1. Background
Haemophilus influenzae type b (Hib) is responsible for numerous upper respiratory tract infections and for the majority of bacterial meningitis cases, in children (1). The disease is rare in children less than 3 month and over 6 years (2, 3). Later studies showed that, usually, H. influenzae is a secondary infection after viral influenza (3, 4). Systemic infection with H. influenzae in children is present worldwide (5). Vaccination has been recommended in immune deficient individuals (6). The bacteria are a small, coccid, facultative anaerobic and gram negative organism (7). Non-capsular strains have lower virulence and cause otitis media in children (8-10). Considerable evidences indicates that the structure of the capsular type-specific antigen of Hib is a polyribosylribitol phosphate (PRP) polymer (11) and its antibodies play a significant protective role against H. influenzae infection (12). Detection of serum anti-PRP antibodies (PRP-Ab), by a sensitive and specific assay, is therefore of great importance. The enzyme-linked immunosorbent assay (ELISA) technique for the measurement of anti-Hib capsular polysaccharide antibodies was first successfully used in 1988 and, since then, it has been widely used all over the world (13-16). The antibody titers obtained by this technique show acute, chronic and post-vaccination steps (17). A comparison among different assays, such as PCR, loop -mediated isothermal amplification (LAMP), radio immune assay (RIA) and ELISA cleared that Elisa has multiple advantages (18-21). The antibody levels against Hib capsular polysaccharide have been investigated in children in Iran, using ELISA kits (16). Although using commercial ready to use ELISA kits is easy and convenient, sometimes, homemade ELISA kits are required because of its affordability and also because of shortage and expensiveness of commercial kits (22). Moreover, using commercial ELISA kits to detect Hib antibody titer, especially for epidemiological studies, can cost much more than homemade kits. Homemade ELISA kits for several pathogens, such as Helicobacter pylori (23) and Toxoplasma gondii (24) have been reported advantageous and cost-effective. Therefore, it was necessary to developed and optimize an indirect-ELISA plate for the detection of Hib infection in children.
2. Objectives
The H. influenzae is the most frequent causative agent of bacterial meningitis, in children aged 5 months to 5 years. The presence of anti-PRP antibody in the serum of non-vaccinated children 3-5 years old is common. Although there are different diagnostic methods to confirm the infection, the most used and preferred method is ELISA immuno-enzymatic method, as a screening test. It is necessary to prepare and develop antigen coated plates to study seroepidemiology of H. influenzae to evaluate its health impact. We designed and optimized anti-Hib enzyme immunoassay kit in our laboratory and compare it to vaccZymeHiBIgG (Binding site-UK).
3. Materials and Methods
3.1. Antigen Preparation
The PRP was prepared from culture supernatants of Hib strains, which were obtained from the type bacteria collection of Pasteur Institute of Iran, Tehran, Iran (PTCC = 1623) grown on culture media, including brain heart infusion broth (BHIB) (Difco, USA) and tripticase soy broth (TSB) (Difco, USA). In order to increase cell density and PRP titer, 60 liter fed batch fermentation was incorporated (Nova-palijas, contact-flow BV, the Netherlands) with 40 L working volume, at 37 ± 1°C (14). The PRP was prepared by precipitation with a mixture of alcohols, including ethanol 70%, methanol 99% and isopropanol 99%, with ratios of 60%, 20% and 20%, respectively. Then, the precipitate was centrifuged for one hour at 4000 rpm. The pellet was washed two times with pyrogen free water. After storing at 4ºC for 24 hours, it was centrifuged for one hour at 4000 rpm. Resuspension of the precipitate was performed in 0.3 M sodium chloride. Orcinol was added to the pellet for assessing ribose (11, 14, 25, 26). The ribose concentration was determined by measuring the absorbance of the solution at 670 nm and comparing it to a standard curve prepared by assaying pure ribose. The PRP concentration was expressed in units of mg PRP per liter (14). After lyophilization, the purity of PRP was determined with nuclear magnetic resonance (NMR) and fourier transform infrared spectroscopy (FTIR).
