1. Background
Toxoplasma gondii is a microscopic protozoa that causes a disease called toxoplasmosis. Primary hosts are cats, but humans and other warm-blooded vertebrates are intermediate hosts. It can cause a serious disease in pregnant women and immune-compromised subjects (1, 2). Initial subclinical infections sometimes lead to chorioretinitis. If the parasites enter the body, for example through an AIDS-positive virus or tissue transplantation, they can damage the fetus (3). There are several case reports demonstrating that diabetic patients have an increased susceptibility to infection. Many infections are more common in diabetic patients, and some are almost exclusive to that specific population. However, other nontoxoplasmic infections can occur to increase the risk of complications in diabetic patients (4, 5). For people who are at risk, timely detection of T. gondii can prevent the disease’s progression (6).
Today, serological tests such as the immunofluorescence antibody assay (IFA) and the enzyme-linked immunosorbent assay (ELISA) are used for the detection and identification of toxoplasmosis. These same tests are also used in diagnosing HIV-positive patients and fetal infections in diabetic mothers, and early diagnosis means that proper treatment can be initiated. However, interpretation of serological tests is difficult in diagnosing pregnant women, and they are not useful for diagnosing cerebral abscesses and ocular toxoplasmosis in immune-compromised patients (7, 8). For the detection of T.gondii in tissue and body fluids such as amniotic fluid, the polymerase chain reaction (PCR) method can be used (9). This technique has helped identify parasites in clinical samples, and it is a powerful tool (10).
There are two types of diagnostic methods: 1, identification of DNA in biological fluids by means of PCR, nested-PCR, and real-time PCR; and 2, molecular methods, such as microsatellite analysis to identify parasite strains (11). The nested-PCR assay has been reported as the most significant and sensitive assay for diagnosing toxoplasmosis (12). In a study by Fallahi et al. conducted on children with cancer to identify T.gondii infections, the nested-PCR method showed the proper sensitivity and specificity for detecting toxoplasmosis (13). Molecular diagnostics can be a very useful tool in diagnosing T.gondii in diabetic patients. In addition to finding the appropriate target gene sequences, the exact design of primers for the correct implementation of the PCR is important. Reports indicate that procedures with multiple gene copies, such as nested-PCR, are better than a single-gene copy (14). Molecular methods used in laboratories worldwide to identify the forms of T. gondii are a 529 bp repeat element (RE) (gene bank accession number AF146527), with 200 - 300 copies (15), and B1 gene (gene bank accession number AF179871) with 35 copies (14).These are used to determine sequences in the genome of T.gondii because high specificity and sensitivity are important. Accuracy and sensitivity of the two genes (RE and B1) have been compared (16-19). Finding a positive PCR in the patient’s sero-negative toxoplasmosis is an important factor, which suggests that the results may not be reliable. This is true when the nested-PCR assay with specific primers does not show false results.
2. Objectives
The aim of this study was the detection of T.gondii in diabetic patients with RE and B1 genes of T.gondii and the comparison of these two genes for diagnosis using the nested-PCR assay method.
3. Patients and Methods
3.1. Patients and Clinical Samples
Patients and the enrollment procedure have been described in our previous study (20). Two hundred and five whole blood samples in EDTA-tubes for DNA extraction and without EDTA for preparation serum were taken from subjects with diabetes. Samples of these patients referred to the diabetic central of Ali Asghar hospital in Zahedan, Iran with instances of myalgia, abortion, blurred vision, arthralgia, malaise, headache, and amount of sugar and albuminuria were selected. The blood samples were stored at -20°C until use.
For the serum samples, an ELISA test was done previously using available kits (EIA-3519 IgG and 3520IgM, Germany). IgM and IgG antibody levels of > 14.5 UL/mL and of > 1.1 UL/mL were considered positive, respectively. Those with positive IgM and IgG were deemed the acute group, those with positive IgG and negative IgM were labeled as being in the chronic stage, and positive IgM and negative IgG were identified as false positives. The ethics committee of Zahedan University of Medical Sciences, Zahedan, Iran (number: 6253) approved the protocol of the study.
