenum | TUEF3 | +++ | ++ | +++ | - | - | - | - | ++ |
| Scopulariopsis flava | TUEF4 | ++ | +++ | +++ | - | ++ | - | - | ++ |
| Gibberella moniliformis | TUEF5 | ++ | +++ | ++ | - | - | - | - | - |
| Aspergillus sydowii | TUEF6 | ++ | +++ | ++ | - | +++ | - | - | ++ |
| Emericella quadrilineata | TUEF7 | +++ | +++ | - | ++ | ++ | ++ | - | ++ |
| A. sydowii | TUEF8 | +++ | +++ | - | - | - | - | ++ | - |
| G. moniliformis | TUEF9 | + | ++ | +++ | - | - | - | ++ | - |
| E. crustaceum | TUEF10 | +++ | +++ | +++ | - | - | - | - | - |
| P. chrysogenum | TUEF11 | +++ | +++ | - | - | - | - | ++ | - |
| Scopulariopsis sp. | TUEF12 | +++ | ++ | +++ | - | - | - | - | - |
| G. moniliformis | TUEF13 | +++ | +++ | - | - | - | ++ | - | ++ |
| A. sydowii | TUEF14 | +++ | +++ | +++ | - | - | - | ++ | - |
| Chaetomium subaffine | TUEF15 | +++ | +++ | ++ | - | ++ | - | - | ++ |
| A. terreus | TUEF16 | +++ | +++ | - | ++ | - | - | - | - |
| E. crustaceum | TUEF17 | +++ | +++ | - | - | ++ | - | ++ | ++ |
| Fusarium cf. solani | TUEF18 | +++ | +++ | ++ | ++ | - | - | ++ | ++ |
| Scopulariopsissp. | TUEF19 | +++ | +++ | + | - | ++ | - | ++ | ++ |
| P. chrysogenum | TUEF20 | +++ | + | +++ | ++ | ++ | - | - | ++ |
| Chaetomium subaffine | TUEF21 | +++ | ++ | ++ | - | - | - | ++ | - |
| F. nygamai | TUEF22 | ++ | +++ | ++ | ++ | - | ++ | ++ | +++ |
| P. chrysogenum | TUEF23 | ++ | +++ | ++ | - | +++ | +++ | ++ | ++ |
| A. sydowii | TUEF24 | +++ | +++ | ++ | ++ | - | - | - | +++ |
| P. chrysogenum | TUEF25 | +++ | +++ | - | - | ++ | - | - | - |
| F. brachygibbosum | TUEF26 | +++ | +++ | +++ | ++ | - | - | ++ | ++ |
| P. chrysogenum | TUEF27 | +++ | +++ | - | +++ | - | - | ++ | ++ |
| G. moniliformis | TUEF28 | +++ | +++ | - | ++ | - | - | - | ++ |
| P. chrysogenum | TUEF29 | ++ | +++ | ++ | - | ++ | - | - | ++ |
| P. chrysogenum | TUEF30 | +++ | +++ | +++ | - | ++ | - | ++ | - |
| P. chrysogenum | TUEF31 | +++ | +++ | +++ | - | - | - | - | - |
| E. crustaceum | TUEF32 | +++ | +++ | +++ | - | +++ | - | ++ | - |
| A. oryzae | TUEF33 | +++ | +++ | - | ++ | - | ++ | ++ | - |
| F. oxysporum | TUEF34 | +++ | +++ | - | - | - | - | ++ | ++ |
| F. oxysporum | TUEF35 | ++ | +++ | ++ | - | ++ | ++ | +++ | ++ |
aInhibition Zone, (-) No activity determined; +, > 2 mm; ++, 2 - 10 mm; +++, < 10 mm.
bF. oxysporum
5. Discussion
275 isolates, belonging to 23 species and 14 genera, were recovered from the stem and root segments of seven medicinal plants collected from salt marshes in North Saudi Arabia. Penicillium chrysogenum was the most frequently isolated species with a colonization frequency of 98.57%, followed by Mycelia sterilia with a colonization frequency of 92.86%. High colonization rates, ranging from 81 - 89%, were reported in palms in Brunei and Australia (22), and up to 95 - 98% in leaf fragments of Guarea guidonia (Meliaceae) in Puerto Rico (23). Fisher et al. (24) found that Camarosporium spp. (85.3% in stems) and Colletotrichum gloeosporioides (30% in leaves) endophytically colonized Suaeda fruticosa in England. Crabtree et al. (25) found that the salt marsh fungus Camarosporium roumeguerii, occurring on plants belonging to Chenopodiaceae, produced a dark green pigment. Other fungi such as Pleospora spp., Stemphylium spp., Cladosporium spp., and Camarosporium spp. were always isolated from plants in deserts and saline lands (26, 27). A possible explanation for the relatively low overall colonization rate noted in this study could be the high salinity of salt marshes at the collection site, and the desert nature of Saudi Arabia.
