1. Background
Hepatitis E Virus (HEV) is the major cause of acute viral hepatitis in developing countries. Hepatitis E Virus virions are small, spherical, non-enveloped, positive-sense single-stranded RNA viruses, which belong to the genus hepevirus of the hepeviridae family (1). The virus transmits predominantly through the fecal-oral route, which is profoundly responsible for epidemics in tropical and subtropical countries (2). During outbreaks, the disease attack rate is more prevalent among young adults. Meanwhile, hepatitis E has been recently considered as a reemerging disease due to increased frequency of HEV seropositivity in industrialized countries (3, 4). Hepatitis E usually causes self-limiting disease with low mortality; however, acute liver failure, chronic infection, or extrahepatic symptoms are possible. The mechanisms of pathogenesis appear to be substantially immune-mediated. Although the infection is rarely progressed into its severe form, this can occur during pregnancy and in immunosuppressive conditions such as solid organ transplantation (4-6). Although chronic hepatitis E infection is not common, it is considered as a major clinical problem in immune compromised (7) and older patients (8). Persistent HEV infections in immune compromised patients, suggest that HEV is controlled by adaptive immune responses. There are scarce studies that have investigated HEV-specific adaptive responses. Like other viral infections, both humoral and cellular immune responses, take part in the pathogenesis of HEV infection. It has been demonstrated that both IgM and IgG antibodies appear at disease onset such as jaundice and persist for variable periods. In spite of their neutralizing activities and being efficient for a long time, the exact role of humoral responses in protection against HEV reinfection remains unclear. Although hepatitis E is associated with a robust antibody response and this is the basis of HEV vaccine design strategy, cellular immune responses to HEV proteins develop in HEV-infected patients (9, 10). Regarding acute hepatitis E, there are a few studies that have shown HEV-specific lymphoproliferative responses of T cells (11) and higher production of interferon gamma by peripheral blood mononuclear cells (PBMCs), stimulated by recombinant HEV Open Reading Frame 2 (ORF2) and ORF3 proteins (10, 12). However, a few studies investigated T-cell immunity in HEV infection yet this issue is open and needs more consideration. It has been shown that polarized human T helper type 1 (Th1) and T helper type 2 (Th2) cells not only produce different sets of cytokines, but also exhibit preferential expression of some activation markers (13-15). The CD30 is a member of the tumor necrosis factor (TNF) family, and appears to be preferentially expressed and released by human CD4+ and CD8+ T cells, producing predominantly Th2 cytokines (16). The CD26 is a cell surface glycoprotein with Dipeptidyl Peptidase IV (DPP IV) enzyme activity in the extracellular domain, correlating with the production of Th1-like cytokines in several diseases (17-19). After cellular activation, the soluble forms of CD30 (sCD30) and CD26 (sCD26) are released into the bloodstream. Therefore, analysis of CD26 and CD30 and their soluble forms has been proposed to be useful in discriminating Th1 and Th2 responses (19-21). However, there are scarce studies thus far to address Th1 and Th2- type responses in hepatitis E infection (22). Therefore, we here aimed to study Th1 and Th2 cytokine patterns in HEV-infected individuals.
2. Objectives
This study aimed to evaluate the Th1/Th2 ratio by investigating serum soluble markers from Th1 and Th2 cells in acute HEV infected patients.
3. Patients and Methods
3.1. Study Population
Seventy individuals were selected for this study. This population included 35 anti-HEV IgM positive patients and 35 age and gender matched anti-HEV negative controls. An informed consent was obtained from all participants. The current study was performed in accordance with ethical guidelines of the 1975 Declaration of Helsinki. In addition to the presence of IgM anti-HEV antibodies, patients were checked for clinical signs of hepatitis such as icterus, dark-colored urine, elevated alanine aminotransferase (ALT) and bilirubin levels in the serum, bile salts and pigments in the urine. The control group had the same epidemiological conditions as the patients, yet had negative anti-HEV IgM test results and no history of an illness resembling viral hepatitis, indicating their previous exposure to HEV. Individuals with infectious disease, pregnancy, chronic renal and liver diseases and those receiving blood or blood products within the last three months were excluded. In addition, all individuals were negative for hepatitis B surface antigen (Dia-Sorin, Saluggia, Italy), IgM anti-hepatitis A virus antibody (Hepanostika HAV IgM; Organon Teknika, Boxtel, the Netherlands), and anti-HCV antibody (Ortho-Clinical Diagnostics, Raritan, NJ, USA). The study protocol was approved by the medical ethics committee of Mazandaran University of Medical Sciences and conformed to the ethical guidelines of the declaration of Helsinki. Informed consent was obtained from each patient or their relatives.