3.2. Antigen Coatin
An amount of 2 mg PRP antigen was dissolved in 1 mL of distilled water, after which 100 µL of 0.1 M sodium periodate was added to this solution to the emerging aldehyde groups from vicinal hydroxyl groups of sugar moieties of PRP (27). The reaction mixture was stirred at room temperature for 20 minutes. The solution was dialyzed in 0.001 M sodium acetate buffer, with a pH of 4.4 and kept at 4ºC for overnight. A two milliliters solution of 0.5 M bicarbonate containing 5 mg/mL BSA, with a pH of 9.6, was prepared. Dialyzed antigen was mixed with 2 mL of prepared BSA and stirred at room temperature for 2 hours. An amount of 400 µL of 4 mg/mL sodium borohydride was gently added to the antigen solution and stirred for one hour. The antigen solution was dialyzed in carbonate buffer at 4ºC for 24 hours. Micro plate wells (Greiner Bio-One GmbH, Germany) were coated with coating buffer (bicarbonate, pH = 9.6) containing prepared antigen solution (PRP-BSA) for 24 hours at room temperature (11). Then, the plates were washed two times with washing buffer (PBS 0.05% Tween 20, pH = 7.2), which contains no other proteins that might compete with the target antigen for attachment to the plastic solid phase. The plates were blocked with blocking buffer (5% BSA) for 1 hour at room temperature. Plates were emptied and were kept at room temperature overnight, to become dry. The BSA is a neutral substance that inhibits passive adsorption of non-specific antigens, while still allowing immunological binding so that it cannot cause cross-reactivity with sera. Consequently, BSA is a common substance that is used in blocking buffer after coating plates (28). We made used of this characteristic to bind PRP to the solid phase in an effective way.
3.3. Sera Collection
Sera were obtained from 83 non-vaccinated children, aged less than 6 years, referred to three Tehran hospitals (Milad, Baharloo and Imam-Hosein, Tehran, Iran).
3.4. Indirect-Enzyme Linked Immunosorbent Assay Procedure
Indirect ELISA method was used for the detection of antibodies against a specific antigen in a two-step incubation procedure. Polystyrene micro well strips (solid phase) are precoated with recombinant, highly immunoreactive antigens. Patient’s serum, which was diluted in diluents buffer, is added to wells. During the first incubation step, anti-specific antibodies, if present in serum, will be bound to the solid phase precoated antigens. The wells are washed to remove unbound serum proteins and anti-human antibodies conjugated to the enzyme horseradish peroxidase (HRP) is added. During the second incubation step, these HRP-conjugated antibodies will be bound to any antigen-antibody complexes previously formed and the unbound HRP-conjugate is then removed by washing. A solution of tetramethylbenzidin (TMB) HRP substrate/chromophore is then added and incubated for 15 minutes, resulting the development of blue color. The color development is stopped and the absorbance is measured spectrophotometrically, at 450 nm. The color intensity can be measured and it is proportional to the amount of antibody captured in the wells and to the amount of antibody in the sample, respectively. Wells containing samples negative for antibody-antigen complexes remain colorless (29).
3.5. Indirect Enzyme-Linked Immunosorbent Assay Optimization
In order to define the optimal dilution for each of the coating antigen and HRP-conjugated anti-human IgG, a titration checkerboard was performed, while the primary concentrations of the samples were 2 mg/L and 1 mg/L, successively. Moreover, to determine the suitable time and temperature for reaction between antibody and antigen and also between the formed complex and enzymatic conjugate, series of tests at different incubation periods and temperatures were performed. Flat-bottom plates (Greiner Bio-One GmbH, Germany) were coated with 100 µL of PRP-BSA with defined appropriate concentration and incubated for 24 hours at room temperature, to perform coating. Washing of the coated plates was performed three to four times, with washing buffer (PBS 0.05%, Tween 20, pH = 7.2). Then, wells were filled with 150 µL of blocking buffer and incubated for 30 minutes at 37°C, after which washing of the plates was performed again three to four times and 100 µL of dilution buffer were added (washing buffer, including 1% BSA). At this point, the plates were ready to add serum samples, consisting of 10 µL of test sera, including positive and negative control (obtained from commercial kit, vaccZymeHiBIgG, Binding site-UK) were added to wells. Plates were incubated at 37ºC for 30 minutes. After washing the wells and adding of 100 μL of HRP-conjugated IgG, with defined appropriate concentration, each plate was incubated at 37ºC for 30 minutes. The washing procedure was repeated and 100 µL of TMB (HRP substrate/chromophore) were added and plates were allowed to rest at room temperature, in the dark, for 15 minutes. An amount of 50 μL of stopping solution (1M hydrochloric acid) was added to each well and then optical density was measured at 450 nm in an ELISA reader (BioTek, Winoovaki, VT, USA) and data were analyzed. The standard positive control of commercial kit (vaccZymeHiBIgG, Binding site-UK) ranging in anti-PRP antibody concentrations from 0 to 10 mg/ml were assayed simultaneously at 450 nm. A commercial ELISA kit (vaccZymeHiBIgG, Binding site-UK) was used to analyze the same sera and results were compared with results obtained from home-made ELISA kit.