3.2. Isolation of DNA
Blood samples were taken from patients using the classical salting out extraction method, with some minor modifications, as outlined in Saremi et al. (21). Briefly, 0.5 mL blood EDTA was transferred to 1.5-mL micro tubes, and was added to 1 mL cell lysis buffer (Tris-HCl (10 mM), sucrose (11% w/v), MgCl2 (5 mM), and Triton X-100(11% v/v). Tubes were mixed for 2 minutes at room temperature and centrifuged at 3400 g. Supernatant was discarded (repeated twice). Next, 300 μL buffer II (10 mM Tris-HCl, 10 mM EDTA, and 10 mM sodium citrate) and 40 μL 10% SDS was added, and incubated at room temperature for 2 minutes. In the next stage, chloroform 600 μL and a saturated salt solution 100 μL were added to microtubes and centrifuged at 3400 g for two minutes, and the supernatant was collected and transferred to other microtube RNA and DNase free, then 700 μL cold isopropanol was added. The microtubes were centrifuged for 1 minutes at 13,000 g and for 2 minutes at 4°C. After decanting the supernatant, 700 μL cold 70% ethanol was added. The suspension was mixed and microtubes centrifuged for 1 minutes at 13,000 g at 4°C. DNA pellets were subsequently dried before being dissolved in 100 μL of distilled water. DNA extraction of T.gondii tachyzoites (RH-strain) was prepared in the department of parasitology of Tarbiat Modares university of Tehran, Iran.
3.3. Nested PCR the B1 Gene
Two pairs of oligonucleotide primers directed against the B1 gene of T.gondii (22) were used to perform a nested PCR (Table 1) with some minor modifications and conditions. The first round PCR amplifications were performed in a final volume of 20 μL. 10 µL master mix (AmpliqonTaq 2x master mix Denmark), 10 pmol /mL primer forward and revers (each of 0.7 µL), 2 µL template DNA (≈ 80 - 100 ng) and 6.6 µL DNase-free water was used for the PCR assay. Each sample was tested in duplicate for each method. PCR cycles were 95°C for 5 minutes, denaturation 95°C for 1 minute, annealing: 57°C for 1 minute, and extension: 72°C for 1 minute. A final extension was done at 72°C for 5 minutes, for 30 cycles. For PCR, a negative control without DNA and a positive-control sample of T.gondii tachyzoites RH strain (100 ng/μL) was used (product size: 287 bp). During the second run, the PCR product was diluted 1:20. To perform the 10 µL master mix (AmpliqonTaq 2 x master mix Denmark), the dilution DNA 2µL, and 10 pmol/mL primer forward and reveres (each of 1 µL) and 6 µL DNase-free water was used for the PCR assay. The cycling conditions were 95 °C for 5 minutes, 95 °C for 30 seconds, 56 °C for 1 minute, 72 °C for 30 seconds, and a final extension of 72 °C for 5 minutes at 35 cycles (product size: 194 bp), (Table 1).
| Assay | Oligonucleotide Sequence | Nucleotide Position | Gene Bank Accession Number |
|---|---|---|---|
| Nested-PCR B1 | AF179871.1 | ||
| F1: 5′-TCAAGCAGCGTATTGTCGAG | 663 - 682 | ||
| R1: 5′-CCGCAGCGACTTCTATCTCT | 949 - 930 | ||
| F2: 5′-GGAACTGCATCCGTTCATGAG | 694 - 714 | ||
| R2: 5′-TCTTTAAAGCGTTCGTGGTC | 887 - 868 | ||
| Nested-PCR RE | AF146527.1 | ||
| F1: 5′CTGCAGGGAGGAAGACGAAAGTTG | 1 - 24 | ||
| R1: 5′-CAGTGCATCTGGATTCCTCTCC | 521 - 499 | ||
| F2: 5′GTGCTTGGAGCCACAGAAGGGAC | 170 - 192 | ||
| 2: 5′GAGGAAAGCGTCGTCTCGTCTGG | 412 - 390 |
Nucleotide Sequences Primers of Nested-PCR Assays B1 and RE Used for T.gondii Molecular Determination
Table 1 shows the primer sequences designed to be used for both parasite genomes by nested-PCR. For the RE gene first-round amplification, each 20 µL PCR reaction was used 10 µL master mix (AmpliqonTaq 2x master mix Denmark), 3 µL template DNA (≈ 80 - 100 ng), and for controls 3 µL negative control without DNA and 100 ng/μL of DNA T.gondii tachyzoites RH strain as positive-control, next, 1.5 µL of each primer (10 pmol/mL), and the remainder volume, was added to DNase-free water. PCR reaction with 95° C denaturation for 5 minutes, denaturation 95 ° C for 60 seconds, annealing at 60 ° C for 90 seconds, and an extension at 72°C for 60 seconds in 35 cycles was carried out (Product size: 520 bp). In the second run, the PCR product was diluted 1:20, so that 2 µL of product was used as a template for the nested amplification. Each 20 µL reaction contained 12.5 µL of master mix, 1 µL of each primer (10 pmol/mL), 2µL of diluted product, and 3.5 µL DNase-free water. Nested PCR steps in 30 cycles were carried out in such a manner: a denaturation at 95°C for 60 seconds, annealing at 58°C for 50 seconds, and extension at 72°C for 60 seconds (product size: 243 bp). Finally, positive and negative controls and sample patiantes was verified in a 2% agarose gel containing 0.6 μg/mL ethidium bromide observed under UV light.
