1. Background
Bacteria of the genus Proteus are Gram-negative rods belonging to the family Enterobacteriaceae (1). Proteus spp. currently include four named species: Proteus mirabilis, P. vulgaris, P. hauseri, and P. penneri, all of which are pathogenic for humans (1). A characteristic feature of Proteus spp. rods is the presence of cilia, which increases their virulence, the phenomenon of swarming growth, and the ability to produce urease (2). Proteus spp. bacteria mainly are found in water, soil, and natural fertilizers, as well as in food products. These microorganisms also may be present in humans and animals, mainly in the gastrointestinal tract (2).
Urinary tract infections (UTI) are one of the most common infectious diseases, especially in a hospital environment (3). The participation of P. mirabilis bacteria in the etiology of UTI is 7%. One of the consequences of UTI caused by Proteus spp. may be the formation of kidney stones (2). The microorganisms of the genus Proteus also can cause skin and subcutaneous tissue infections as well as infections arising in the case of hernia plastic surgery or acute otitis media, and the microorganisms of the genus P. mirabilis can cause purulent meningitis in newborns (2, 4, 5).
Proteus mirabilis and P. vulgaris rods have a number of virulence factors, for example, lipopolysaccharide (LPS), flagella, fimbriae, glycocalyx, and the phenomenon of adhesion and hydrophobicity of bacteria surface (2). An important adaptation of the bacteria Proteus spp., to cause infections in the urinary system, is the ability to form a biofilm. A biofilm is a formation of communicating microorganisms that adhere to certain surfaces and to neighboring cells and are covered with an extracellural matrix (6). It is often formed on catheters and surgical implants (6). The structure of a biofilm provides protection for bacterial cells against adverse environmental factors, including trade disinfectants and antibiotics (7).
2. Objectives
The aim of this study was to assess the degree of a biofilm formation by strains of P. mirabilis and P. vulgaris and to determine the effect of ciprofloxacin on planktonic cells and on biofilm of studied microorganisms.
3. Materials and Methods
3.1. Characteristics of the Strains
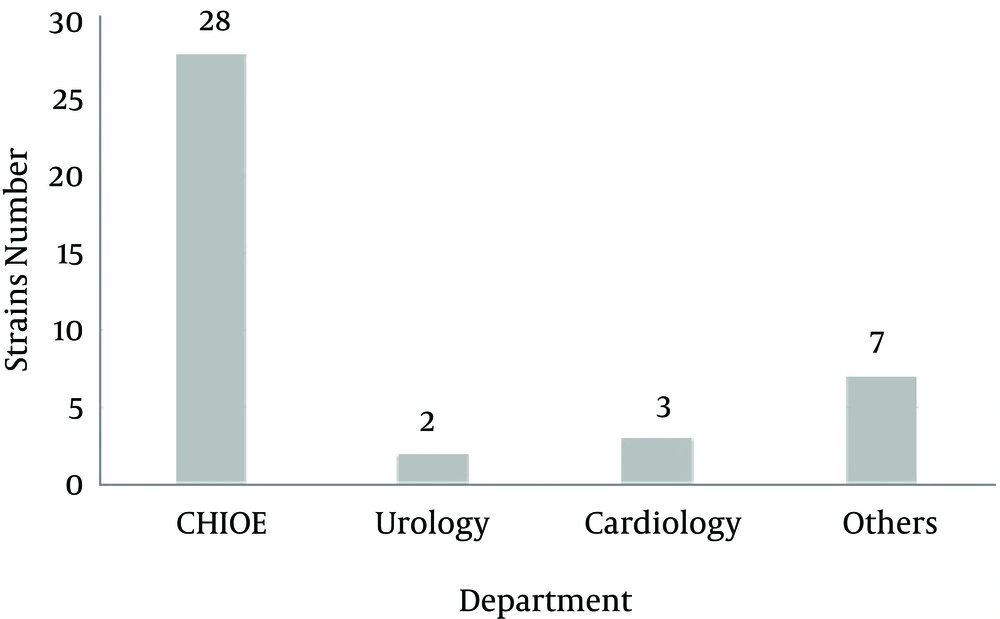
The material for this study consisted of 20 strains of P. mirabilis and 20 strains of P. vulgaris from the collection of the department of microbiology of the Ludwik Rydygier medical college in Bydgoszcz, Poland, Nicolaus Copernicus University. The strains were isolated from patients treated in the clinics of the Antoni Jurasz university hospital in Bydgoszcz in 2010 - 2014. Most strains (28; 70.0%) were isolated from patients of the department of general surgery and endocrinology (Figure 1). In the case of P. mirabilis and P. vulgaris, 25.0% (n = 5) were from urine and 75.0% (n = 15) from a wound swab.