4. Results
4.1. Coating Antigen Synthesis
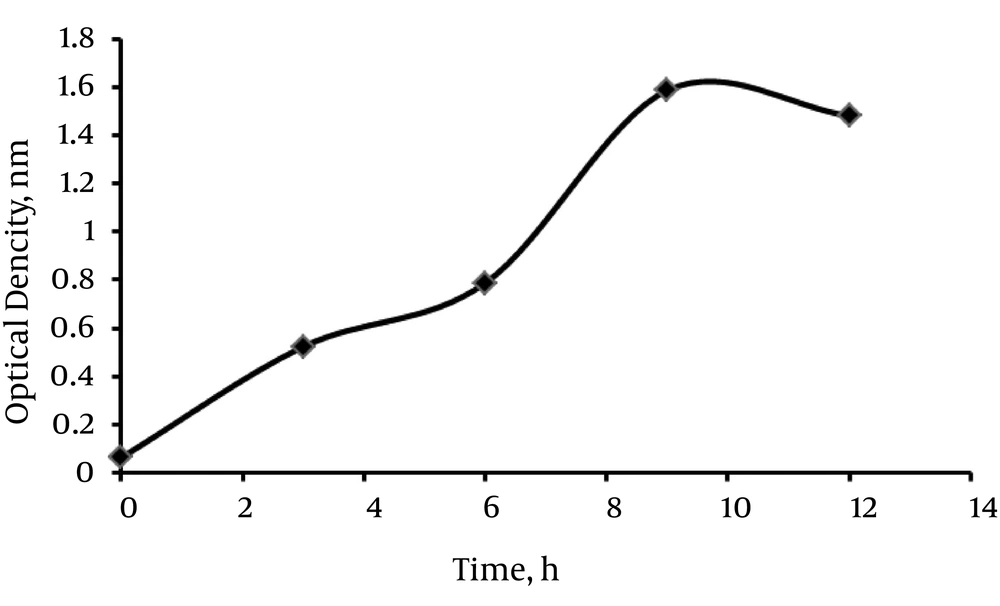
Fed batch fermentation was used to increase cell density and PRP titer. To evaluate the best time for harvesting PRP, we measured the optical densities (OD) of several samples of antigen concentrations, at 450 nm at different times. As the curve shows in Figure 1, the optimal concentration for antigen harvest was reached after 9 hours. The NMR and FTIR analyses were performed on vials of lyophilized PRP, containing 9.8 g powder of PRP. Data indicate > 90% similarity between purified PRP and standard PRP obtained from previous studies (25).