3.4. Statistical Analysis
The data were analyzed by SPSS 18. A chi-square test compared the sero-prevalence values with the genes RE and B1 of T. gondii. Confidential intervals at 95% and P < 0.05 were considered levels of significance.
4. Results
In line with our previous study, the number of participants in the present study is 42 men (20.5%) and 163 women (79.5%). The mean age was 37 ± 8 years. The total seroprevalence of toxoplasmosis in our previous study (20) in diabetic patients was 70.7% (145/205 cases), and encompassed 53/145 (36.6%) (Positive IgG, IgM), 72/145 (49.6%) (Positive IgG, Negative IgM), 20/145 (13.8%) (Negative IgG, Positive IgM), and 60/205 (29.3%) (Negative IgG, IgM).
We completed nested-PCR experiments with two genomic targets, RE and B1, and the following results were obtained.
4.1. B1 Gene
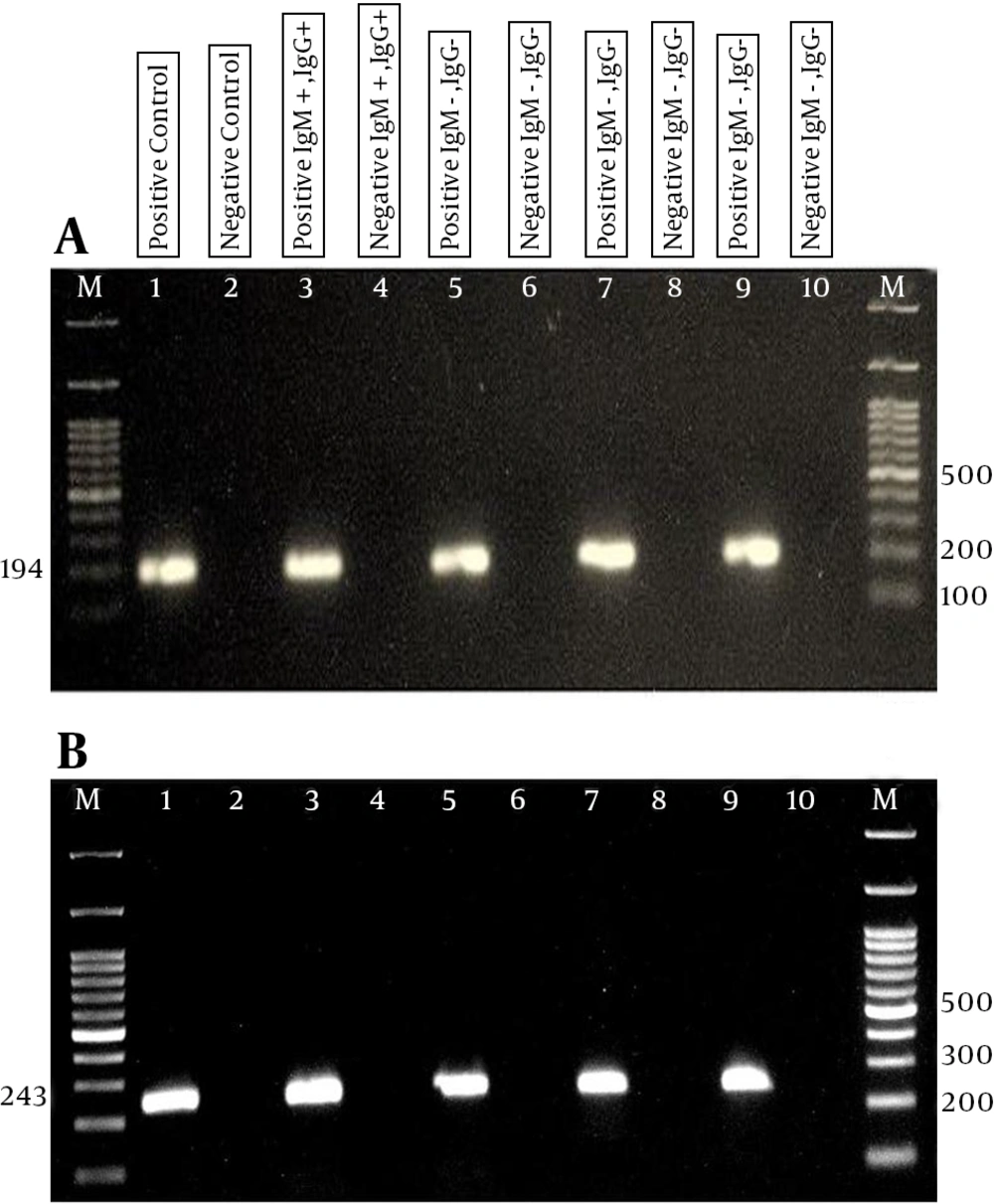
Fifty-three of the sero-positive and 60 of IgM, IgG the sero-negative nested-PCR assays in duplicate on the B1 genomic T.gondii were done, and the results showed 30 (56.60%) and 41 (77.35%) of the samples were identified as positive, respectively. In a duplicate test on the B1 genomic target, in the chronic group, 51/72 (70.83%) were false positives, and 17/20 (85%) were positive (Table 2). The prevalence of toxoplasmosis in diabetic patients, based on the B1 gene, produced 139 (67.8%) positive and 66 (32.2%) negative (Figure 1) results.
| Assay | IgG+, IgM+ | IgG-, IgM- | IgG-, IgM+ | IgG+, IgM- |
|---|---|---|---|---|
| B1-nested-PCR + | 30 (56.6) | 41 (68.3) | 17 (85) | 51 (69.8) |
| B1-nested-PCR - | 23 (43.4) | 19 (31.7) | 3 (15) | 21 (28.7) |
| RE-nested-PCR+ | 21 (39.6) | 38 (63.3) | 18 (90) | 40 (55.5) |
| RE-nested-PCR- | 32 (60.4) | 22 (36.7) | 2 (10) | 32 (44.5) |
| Total | 53 (100) | 60 (100) | 20 (100) | 72 (100) |
Comparative Results of the Nested-PCR Assays Targeting the B1 and RE on 53 (IgM+, IgG+), 60 (IgM-, IgG-), 20 (IgG-, IgM+), and 72 (IgM-, IgG+) in Patients with Diabetesa