3.2. Susceptibility of the P. mirabilis and P. vulgaris Strains
The strains were examined by the disk diffusion method to piperacillin, amoxicillin with clavulonic acid, piperacillin with tazobactam, cefotaxime, ceftazidime, cefepime, imipenem, amikacin, ciprofloxacin, norfloxacin, and trimethoprim-sulfamethoxazole (Emapol). The results were interpreted according to the european committee on antimicrobial susceptibility testing (EUCAST) recommendation (8).
3.3. Evaluation of Ciprofloxacin MIC Values of Planktonic Cells
In order to determine the minimal inhibition concentration (MIC) of the tested bacteria, the microdilution method in a microtiter plate was used in accordance with the recommendations of the clinical and laboratory standards institute (CLSI) (9). The growth of bacteria in the presence of ciprofloxacin at concentrations ranging from 0.01 - 128.0 μg/mL was evaluated in the studies. Pseudomonas aeruginosa strain ATCC 27853 was used for the control assays.
The MIC value was read visually by the presence of turbidity of the suspension in the wells of microtiter plates at a given concentration of antibiotic. The results were interpreted on the basis of recommendations introduced by the EUCAST (8). The limit for the minimum inhibitory concentration of ciprofloxacin is ≤ 0.5 μg/mL for susceptible strains. For intermediately susceptible microorganisms, MIC breakpoints fall within the range of 0.5 - 1.0 μg/mL. In the case of strains that have resistance to ciprofloxacin, the limit of MIC is > 1 μg/mL. MIC50 and MIC90 values were calculated on the basis of the obtained values of MIC. To be more precise, the values include ciprofloxacin concentration, at which the growth of 50% and 90% of strains was inhibited.
3.4. Evaluation of biofilm Formation
A biofilm formation by the strains of P. mirabilis and P. vulgaris was evaluated based on the method described previously (10) using a 0.1% TTC (2, 3, 5-Triphenyl-tetrazolium chloride solution, AVANTORTM) with modifications. After a 24-hour incubation, the planktonic, nonadsorbed cells were removed from the wells of the microtiter plates. The wells were washed three times with 600 μL sterile distilled water per cell. 100 μL of tryptic soy medium (TSB, Becton Dickinson) and 100 μL of sterile 0.1% TTC solution were added to each cell and incubated. After two hours, the contents of the wells were removed and washed again with distilled water. The formazan was dissolved in 200 μL methanol (AVANTORTM). The contents of the wells were transferred to sterile microtiter plates. An absorbance measurement was made using a BIO-TEK spectrophotometer at a wavelength of 470 nm. The KC4TM v.3.4 and KC4TM Signature program was used to read the data.
The results were interpreted in accordance with the criteria described in the previous publication (10). The obtained results were compared by the Mann-Whitney U test, with differences at p ≤ 0.05 considered as statistically significant using the StatSoft Inc. (2011) STATISTICA 10.0 program (data analysis software system).
3.5. Assessment of the Ciprofloxacin Influence on P. mirabilis and P. vulgaris Biofilms
The study evaluated the influence of ciprofloxacin on the 24-hour biofilm of P. mirabilis and P. vulgaris by the microdilution method in microtiter plates in accordance with the methodology described in the work of Kwiecinska-Pirog et al. (10). As part of the experiment, the minimum biofilm eradication (MBE) values of MBE50 and MBE90 were rated. Those values denote the concentration of antibiotic that leads to an inhibition of biofilm formation by P. vulgaris and P. mirabilis, respectively, ≥ 50.0% and ≥ 90.0% of the tested strains.