4.2. Determination of Optimal Conditions for Enzyme-Linked Immunosorbent Assay
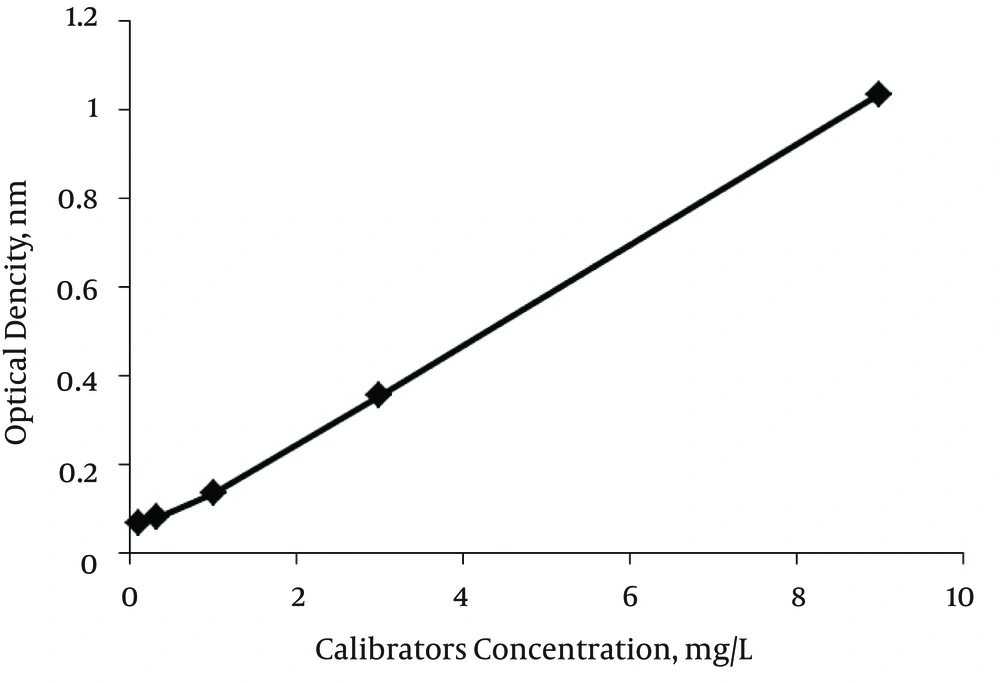
Developing the ELISA kit for anti-PRP antibody detection was accompanied by optimizing the reagents concentrations, incubation temperature and incubation time. In order to obtain higher absorbance for positive control and lower for negative ones, all these factors were experimentally determined by testing positive and negative control. A limitative factor for ELISA reactions is represented by the amount of antigen which can be fixed on the solid phase. To define the optimal working dilution of coating antigen, several dilutions of PRP-BSA [while keeping the concentration of coating antigen constant] from 1/10 to 1/200 were prepared and assayed simultaneously. As indicated in Table 1, the highest OD for positive control and the lowest OD for negative control and blank were at a dilution of 1/50. To define the optimal dilution of HRP-conjugated anti-human IgG, three dilutions were used in the ELISA test. As shown in Table 2, the optimal dilution, in which the ratio between the absorbance of negative control and the positive one has the lowest value, was defined at the dilution of 1/5000. To define the optimal incubation temperature, positive control, negative control and blank were analyzed at 37ºC and room temperature, and the optimal incubation temperature for sera and conjugate was defined at 37ºC. In order to determine the suitable time for reaction of antibody-antigen and also the HRP-enzymatic conjugates, series of tests at two different incubation time periods, 30 minutes and 60 minutes, for the controls, as well as for the conjugate, were performed. In all diagnostic tests, those in which sensitive and correct results are provided in a shorter period of time are more advantageous. The optimal incubation time, at which the ratio between the absorbance of negative control and the positive one has the lowest value, was 30 minutes ELISA showed absorbance at 450 nm as a function of the concentration of anti-PRP antibodies, in standard positive control, as depicted in Figure 2.
| OD at 450 nm | ||||
|---|---|---|---|---|
| PRP dilutions | 1/200 | 1/100 | 1/50 | 1/40 |
| Blank | 0.013 | 0.007 | 0.012 | 0.044 |
| Negative | 0.166 | 0.114 | 0.116 | 0.033 |
| Positive | 0.905 | 1.367 | 2.990 | 2.555 |
Abbreviations: OD, optical density; PRP, polyribosylribitol phosphate.
aOptimal dilution of PRP was at dilution of 1/50.
| OD at 450 nm | ||||
|---|---|---|---|---|
| Dilutions of conjugated antibody | 1/5000 | 1/4000 | 1/2000 | 1/1000 |
| Blank | 0.063 | 0.108 | 0.120 | 0.140 |
| Negative | 0.247 | 0.309 | 0.306 | 0.261 |
| Positive | 1.447 | 0.993 | 1.079 | 1.353 |
Abbreviations: OD, optical density.
aOptimal dilution of conjugate antibody was 1/5000.