A, B1-nested PCR analysis of T.gondii DNA; B) RE-nested PCR analysis of T.gondii DNA. Line M, ladder; line 1, positive control; line 2 negative control; lines 3 and 4 positive and negative for IgM+ and IgG+; lines 5 and 6 positive and negative for IgM- and IgG-; lines 7 and 8 positive and negative for IgM- and IgG+; lines 9 and 10 positive and negative for IgM+ and IgG-.
4.2. RE Gene
Subsequent to the duplicate RE nested-PCR on 53 sero-positive samples (IgM, IgG), 21/53 (39.63%) were found to be positive, whereas, following the duplicate RE nested-PCR on the 60 sero-negative samples, 38/60 (63.33%) were identified as positive. In the following duplicate on the RE genomic target, in the chronic group (positive IgG, negative IgM), 40/72 (55.56%) and 18/20 (90%) of the false positives (positive IgM, negative IgG) were found to be positive (Table 2). Based on this study, the baseline antibody titer for IgM and IgG on these patients’ serologic tests was positive, but in the nested-PCR assay, only 17 (B1) and 18 (RE) were positive, indicating an error in the ELISA method, which increased the report of positives. In the overall frequency of the RE gene of toxoplasmosis, 117 (57.1%) were positive and 88 (42.9%) were negative. The details of the results are presented in Figure 1 B. The data showed no significant difference between the RE and B1 genes in the detection of T.gondii (P ≥ 0.05), but the percentage of positivity of the B1 gene is more than that of the RE gene, and so it is diagnostically more effective.
5. Discussion
The results of the study on diabetic patients found that a positive PCR in patients, especially with suspected toxoplasmosis, is a valuable and important point that suggests serological results may not be reliable. We found the RE and B1 genes could be used as molecular tools via nested PCR assays in recognition of T. gondii infections. The results also show that RE is the better B1 gene, although no superiority between the two gene molecular diagnostics can be used to identify individuals.
