1. Background
Influenza A virus infects a wide variety of animals and, also, human hosts. Among the avian influenza virus (AIV) subtypes, H9N2 virus has the potential to cause influenza pandemic and vaccination is a prevalent solution for this problem. The vaccine used for rapid interposition should be safe to use and highly effective, after administration (1). Aluminum salts and oil-based emulsions were previously used as adjuvant, to enhance the immunogenicity of inactivated influenza vaccines (2). Chitosan was introduced as an effective adjuvant for delivery of biological materials, such as drugs, and also, vaccines containing specially inactivated viral types, such as influenza, in several publications. Chitosan adjuvant vaccines enhanced antibody titers against influenza, in comparison to vaccines without chitosan (3, 4).
Since the 1970s, the ecology of influenza viruses in birds has been better understood, when surveillance studies showed the enormous pool of viruses presenting in the feral bird population, particularly waterfowl, and the great variation in these viruses. At present, delivery-depot effect, or specific immune activation, are regarded as two mechanisms constituting the main core of all recently developed adjuvant systems. However, multiple kinds of adjuvant systems have been extended and approved, by preclinical methods, and several of them are useful for human beings. The first restrictions to the application of recent adjuvant systems for medicine concern the safety issues. However, investigation and research plans have decreased the toxicity of adjuvants, over the last 80 years. The safety barriers, presented by regulatory and liability issues, have continued to increase. In medicine, the safety issues are more fundamental for prophylactic vaccines. As a matter of fact, the vaccines given to infants or children, today, heighten the safety concerns of vaccine adjuvants (1).
There are different methods by which adjuvants can improve the immune response against vaccines: a) Developing the immunogenicity of faint antigens; b) Boosting the velocity and the length of the immune response; c) Adjusting antibody avidity, specification, isotype, or subclass dissemination; d) Stimulation of cytotoxic T lymphocyte response; e) Increasing the induction of mucosal immunity; f) Reducing the antigen volume in the vaccine, for lower costs.
Prophylaxis of influenza has been successfully used for more than 50 years for inactivated influenza vaccines. However, the results of presenting inactivated vaccines are less impressive in the aged population and are incapable to protect from influenza virus drift variants. Chitosan is a polymer formed by the reaction between two different monomers, with units of more than one kind of glucosamine and N-acetyl glucosamine, taken from the sectional depolymerization and deacetylation of chitin. Characteristically, is a biocompatible, biodegradable, non-toxic polymer. Moreover, chitosan was found to represent immune adjuvant characteristics, by improving humoral and cell-mediated immune responses, followed by inducing vaccination (3).
2. Objectives
This research is based on the optimization of chitosan concentration, for vaccine delivery, and, also, the evaluation of antigen loading capacity for vaccine preparation. Chitosan nanoparticles (CNP) represent an interesting new platform for antigen delivery and a promising adjuvant candidate for H9N2 inactivated influenza vaccine.
3. Materials and Methods
3.1. Virus Stock Preparations
Standard vaccine strain AIV (A/Chicken/Iran/99/H9N2) was inoculated into 11-day-old specific pathogen-free (SPF) eggs. The eggs were observed for 24 - 72 hours post inoculation, according to the International Office of Epizootics (OIE) Terrestrial Manual (2008). The amnion-allantois fluids of the inoculated eggs were collected and centrifuged at 1200 rpm, for 30 minutes (5). The 50% of egg-infective dose (EID50) was calculated by Reed and Munch method (1938) in 11-day-old embryonic SPF eggs. Hemagglutination assay (HA) was performed in V-bottom 96-well plates, with 1% chicken red blood cell, as described by Burleson et al. (1992) (6).
3.2. Egg Infective Dose 50 (EID50)
The EID50 assay was carried out to measure the concentration of infectious influenza disease virus in a suspension, where every well is represented by an unique egg. Each virus dilution is injected into replicate eggs, which are tested, after a period of incubation, for signs of virus growth. In case of influenza, we use the HA. The infectivity titer of a suspension of influenza disease virus is the number of infectious units of virus per unit volume, usually expressed per ml. An adaptation of the mathematical technique, devised by Reed and Muench (1938), is used to calculate the dilution of the suspension of virus being tested that produced the end point. The end point contains one unit of infectivity (1 EID50). This dilution is then used to calculate the infectivity titer.