The reduction of the biofilm was determined by a calculation that uses the absorbance values of the tested strains. These values have been registered with a spectrophotometer. The absorbance values of the positive control sample and the average value of the results of the absorbance of the tested strains were used for calculation. The degree of reduction was calculated using the following Equation:
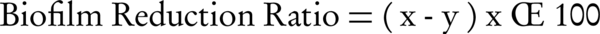
x, absorbance value of positive control; y, absorbance value of the examined strain.
4. Results
4.1. Susceptibility of the P. mirabilis and P. vulgaris Strains
All examined strains were susceptible to piperacillin with tazobactam, cephalosporines, imipenem, ciprofloxacin, and norfloxacin. One P. vulgaris strain (5.0%) was resistant to piperacillin. Five Proteus spp. strains (12.5%) were resistant to amoxicillin with clavulonic acid (4, 20.0% of P. mirabilis, and 1, 5.0% of P. vulgaris). One strain of P. mirabilis (5.0%) was resistant to amikacin. Five strains of P. mirabilis (25.0%) and one of P. vulgaris (5.0%) were resistant to trimethoprim-sulfamethoxazole. The MIC90 value of ciprofloxacin in the case of strains of P. mirabilis and P. vulgaris isolated from a wound swab was 0.5 μg/mL. The value of the isolates from the urine was equal to 1.0 μg/mL and 0.25μg/mL, respectively, for P. mirabilis and P. vulgaris (Table 2).
4.2. The assessment of the Impact of Ciprofloxacin on Planktonic Cells
All tested strains of P. mirabilis and P. vulgaris were susceptible to ciprofloxacin. However, among the 20 strains of P. mirabilis three (15.0%) were classified as the intermediate group. Ciprofloxacin-intermediate strains were isolated from urine. The MIC values of ciprofloxacin for planktonic bacteria cells of both tested species are presented in Table 1. The MIC50 ciprofloxacin values for planktonic cells of P. mirabilis and P. vulgaris strains were 0.06 μg/mL for both isolates from the wound swabs and those from urine (Table 2).
| Proteus mirabilis | Proteus vulgaris | |||
|---|---|---|---|---|
| MIC, µg/mL | Strains | Cumulated Value | Strains | Cumulated Value |
| 0.01 | 2 (10.0) | 2 (10.0) | 2 (10.0) | 2 (10.0) |
| 0.03 | 5 (25.0) | 7 (35.0) | 5 (25.0) | 7 (35.0) |
| 0.06 | 5 (25.0) | 12 (60.0) | 6 (30.0) | 13 (65.0) |
| 0.12 | 2 (10.0) | 14 (70.0) | 1 (5.0) | 14 (70.0) |
| 0.25 | 1 (5.0) | 15 (75.0) | 3 (15.0) | 17 (85.0) |
| 0.50 | 2 (10.0) | 17 (85.0) | 3 (15.0) | 20 (100.0) |
| 1.00 | 3 (15.0) | 20 (100) | 0 (0.0) | 20 (100.0) |
aValues are expressed as No. (%) unless otherwise indicated.
| Parameter | Proteus mirabilis | Proteus vulgaris | ||||
|---|---|---|---|---|---|---|
| Wound swab | Urine | All Strains | Wound swab | Urine | All Strains | |
| MIC50 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 |
| MIC90 | 0.5 | 1.0 | 1.0 | 0.5 | 0.25 | 0.5 |
| MBE50 | 1.0 | 4.0 | 1.0 | 1.0 | 0.25 | 1.0 |
| MBE90 | 256.0 | 512.0 | 512.0 | 4.0 | 2.0 | 64.0 |
aValues are expressed as µg/mL.