Many factors, such as food culture, duration of exposure to the pathogen, age, and duration of diabetes can cause acute infections are in people with diabetes. Recent studies show the destruction of the pancreas for the effectiveness of insulin on the proliferation of T.gondii in pancreatic cells in patients with diabetes (4).
Reports indicate that the prevalence of T. gondii infection and diabetes worldwide and in Iran is high (4). In our previous study of diabetic patients, we found that undercooking and poor eating habits regarding the consumption of meat has an observable increase in seropositive populations. In serological tests, the reported seropositive or seronegative status is based on a cut-off obtained in the study area. This cut-off is different in each region or country. The cut-off determines positive and negative status and the prevalence of T.gondii.
The serological methods and PCR diagnosis of toxoplasmosis is not standardized for all parasitic diseases. Laboratory methods currently lack the sensitivity and accuracy for the detection of toxoplasmosis. For example, serological tests for the diagnosis of ocular T.gondii have limitations; in some cases, antibodies against the parasite may be negative, although the cause of retinal lesions is T.gondii. Conversely, despite the high sensitivity of serological tests, no signs of toxoplasmosis positive status have been seen in large populations. Positive and negative results are still a major problem (23, 24). Hence, we must run very precise and highly sensitive diagnostic methods for the parasite T.gondii in the diagnosis of ocular and cerebral toxoplasmosis, due to the high rate of mortality and morbidity and the fact that diabetic patients are very vulnerable. Nested-PCR is a very important method for evaluating appropriate cases with toxoplasmosis (25, 26).
The nested PCR method shows the sensitivity and specificity for the detection of toxoplasmosis (13), and it has been shown to be significantly more cost-effective and to have a high capacity, compared with other PCR modifications in present studies (27). Detection of T.gondii using nested-PCR can be valuable in alignment with serological methods.
Determining, the consensus on the best sequence to be amplified will be more difficult. Studies have shown that when many copies of a sequence in the genome are conserved, the high copy number can lead to increased sensitivity in the detection of a target element in routine laboratory conditions (10). The obtained RE sequence information shows that the sequence between the various strains of T.gondii is maintained. Several studies have shown that the B1 gene is highly specific for T.gondii and that it is well conserved among all the strains tested to date and sensitivity of 35-fold repeat B1 is much greater than that of the ribosomal gene (7, 27).
In this study, we found that primers specific to the genome of the parasite (two genomic repeated goals) attached and had no interference with the human genome, so they are specific for the detection of T.gondii. The findings of this study determined that the B1 gene is better than the RE for the detection of toxoplasmosis. Thus, this genomic target offers an improvement in the efficacy of PCR assays for the detection of T.gondii DNA in suspected individuals, especially in people who are at risk, including diabetic patients. Our findings are similar to the results of Cardona et al. (28) and Wahab et al. (18). They reported that the targeting of the B1 gene was more efficient than the RE genomic repeated element. They also concluded that, in the strains of the parasite, part or all of the 529 bp RE has been removed or absent in 4.8% of human T.gondii-positive samples tested. Our data were opposite the results of Reischl et al. (17), Cassaing et al. (10), and Fallahi et al. (13), as they show the RE gene may be the preferred diagnostic target over the B1 gene for the detection of T.gondii. Fallahi et al. assessed the analytical sensitivity of the nested-PCR assays, which were evaluated against a 10-fold serial dilution of T.gondii tachyzoite DNA from 1 ng to 1 fg and suggested that the sensitivity of PCR in the B1 gene is 10 to 100 times lower than the RE gene targeting (13).
In this study, of the 20 people who had a false positive result, two samples for RE genes and three samples for the B1 gene were negative for diagnosis of toxoplasmosis. In our previous study, based on the specified cut-off level in our area, they were considered positive, but using the method Nested PCR only, 17 (B1) were positive, indicating an error in the interpretation of the ELISA method results. This caused an increase in the report of positive results. The main tool used to identify and confirm people infected with T.gondii is the seronegative test. Using a nested PCR method is useful, because negative serological test is not reliable.
With attention to high prevalence of toxoplasmosis, the method nested-PCR is suitable for accurate detection of T.gondii and can be very helpful. The findings of the present study suggest that the B1 gene, in comparison with the RE gene, has no superiority of molecular recognition, although B1 genes, more so than RE genes, percent positive and is better for diagnosis.