3.3. Formalin Treatment (Virus Inactivation)
Formaldehyde (Merck KGaA, Darmstadt, Germany) solution was diluted in double-distilled water. The final concentration of 0.1% was added to virus solution. The viruses were mixed and incubated for 16 hours, at 37°C.
3.4. Hemagglutination Assay (HA)
The HA identifies the presence of certain viruses that agglutinate red blood cells. The presence of virus will hold the red cells in a diffuse matrix and prevent them from settling out to the bottom of the well.
3.5. Hemagglutination-Inhibition Assay (HAI)
The test is performed based on the inhibition of viral agglutination, by a specific antibody, and can be used for virus identification or antibodies measurement.
3.6. Chitosan Nanoparticles
The CNP (Sigma-Aldrich, St. Louis, MO, USA) were prepared according to a modified ionic gelation method. In this procedure, deacetylation of chitosan was 95% and was distinguishable by essential analysis. Molecular weight of chitosan, in this study, was 220 kDa, as specified by isometric methods. Chitosan solution was prepared by dissolving 0.37 g of both low and medium molecular weights in 89.25 ml of acetic acid (0.1 M) and heated at 37°C, to dissolve. In the next step, the solution was filtered using pre-filter membranes under vacuum (for excluding large particles). Then, 160.78 ml of acetate sodium (0.1 M) were added to the prepared solution. In this study, chitosan derivatives solution 0.5% and 1% (deacetylated by 85%) in 0.2 M glutamate buffer were used, for immune response activity. Fleetingly, chitosan was dissolved in different concentrations (0.5% and 1%) of acetic acid. For better dissolving and to form an unique complex with microorganisms, especially viruses, a pH 4.6 - 4.8 must be reached, by using NaOH (7).
Inactivated influenza virus was added to the prepared solution to reach a final concentration of 5 hemagglutination units (HAU) (suspension was gently stirred for 10 min). Tween 80 (Sigma-Aldrich, St. Louis, MO, USA) was added to suspension, in concentration of 0.01%, as an emulsifier. Then, sodium tripolyphosphate (TPP) was separately dissolved in deionized water (80 ml), at a concentration of 0.1% w/v. Nanoparticles were formed instantaneously, upon the drop-wise addition (using pumping device) of TPP solution to chitosan solution. The nanoparticle suspensions were gently stirred for 60 minutes, at room temperature, before characterization. The suspension was centrifuged at 10000 rpm, for 15 min, to remove the supernatant containing free AIV. The loading capacity of CNP were calculated by difference of supernatant HA titer and initial antigen loading. Remaining pellets were dissolved in equal volume of citrate buffer [92 ml of sodium citrate (0.1 M) in 8 ml of citric acid (0.1 M)] (3, 7).
3.7. Study Plan
For vaccine-antibody response assay, different solutions of prepared vaccines (0.5% and 1% of chitosan) were injected via intramuscular route in 21-day-old SPF chickens. Vaccine induced AIV-specific antibodies, after single dose vaccination. Sera were collected every week after vaccination, for seven weeks, and measured by hemagglutination inhibition (HI) test.
Fifty SPF broilers (21 days old) were randomized into five groups of 10 chickens. Before the chickens were introduced into the laboratory experimental facilities, they were tested for the major viral diseases, affecting chickens. The SPF birds were maintained in air-filtered bio-security isolation units, with feed and water under controlled condition.
Group 1 (with chitosan of low molecular weight and a concentration of 0.5%) was vaccinated with CNP-AIV vaccine (0.2 ml/chicken); group 2 (with chitosan of low molecular weight and a concentration of 1%), group 3 (with chitosan of medium molecular weight and a concentration of 0.5%) and group 4 (with chitosan of medium molecular weight and a concentration of 1%) were vaccinated with the same volumes of vaccine, respectively. All vaccines were delivered through intramuscular route. Blood samples were collected on day 0 (vaccination day) and at the end of each week (for 7 week) post vaccination. Sera were separated, heat inactivated and stored at -20°C, for future use.
3.8. Statistical Analysis
All data were analyzed by using SPSS version 17 (SPSS Inc., Chicago, IL, USA) statistical package and correspondences evaluated using one - way ANOVA tests.
4. Results
4.1. Loading Capacity
The loading capacity of CNP was calculated by using the HA method. The maximum avian influenza antigen loading capacity for our nanoparticles was of four HAU.
4.2. Clinical Evaluations
No respiratory or clinical complications were observed right after vaccination and in the weeks following vaccination. The vaccines were, therefore, safe to use.