4.3. Evaluation of Biofilm Formation
All tested strains formed a biofilm. The absorbance value measured for the strains of P. mirabilis was in the range of 0.656 - 2.319, while P. vulgaris was within the range of 0.708 - 1.800. According to the criteria (10), none of the strains formed a biofilm in a weak degree. In the case of two tested species, most strains formed a biofilm of medium intensity 12 (60.0%) isolates of P. mirabilis and 11 (55.0%) of P. vulgaris. Strong biofilm formation was observed for eight (40.0%) strains of P. mirabilis and nine strains (45.0%) of P. vulgaris (Table 3).
| Biofilm Formation | Proteus mirabilis | Proteus vulgaris | Total | ||
|---|---|---|---|---|---|
| Wound swab | Urine | Wound swab | Urine | ||
| Lack | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Weak | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Moderate | 10 (50.0) | 2 (10.0) | 7 (35.0) | 4 (20.0) | 23 (57.5) |
| Strong | 5 (25.0) | 3 (15.0) | 8 (40.0) | 1 (5.0) | 17 (42.5) |
| Total | 15 (37.5) | 5 (12.5) | 15 (37.5) | 5 (12.5) | 40 (100.0) |
aValues are expressed as No. (%).
4.4. Assessment of the Impact of Ciprofloxacin on P. mirabilis and P. vulgaris Biofilms
The MBE50 value of ciprofloxacin was 1.0 μg/mL for the strains of both examined species (Table 4). This value was 16 times higher than the MIC50 for planktonic cells. The MBE90 value of ciprofloxacin for isolates of P. vulgaris was 64 μg/mlL, and 512 μg/mL for P. mirabilis strains (Table 2). The MBE90 value was 512 and 128 times higher than MIC90, respectively, for P. mirabilis and for P. vulgaris. MBE90 and MBE50 values of ciprofloxacin, depending on clinical material as well as the tested species, are presented in Table 2. No statistically significant difference was observed in resistance of the biofilm formed by P. mirabilis and P. vulgaris strains (P = 0.2315) and by strains isolated from urine and from wound swabs (P = 0.6335).
| MBE, µg/mL | Proteus mirabilis | Proteus vulgaris | ||
|---|---|---|---|---|
| Strains | Cumulated Value | Strains | Cumulated Value | |
| 0.06 | 1 (5.0) | 1 (5.0) | 2 (10.0) | 2 (10.0) |
| 0.12 | 1 (5.0) | 2 (10.0) | 1 (5.0) | 3 (15.0) |
| 0.25 | 1 (5.0) | 3 (15.0) | 4 (20.0) | 7 (35.0) |
| 0.5 | 4 (20.0) | 7 (35.0) | 2 (10) | 9 (45.0) |
| 1.0 | 3 (15.0) | 10 (50.0) | 4 (20.0) | 13 (65.0) |
| 2.0 | 0 (0.0) | 10 (50.0) | 0 (0.0) | 13 (65.0) |
| 4.0 | 3 (15.0) | 13 (65.0) | 3 (15.0) | 16 (65.0) |
| 8.0 | 2 (10.0) | 15 (75.0) | 0 (0.0) | 16 (65.0) |
| 16.0 | 0 (0.0) | 15 (75.0) | 0 (0.0) | 16 (65.0) |
| 32.0 | 0 (0.0) | 15 (75.0) | 1 (5.0) | 17 (70.0) |
| 64.0 | 0 (0.0) | 15 (75.0) | 2 (10.0) | 19 (95.0) |
| 125.0 | 1 (5.0) | 16 (80.0) | 0 (0.0) | 19 (95.0) |
| 256.0 | 1 (5.0) | 17 (85.0) | 0 (0.0) | 19 (95.0) |
| 512.0 | 2 (10.0) | 19 (95.0) | 0 (0.0) | 19 (95.0) |
| > 512.0 | 1 (5.0) | 20 (100.0) | 1 (5.0) | 20 (100.0) |
aValues are expressed as No. (%).
Based on the results of spectrophotometric study, the degree of the reduction of the biofilm of both tested strains was determined (Tables 5 - 6). Only one out of five (20.0%) P. mirabilis strains isolated from urine had a biofilm reduction ratio of over 50% at the lowest tested concentration of ciprofloxacin (0.06 µg/mL). Among the strains of the species isolated from the wound swabs, a degree of reduction above 50% for the concentration of 0.06 µg/mL of the tested antibiotic was observed in the case of eight strains (53.3%). For strains of P. vulgaris, there was no difference in the degree of reduction of the biofilm at the lowest tested concentrations of ciprofloxacin, depending on the isolation of the strain. The concentration of 0.06 µg/mL resulted in a reduction of more than 50% of the P. vulgaris biofilm among as many as 60% of the tested strains.