3.2. Serological Enzyme Immunoassays
From eligible subjects, a 5-mL blood sample was drawn and serum was obtained by centrifugation at 1450 g for 15 minutes at 4ºC and stored at -80ºC until further processing. Specific anti-HEV IgM antibody (anti-HEV) to immunodominant determinants derived from ORF2 and ORF3 of all the four HEV subtypes was measured using an enzyme immunoassay (DiaPro, Milan, Italy), according to the manufacturer’s instructions. Serum levels of CD26, CD30, IFN-γ and IL-4 were detected using commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kits with paired cytokine-specific monoclonal antibodies, according to the manufacturer’s recommended procedure (Bender Med Systems, Vienna, Austria). The detection limit for CD26 and CD30 were 7.26 pg/mL and 1.9 U/mL, respectively.
3.3. Statistical Analysis
Significance analysis was performed for comparison of differences between HEV positive and non HEV-positive subjects using Mann-Whitney U test, independent t-test and Yate’s correction of contingency where appropriate. Relationships between clinical variables and levels of soluble markers were evaluated by determining the Pearson correlation coefficient. A P < 0.05 was considered to be significant. Linear regression analyses were carried out to determine a possible correlation between values of sCD30 and sCD26 and several biological markers such as IFN-γ and IL-4.
4. Results
The basic characteristics of the study population are shown in Table 1. There was no significant differences between patients with acute HEV infection and HEV negative controls with respect to age and gender (P = 0.119 and P = 0.6, respectively). All individuals had an age range of 11 to 25 years old and 48.6% of acute HEV infected patients and 40% of HEV negative subjects were males. In addition, there was no significant difference between the two groups in terms of location (urban or rural). As shown in Table 1, most of the patients exhibited higher sCD26 serum levels (104 - 536 pg/mL) than the controls (16 - 324 pg/mL), indicating significant differences (P = 0.001). The median serum levels of CD30 were 48 and 40.9 U/mL in patients with HEV infection and HEV negative subjects, respectively, which shows no significant differences (P = 0.159).
| Acute HEV Patients | Controls | P Value | |
|---|---|---|---|
| Age | 19.7 ± 3.3 | 18.4 ± 3.2 | 0.119 |
| Gender | |||
| Male | 17 | 14 | 0.6 |
| Female | 18 | 21 | 0.6 |
| Location | |||
| Urban area | 14 (40) | 16 (45.7) | 0.8 |
| Rural area | 21 (60) | 19 (54.3) |
aN = 35
bValues are expressed as mean
To have a clear picture of Th1- and Th2-type responses in acute HEV infection, serum levels of IFN-γ and IL-4, with respect to Th1 and Th2 responses, were compared between the two groups. As shown in Table 1, IFN-γ production was higher in acute HEV patients than HEV negative controls (median; 23.7 vs. 7.2 pg/mL, P < 0.0001). Acute HEV patients also produced more serum IL-4 than the HEV negative controls yet these differences did not meet statistical significance (median; 7.1 vs. 5.5 U/mL, P = 0.354). The sCD26/sCD30 ratio was calculated for each individual, and the medians of ratios were then compared between acute HEV infection patients and HEV negative groups. The sCD26/sCD30 ratio was higher in acute HEV patients (5.17) than that of HEV negative subjects (2.22) and the difference was significant (P = 0.001). On the other hand, the ratio of sCD26/sCD30 in the acute HEV group was more than two folds higher than the HEV negative group. Levels of sCD26 and sCD30 did not show any significant correlation with age, gender and location of each group.