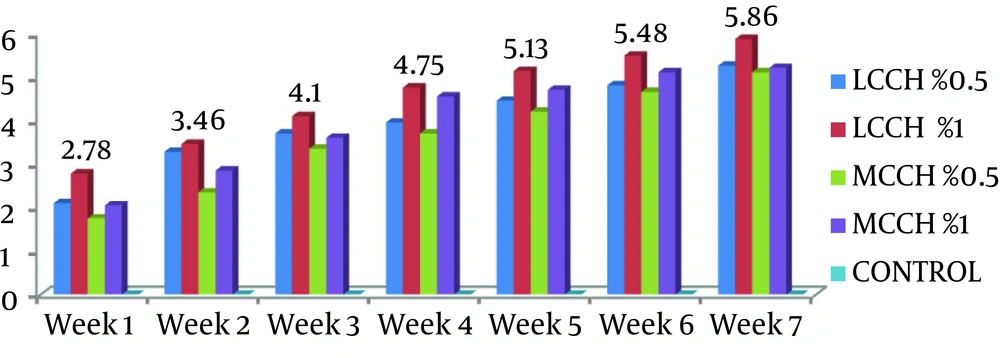
Mean HI titers, in the different weeks of the study groups, are shown in Figure 1 and Table 1. The mean HI titer for the group of chitosan with low molecular weight and a concentration of 1% was significantly higher than that of the other groups (P < 0.05).
| Groups | Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 |
|---|---|---|---|---|---|---|---|
| LCCH 0.5% | 2.10 | 3.28 | 3.70 | 3.95 | 4.45 | 4.80 | 5.25 |
| LCCH 1% | 2.78 | 3.46 | 4.10 | 4.75 | 5.13 | 5.48 | 5.86 |
| MCCH 0.5% | 1.75 | 2.35 | 3.35 | 3.70 | 4.20 | 4.65 | 5.10 |
| MCCH 1% | 2.05 | 2.85 | 3.60 | 4.55 | 4.70 | 5.10 | 5.20 |
| Control | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
aAbbreviations: LCCH, Low Molecular Weight of Chitosan; MCCH, Medium Molecular Weight of Chitosan.
5. Discussion
An efficient vaccine requires antigens for being presented to the immune system and it would be have maximal results if combined with suitable and premiere adjuvant, for long-term immunity, and also for increasing the immunogenicity. Adjuvants that were utilized in boosting humoral and cellular immunity had a limited release from the injection site. On the other hand, the side effects of adjuvants, instead of swelling, tissue injury and also pain, are very important in histopathology for medicine development and vaccine protocol guidelines. Oily inactivated vaccines could enhance the immune responses and lengthen duration of immunity. Meanwhile, residues of the adjuvant, especially mineral, at the injection site, caused multiple problems, like tissue damage and certain cases of necrosis, which could present carcinogenic transformation and decrease the standard commercial values (8, 9). Therefore, CNP adjuvant was used to evaluate adjuvant effects. Chitosan adjuvant is a polymeric particle adjuvant. In chitosan loaded vaccines, the use of antigen was obviously reduced and the vaccine caused clearly much less inflammatory response, after inoculation. Also, chitosan adjuvant has immune enhancing effects (10, 11).
This research also paved the way for the introduction of the optimum concentration of chitosan for nanoparticle preparation and, also, realized an evaluation of the loading of AIV into CNP. Although aluminum salts are used in the vaccine industry, for many years, however, research in this field, in human being, did not have high efficacy (12). Due to the ability of stimulating local factors, the mucosal affinity of adjuvant is the goal in vaccine preparation and administration. Recently, scientists indicated the potential efficacy of CNP that were formulated by inactivated flu virus, for intranasal route administration (13).
Ghendon et al. (2008) (14) showed that chitosan-adjuvant formulations also significantly increased antibody titers against homologous and heterologous virus strains. Chitosan, with an antigen increased proliferation of antigen-specific CD4+ T lymphocytes in mice spleen, as well as the quantities of Th-1 lymphocytes, amplifying T-helper function and inducing cytotoxic T-lymphocytes. Ghendon et al., (2008) (14, 15) represented that using low and high molecular weights of chitosan, with inactivated influenza, enhanced the activity of cytotoxic natural killer (NK) lymphocytes. This process could be more effective when the nanoparticles used low molecular weights, by increasing CD3, CD3/NK and CD25 T-lymphocytes (16).