| Ciprofloxacin Concentration, µg/mL | Proteus mirabilis | Proteus vulgaris | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 1 | 2 | 3 | 4 | 5 | |
| 512.0 | 93 | 95 | 95 | 95 | 90 | 95 | 94 | 96 | 93 | 88 |
| 256.0 | 95 | 93 | 93 | 93 | > 50 | 95 | 93 | 95 | 95 | 91 |
| 128.0 | 92 | 91 | 91 | 92 | > 50 | 96 | 94 | 95 | 89 | 92 |
| 64.0 | 92 | 93 | 93 | 95 | > 50 | 95 | 88 | 96 | 94 | 91 |
| 32.0 | 94 | 93 | 93 | 94 | > 50 | 95 | 87 | 95 | 91 | 91 |
| 16.0 | 94 | 92 | 92 | 94 | > 50 | 95 | 93 | 95 | > 50 | 91 |
| 8.0 | 95 | 90 | 90 | 94 | > 50 | 95 | 94 | 95 | > 50 | 90 |
| 4.0 | 94 | 93 | 93 | 93 | < 50 | 95 | 76 | 96 | > 50 | 91 |
| 2.0 | 95 | > 50 | > 50 | > 50 | < 50 | 95 | 80 | 96 | > 50 | 86 |
| 1.0 | 89 | < 50 | < 50 | < 50 | < 50 | 69 | 87 | 95 | > 50 | 93 |
| 0.5 | 94 | < 50 | < 50 | < 50 | < 50 | 94 | 95 | 95 | > 50 | 91 |
| 0.25 | 95 | < 50 | < 50 | < 50 | < 50 | 95 | 92 | 69 | < 50 | 58 |
| 0.125 | > 50 | < 50 | < 50 | < 50 | < 50 | > 50 | > 50 | 90 | < 50 | 83 |
| 0.06 | > 50 | < 50 | < 50 | < 50 | < 50 | < 50 | > 50 | 92 | < 50 | 64 |
| Ciprofloxacin Concentration, µg/mL | Biofilm Reduction of Strains, % | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | |
| Proteus mirabilis | |||||||||||||||
| 512.0 | 95 | 91 | 92 | 94 | 96 | 90 | 94 | 95 | 90 | 96 | 94 | 84 | > 50 | 86 | 88 |
| 256.0 | 93 | 92 | 93 | 95 | 92 | 93 | 87 | 94 | > 50 | 94 | 86 | 87 | > 50 | 92 | 89 |
| 128.0 | 94 | 94 | 93 | 92 | 92 | 92 | > 50 | 95 | > 50 | 88 | 90 | 91 | > 50 | 90 | 86 |
| 64.0 | 94 | 95 | 94 | 90 | 89 | 92 | > 50 | 94 | > 50 | 86 | 89 | 92 | > 50 | > 50 | 84 |
| 32.0 | 95 | 95 | 92 | 91 | 94 | 91 | > 50 | 94 | > 50 | 90 | 82 | 91 | > 50 | 94 | 85 |
| 16.0 | 95 | 90 | 87 | 91 | 94 | 89 | > 50 | 95 | > 50 | 92 | 92 | 92 | > 50 | 91 | 84 |
| 8.0 | 95 | 95 | 89 | 91 | 90 | 85 | > 50 | 96 | > 50 | 91 | 92 | 92 | > 50 | 90 | 85 |
| 4.0 | 95 | 95 | 91 | 86 | 92 | 88 | > 50 | 94 | <50 | 91 | > 50 | 91 | > 50 | > 50 | > 50 |
| 2.0 | 95 | 94 | 91 | 88 | 94 | 91 | > 50 | 96 | <50 | 92 | > 50 | 91 | > 50 | > 50 | > 50 |
| 1.0 | 95 | 94 | 90 | 89 | 93 | 87 | > 50 | 95 | <50 | 89 | < 50 | 86 | > 50 | > 50 | > 50 |
| 0.5 | 93 | 88 | 91 | > 50 | > 50 | > 50 | > 50 | 88 | > 50 | 86 | < 50 | 87 | > 50 | > 50 | < 50 |
| 0.25 | > 50 | > 50 | 93 | > 50 | > 50 | > 50 | < 50 | > 50 | < 50 | > 50 | > 50 | 91 | > 50 | > 50 | < 50 |