4.1. Effect of Acute Hepatitis E Virus Infection on the sCD26 and sCD30
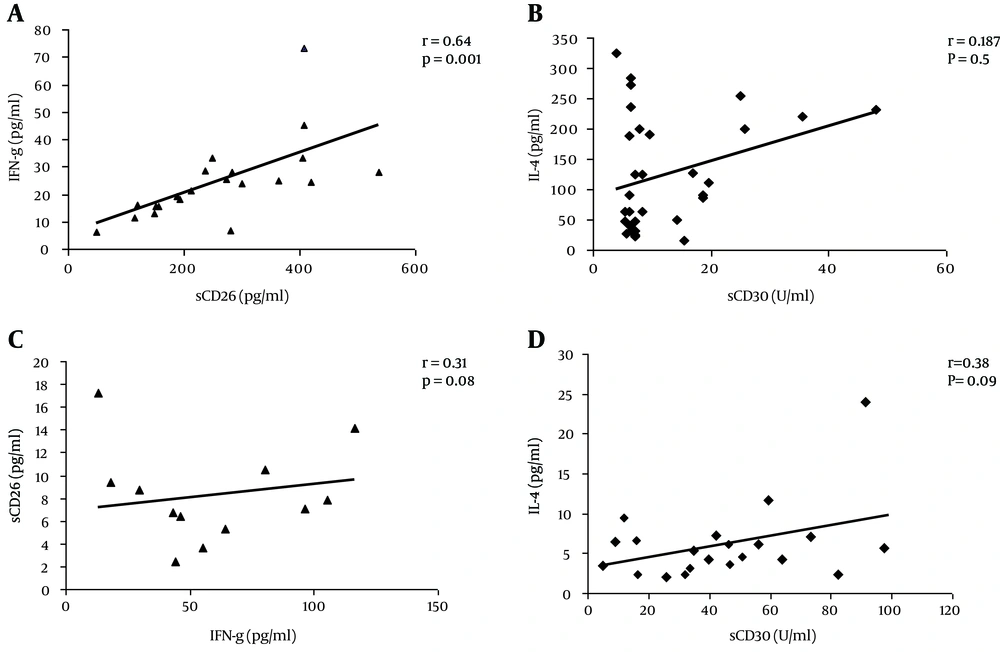
To examine the impact of acute HEV infection on development of Th1 or Th2 response, correlation of serum levels of appropriate cytokines (IFN-γ and IL-4) with expression of CD26 or CD30 were assessed. As shown in Figure 1A, a positive correlation was found only between the serum levels of sCD26 and IFN-γ in acute HEV patients (r = 0.64, P = 0.001). Nevertheless, the levels of sCD30 did not have any significant correlation with serum levels of IL-4 (r = 0.25, P = 0.37) in this group (Figure 1C). There was no significant correlation between sCD26 or sCD30 and the levels of IFN-γ or IL-4 in HEV negative subjects (Figure 1A and D).
A, A direct positive correlation was seen between serum levels of IFN-γ and sCD26 only in acute HEV infected subjects; C and D, The correlation between sCD30 and IL-4 in acute HEV patients or HEV negative subjects, respectively. A, R = 0.64, P = 0.001; B, R = 0.31, P = 0.08; C, R = 0.187, P = 0.5; D, R = 0.38, P =0.09.
| Anti-HEV Positive (n=35) | Anti-HEV Negative (n=35) | P Value | |
|---|---|---|---|
| IFN-γ (pg/mL) | 23.7 (6.3 - 73) | 7.2 (4.3 - 48) | < 0.0001 |
| IL-4 (pg/mL) | 7.1 (2.3 - 17.2) | 5.5 (2.1 - 24) | 0.354 |
| sCD26 (pg/mL) | 248 (104 - 536) | 91 (16 - 324) | 0.001 |
| sCD30 (U/mL) | 48.0 (12.8 - 116.25) | 40.9 (5 - 99) | 0.159 |
| sCD26/sCD30 | 5.17 (1.6 - 30) | 2.22 (0.28 - 10.8) | 0.001 |
aValues are expressed as median (range).
bAll statistical comparisons were determined by the Mann-Whitney U test.