Molecular analysis of fungal rDNA sequences provides a powerful technique for assessing fungal diversity at the genus level. Most fungal isolates described in this study belong to Ascomycota (58.54%), Deuteromycota (39.27%), and Zygomycota (1.45%), confirming the previous reports by Huang et al. (10) and other reports on the fungal endophytes of T. nilotica, A. fragrantissima, and Artemisia plants from various locations (9, 28). The ITS sequences of the isolated species submitted to EMBL-EBI were compared with the previously deposited sequences using BLAST. Isolates used for the sequencing analysis, their codes, and GenBank accession numbers are listed in Table 3. Most of the isolates belonged to Ascomycota, which confirms Petrini’s findings (29) that fungal endophytes are mainly ascomycetes. Khan et al. (30) reported that all the endophytic fungi, collected from Calotropis procera and situated at different locations within Karachi university campus, belonged to Ascomycetes. Similar results were also obtained by Gherbawy et al. (13) during their research on C. procera from the Taif region of Saudi Arabia.
Some of the fungi isolated in this study (A. alternata, G. moniliformis, and some species of M. sterilia) are well-known plant pathogens. It is known that an endophyte in one plant may act as a pathogen in another plant, depending on the balance between the pathogenicity and endophytism of the microorganism in different hosts. The fungi isolated during this study have previously been isolated as endophytes from a wide range of different host plants such as Pinus tabulaeformis (31), Avicennia marina (32), ginseng leaves (33), Thymus decussatus (34), and C. procera (13). Endophytic fungi were tested for antifungal activity using a dual culture method. The results clearly showed that the endophytic fungi have stronger inhibition against plant pathogenic fungi, compared to human pathogenic bacteria. This could be because the endophytic fungi and plant pathogenic fungi naturally exist in the same habitat.
The antibacterial activity of the endophytic isolates from medicinal plants shown in this work against human pathogenic bacteria indicates the probable role of endophytes in the medicinal activity of these plants, partially agreeing with Jahanpour et al. (6), who demonstrated that the ethanolic extract of P. harmala seed and P. granatum peel exhibited potential activity against all Mycobacterium tuberculosis isolates with a mean inhibitory zone of 18.7 and 18.8 mm, at a 200 mg/mL concentration. Several groups (11, 12, 18) have previously reported the antifungal activities of plant endophytic fungi and actinomycetes. Bernardi-Wenzel et al. (35) evaluated the antibacterial activity of nine endophytic fungi isolated from leaves of Luehea divaricata against phytopathogens and the human pathogenic bacteria E. coli and S. aureus. The endophytes had varied effects on A. alternata. One isolate produced an inhibition halo against Moniliophthora perniciosa and E. coli.
Cui et al. (36) isolated 28 fungal endophytes from agarwood and found that 13 (46.4%) showed antimicrobial activity against at least one of the test human pathogenic bacteria. The diameters of the inhibition zones of YNAS07, YNAS14, HNAS04, HNAS05, HNAS08, and HNAS11 were equal to or higher than 14.0 mm against S. aureus, E. coli, Bacillus subtilis, B. subtilis, A. fumigatus, and B. subtilis, respectively. Production of active metabolites by endophytes may be related to the characters of the host plants and to a genetic recombination of the endophyte with the host that may have occurred in evolutionary time.
This is probably the first study that demonstrates the diversity of molecular identification of endophytic fungi from the stems and roots of medicinal plants, collected from salt marshes in Al-Gouf Governorate, Saudi Arabia. This is also perhaps the first study that has examined these host plants. The successful colonization of these plants by such fungi suggests that they can be utilized in future applications, such as delivery of degradative enzymes for controlling certain plant diseases. Meanwhile, the use of endophytes as producers of bioactive agents will help in the conservation of medicinal plants and the maintenance of environmental biodiversity.