| 0.125 | > 50 | < 50 | 93 | > 50 | < 50 | > 50 | < 50 | > 50 | < 50 | > 50 | > 50 | 93 | > 50 | > 50 | < 50 |
| 0.06 | < 50 | < 50 | > 50 | > 50 | > 50 | > 50 | < 50 | < 50 | < 50 | > 50 | < 50 | 92 | > 50 | > 50 | < 50 |
| Proteus vulgaris | |||||||||||||||
| 512.0 | 97 | 96 | 85 | 85 | 87 | 93 | < 50 | 92 | 92 | 79 | 98 | 95 | 91 | 99 | 96 |
| 256.0 | 96 | 96 | 85 | 85 | 84 | 85 | < 50 | 94 | 95 | 80 | 96 | 90 | 92 | 97 | 96 |
| 128.0 | 95 | 95 | 91 | 91 | 87 | 90 | < 50 | 89 | 95 | 93 | 94 | 91 | > 50 | 96 | 96 |
| 64.0 | 96 | 96 | 88 | 88 | 90 | 81 | < 50 | 86 | 95 | 94 | 89 | 85 | 89 | 95 | 95 |
| 32.0 | 97 | 97 | 87 | 87 | 89 | 82 | < 50 | 83 | 96 | > 50 | > 50 | 84 | 89 | 94 | 96 |
| 16.0 | 97 | 97 | 86 | 86 | 89 | 89 | < 50 | > 50 | 95 | 90 | > 50 | 89 | 82 | 95 | 97 |
| 8.0 | 97 | 97 | 92 | 92 | 89 | 93 | < 50 | > 50 | 94 | 96 | < 50 | 87 | 84 | 95 | 97 |
| 4.0 | 96 | 96 | 92 | 92 | > 50 | 89 | < 50 | > 50 | 93 | 93 | 51 | 95 | 85 | 95 | 97 |
| 2.0 | 96 | 96 | 89 | 89 | 82 | > 50 | < 50 | < 50 | 91 | > 50 | < 50 | 89 | > 50 | 86 | 97 |
| 1.0 | 91 | 89 | 92 | 92 | 88 | > 50 | < 50 | < 50 | 95 | > 50 | < 50 | 93 | > 50 | 91 | 97 |
| 0.5 | 96 | 96 | > 50 | > 50 | 92 | > 50 | < 50 | < 50 | 93 | > 50 | > 50 | > 50 | < 50 | 95 | 87 |
| 0.25 | 96 | 96 | > 50 | > 50 | 85 | > 50 | < 50 | < 50 | > 50 | < 50 | < 50 | < 50 | < 50 | 95 | 85 |
| 0.125 | 90 | > 50 | > 50 | > 50 | 82 | > 50 | < 50 | < 50 | < 50 | < 50 | < 50 | < 50 | < 50 | < 50 | > 50 |
| 0.06 | > 50 | > 50 | > 50 | > 50 | 86 | 85 | < 50 | < 50 | > 50 | < 50 | < 50 | < 50 | < 50 | > 50 | > 50 |
5. Discussion
In our study, we evaluated the sensitivity of planktonic forms to ciprofloxacin. It was shown that all tested strains of two species of Proteus spp., except three isolates (15.0%) of P. mirabilis coming from urine, were sensitive to this chemotherapeutic agent. This is consistent with results obtained by other researchers (11-18). Among the 80 strains of P. mirabilis analyzed by Saito isolated from urine, 13 (16.0%) were resistant to ciprofloxacin (16). This percentage of resistance strains is similar with results obtained by Hernandez et al. (13) and by Ko et al. (14), who indicated that 16.2% and 13.6%, respectively, of strains were resistant to ciprofloxacin.