5. Discussion
To the best of our knowledge, the present work is the first study that evaluated the correlation between serum levels of sCD26 and sCD30 in which corresponding to Th1 and Th2-type immune responses on acute HEV infected patients in a HEV endemic area (23). The main findings of this study were increased serum levels of IFN-γ and sCD26 in acute HEV patients but not in HEV negative controls. Furthermore, T cell activation, occurring in acute HEV infected patients, was compared to HEV seronegative controls and demonstrated an increase in IFN-γ production in acute HEV patients. T cell-mediated immune responses play a critical role in eliminating viral infections (24). Meanwhile, T-effectors responses may also determine the outcome of infection by mediating injury in virus-infected cells. Interferon-γ, a key cytokine of Th1-type immune response, plays an important role in protective immune response against viral infections (25-28). It increases antigen presentation by antigen-presenting cells, in order to enhance MHC class I and co-stimulatory molecule expression on the cells. Gamma interferon enhances the antiviral state by increasing cytotoxic activity of NK, NKT, and CD8+ T cells on virus-infected cells (17). Moreover, it inhibits the development of Th2 type immune response by decreasing the production of Interleukin (IL)-4. Although Th1-type responses have been demonstrated to be correlated with hepatic inflammatory activity of viral hepatitis, via favoring the clearance of virus, Th2 cells may be associated with the persistence of those infection (29). Increased levels of IFN-γ in acute hepatitis E infected patients compared with the controls might contribute to the successful clearance of the virus. In addition, no significant overproduction of IL-4 in acute HEV infected patients suggested the Th1/Th2 balance trend toward Th1 dominant immune response yet with a coincidental weak Th2 response in acute hepatitis E infection. These findings are in line with data obtained from other studies, which have shown a strong HEV-specific CD4+ and CD8+ T cell responses in the acute form of hepatitis E (12, 30) and B infections (31). The elevated levels of CD8+ T cells were observed in acute viral hepatitis-E in the study of TrehanPati et al. They also found an increase in CD38+CD69+ T cells, mRNA expression of IFN-γ, Tumor Necrosis Factor (TNF)-α, IL-4 and down regulation of IL-2 and IL-10 in CD4+ T cells in acute hepatitis E infection (32). Analysis of cytokine production or intracellular cytokine expression is routinely used to classify the immune response into Th1 and Th2 responses. It was demonstrated that Th1/Th2 responses are associated with hepatitis E infection and progression of fulminant hepatic failure (FHF) by detecting significant high levels of IFN-γ, IL-2, TNF-α, and IL-10 cytokines (33). However, these are labor-intensive and time-consuming methods. Recently, analysis of CD26 and CD30, and their serum soluble forms have been proposed to be simple, useful, and efficient tools in discriminating Th1 and Th2 responses. These molecules are T cell activation markers associated with Th1- and Th2-related conditions, respectively (13, 15, 34, 35). Analysis of serum sCD26 and sCD30 in acute HEV infection showed significantly higher levels of sCD26 in acute HEV patients compared with HEV seronegative controls. Moreover, the presence of a direct close correlation between serum levels of sCD26 and IFN-γ in acute HEV patients suggests that sCD26 might be a surrogate indicator of Th1 immune response or a marker of T cell activation in acute HEV infection. Although CD8+ T cells and NK cells could produce IFN-γ, Th1 cells are more common sources of IFN-γ production. Therefore, it would be logical to propose that higher production of IFN-γ in acute HEV infected patients is reasonable for manifesting Th1 cell activation due to increased expression of CD26 molecules. Concordantly, this speculation was reported by Prabhu et al. they showed predominant enumeration of CD4+T cell in the liver of HEV infected patients (36). A significant increase in trend of HEV specific IFN-γ producing T-cell response in Elispot of acute hepatitis E patients, which diminished after successful clearance of HEV infection (22). In line with our case, other researchers highlighted specific T cell reactivity in acute hepatitis E infection, indicated by increased frequency of recombinant ORF2 protein specific IFN-γ- secreting cells (12). Therefore, the elevated levels of IFN-γ, in acute HEV infection suggest that Th1-mediated effects may contribute not only to liver cell injury, but probably also play a major role in successful control of infection. In addition, higher levels of CD26 have been shown after successful treatment of chronic hepatitis B (37) and in remission of vasculitis (38). Interleukin (IL)-4 is one of the key cytokines in Th2 response. It is a pleiotropic cytokine, which also shows prominent anti-inflammatory effects on Th1-driven inflammation by modulating macrophage activation and antagonizing the effects of IFN-γ. Therefore, IL-4 has as an important role, acts like a suppressor of the cell-mediated response and increases the level of humoral immunity. The present study showed that little increase in IL-4 levels in acute HEV patients indicates the activity of the Th2 phenotype in acute HEV infection, which is decisive for the production of neutralizing antiviral antibodies, resolution of infection, and inhibition of the overwhelming Th1-type inflammatory response. On the other hand, a shift to the Th1 cytokine environment favors the clearance of HEV infection in acute infected patients. There are concordant literature data showing very low levels of IL- 4 in the sera of patients with acute HBV infection (31). However, high expression/release of CD30 in acute HEV patients compared with healthy subjects possibly reflects the activation of CD30 harboring CD4+ or CD8+ T cells during disease. Meanwhile, no significant correlation existed between the levels of sCD30 and IL-4 in acute HEV patients suggesting that CD4+ Th1 but not CD8+ T cells might be the main sources of sCD30-producing cells. Taken together, the key finding of this study was a direct significant positive correlation between the serum levels of sCD26 and IFN-γ in acute HEV infected patients, which confirms that the trend of sCD26 levels is a valuable factor for predicting disease progression.