After measuring absorbance for all 83 samples, data were analyzed and signals were plotted versus the concentration of antibody. Since ELISA test was performed three times for each serum sample, average data were provided. Serum samples were divided into three age groups: 14.45% < 2 years old, 46.98% were 2 - 4 years old and 38.55% were 4 - 6 years old. Concentrations of anti-PRP IgG antibody were determined using standard calibrators, supplied with the vaccZym kit and were expressed in μg/mL. The lower and upper limits of detection were 0.11 and 9 μg/mL, respectively (27). In age group < 2 years old, about 58% lacked serum antibody against PRP capsule of Hib and about 42% had the minimum acceptable level or a higher antibody level, which has been considered an immunological correlate of natural protection against invasive Hib disease. In age group 2 - 4 years old, about 33% lacked serum antibody against Hib and 67% had the minimum acceptable antibody level or greater, indicating protection against Hib. In age group 4 - 6 years old, 22% lacked serum protective antibody and 78% had the minimum acceptable level or more than minimum acceptable level, indicating protection against Hib infection. Comparison between results obtained from optimized ELISA kit and vaccZymeHiBIgG binding site ELISA kit is presented in Table 3.
| Age, y | Home-Made Kit | vaccZym Kit | ||
|---|---|---|---|---|
| Minimum Immunity 0.11, µg/mL | Without Minimum Immunity < 0.11, µg/mL | Minimum Immunity 0.11, µg/mL | Without Minimum Immunity < 0.11, µg/mL | |
| 0 - 2 | 41.16 | 58.33 | 41.66 | 58.33 |
| 2 - 4 | 66.66 | 33.34 | 64.09 | 35.89 |
| 4 - 6 | 78.12 | 21.87 | 78.12 | 21.88 |
aValues are expressed as percentage.
5. Discussion
The aim of this study was to develop and optimize a home-made ELISA kit for detection of anti-PRP antibody in infected children. All these experiments permitted to develop a home-made indirect-ELISA method kit, for detection of Hib infection. On the basis of the analysis of 83 serum samples, the results obtained by home-made ELISA showed an overall good agreement with those of vaccZyme kit. This means that the obtained data from home-made kit results are in the same range with the commercial kit. We suggest that the agreement of results is due to the high purity of produced PRP, optimization of factors affecting reactions of the test, including antigen-HRP-conjugated anti-human IgG concentrations, incubation time and temperature. Moreover, using BSA was important to make coating with PRP possible. This method of optimizing ELISA has also been shown in previous studies (27-30). Our results are in agreement with those of Mohammadi et al. (23). In their study, a home-made ELISA kit was developed, incorporating soluble antigens extracted from Iranian H. pylori strains and evaluated against commercially used imported ELISA kits. Cost effectiveness and efficiency of the home-made kit was emphasized (23). Moreover, in a study conducted by Jalallou et al. (24). The sensitivity and specificity of home-made ELISA for the detection of anti-Toxoplasma-specific IgM and diagnosis of acute infection, compared to a standard commercial ELISA, were 80% and 90%, respectively. Liu et al. study in China, on the comparison of homemade and imported HbsAg ELISA kits for screening blood samples, confirms that the imported ELISA kit had the highest sensitivity, although its specificity was not as good as that of the home-made ELISA kit (31). Based on the results of this study, the proportion of natural antibody was increased by age. The exposure rate of children with Hib is high, particularly in children younger than 2 years old. This has also been observed in a study conducted by Jahromi and Rahmanian (22), revealing the need to introduce Hib conjugate vaccine in national immunization programs; however, they used a commercial ELISA kit that is not cost-effective, particularly for more extensive epidemiological studies. We recommend further studies, incorporating home-made ELISA kits to detect anti-PRP antibodies, using a larger population of children, to emphasize more the need of immunization.