The presented studies determined that some strains intermediate to ciprofloxacin were among the strains isolated from urine, and all strains isolated from the wound swabs were susceptible to this antibiotic. According to Guggenheim et al. (12), 100% of wound-swab-derived strains of Proteus spp. were susceptible to ciprofloxacin. Yah et al. (18) obtained a lower percentage (5.2%) of P. mirabilis strains from wound swabs that were resistant to that antibiotic. According to Gales et al. (11), 18.5% of P. mirabilis strains isolated from urine were resistant to ciprofloxacin. Wagenlehner et al. (17) determined that 0% - 11.6% of Proteus spp. strains isolated from urine between 1994 and 2000 were resistant to ciprofloxacin. In contrast, Abdi-Ali et al. (19) found that Acinetobacter baumannii isolated from urinary catheters were less resistant to the ciprofloxacin than those isolated from wounds.
Saito et al. (16) also drew attention to a clear increase in the incidence of P. mirabilis infections’ resistance to broad-spectrum fluoroquinolones and cephalosporins. According to the study of Kanayama et al. (20), 74.2% ESBL-positive strains were nonsusceptible to ciprofloxacin, whereas only 17.7% of the ESBL-negative strains were. The presence of ESBL among these strains can be explained by previous usage of fluoroquinolones. Saito et al. (16) also note that seven (9.0%) resistant strains were isolated from patients previously treated with antibiotics. In this study, the presence of strains intermediate to ciprofloxacin among strains delivered from urine can be caused by earlier treatment of UTI with fluoroquinolones, recommended in Poland.
A very important factor that reduces the effectiveness of antibiotics on bacterial cells is the formation of a biofilm. The strains able to form a strong biofilm can have a high level of resistance to antibiotics related to the presence of genes involved in resistance mechanisms (21). Studies have shown that all strains of P. vulgaris and P. mirabilis, regardless of the clinical material from which they were isolated, form a biofilm. This is consistent with the literature reports (3, 22-24). Jacobsen et al. (3) proved that all 50 strains of Proteus spp., isolated from urinary catheters, form a biofilm. Incubation of the tested strains, as in the case of our research, lasted 12 - 24 hours. In our experiment, it was established that 12 (60.0%) strains belonging to P. mirabilis formed a medium biofilm, and eight (40.0%) strains formed a strong biofilm. Among P. vulgaris rods were respectively 11 (55.0%) and nine (45.0%) strains. Myszka et al. (21) received comparable results of their own research. From the 50 strains of P. vulgaris, only five (10.0%) were characterized by a poor production of a biofilm, and the remaining 45 (90.0%) formed a strong biofilm (22).
In our study, it was found that the MIC50 and MIC90 values of Proteus spp. strains are lower than the MBE50 and MBE90 values. Relatively small differences in MBE and MIC values of ciprofloxacin can be explained by a good penetration of the antibiotic into the biofilm (25). Ciprofloxacin also can decrease bacteria’s ability to form biofilm (26). This is confirmed by the results of drug susceptibility of planktonic cells and biofilm strains of Klebsiella pneumoniae obtained by Bellifa et al. (27). Ciprofloxacin compared to cefotaxime and gentamicin has the lowest coefficient values of MBE to MIC (27). Singh also observed no difference in the zones of growth inhibition of biofilm cells and planktonic cells of S. aureus and S. epidermidis under the influence of ciprofloxacin (28).
Wasfi et al. (29) indicated that the reduction of P. mirabilis and P. vulgaris biofilm increased with the increase of the concentration of ciprofloxacin. The concentration of 0.5 MIC resulted in the reduction of the biofilm of P. mirabilis of 64.0% - 93.0% and a concentration of 0.25 MIC by 28.0% - 91.0%, depending on the strain (29). Similar results for P. aeruginosa strains were obtained by Ołdak and Trafny (30). Single application (concerning a 24-hour biofilm) or several times (once every 24 hours from 1 - 4 nights) at subinhibitory concentrations of ciprofloxacin reduced the biofilm mass. Also, application of ciprofloxacin at a concentration of 0.5 MIC on 24-, 48-, and 72-hour biofilms of Escherichia coli effectively reduces its weight and metabolic activity (31).
In summary, the MBE values of ciprofloxacin are close to the MIC values. The biofilm of P. mirabilis strains isolated from urine is more resistant to ciprofloxacin than the biofilm created by strains isolated from wound swabs; however, the differences are not statistically important.