1. Background
About 350 - 400 million individuals have chronic hepatitis worldwide; in other words, 5% of individuals all around the world are chronic carriers of hepatitis B virus (HBV). Thus, this infection represents a worldwide public health problem (1). Co-infection with HIV augments the risk of HBV chronicity, development of liver cirrhosis, and hepatocellular carcinoma (2). Hepatitis B viruses are classified into nine serotypes (ayw1, ayw2, ayw3, ayw4, ayr, adw2, adw4, adrq+, and adrq) and ten genotypes (A-J) (3). The position located in HBsAg at codon 124 to 147 within the major hydrophilic region (MHR) is named “a” determinant. This region of the HBs surface antigen stimulates the production of anti-HBs antibodies during the course of the initial immune response. The structure of “a” determinant is made of two loops that link together with disulfide bonds in amino acids 124/137 and 139/147; this is defined as a major B-cell epitope which is highly conserved (4). The substitution at codon 145 in “a” determinant region affects the antigenicity of the surface gene and leads to a defeat in neutralization of HBs antigen by anti-HBs antibody. This results in the escape from the host immune response.
The mutations of HBV can emerge in patients following pressure selection from either immune response or treatment options. Compared to wild-type viruses, mutations that fall out of the immunodominant epitopes of hepatitis B virus surface antigen are at undetectable levels. In some immunoassay tests, HBsAg mutants have been shown as false-negative results (5). Co-infection of HBV in HIV positive patients is common and estimated to be 5% - 15% (6). Co-infection with HIV augments the risk of HBV chronicity, development of liver cirrhosis, and hepatocellular carcinoma (HCC) (7). HIV infection has a substantial impact on the natural promotion of HBV infection, with increased levels of HBV DNA and high rate of liver- associated mortality (8).
HIV infection has a severe effect on the outcome of chronic viral hepatitis infections, and the management of these co-infected patients confronts many problems. Chronic hepatitis in HIV-positive individuals may occur due to HBV and HCV. The highest mortality is reported among multiple hepatitis compared to alone infection (9). Treatment during HIV/HBV co-infection leads to a decrease in symptoms and virus circulation of HBV DNA and HIV RNA. The accumulated mutations during the natural course of HBV have been shown in a selected manner and may be responsible for the reactivation (10). The most common HBV S gene mutations in HIV positive patients are L175S, V164E, I103T, F134V, D144E, M133I, and K122R.
2. Objectives
The aim of this study was to determine the genotype and subtype of hepatitis B virus and HBV S gene mutations in patients co-infected with HIV.
3. Methods
This study conducted on one hundred twenty four patients with confirmed HIV/ HBV co-infection, who had been referred by specialist for HBV infection diagnosis to virology diagnostic laboratory of Golestan University of Medical Sciences. None of the patients were vaccinated for HBV or had a history of immunoglobulin therapy; all patients were negative for antibodies against hepatitis C and hepatitis D viruses. Collection of samples was from May to August 2014 after approval of the study by the university ethics committee. Prior to tests being carried out, the patient's consent was obtained. Blood samples were collected in anti-coagulant EDTA 5% and plasma was separated and used for further examination. All patients were tested for serological marker of HBV (HBsAg) using commercial ELISA kit (from ACON Biotech Hangzhou, China). All the samples were HBsAg positive.
3.1. DNA Extraction and Primer Design
Extraction of HBV-DNA was performed using High Pure Viral Nucleic Acid Kit (Roche, Hamburg, Germany). 200 μL of plasma was tested for each sample according to manufacturer’s recommendations and then stored at - 20°C for PCR. Appropriate primers were used to amplify the S gene with maximum sensitivity for HBSAg and the first loop of “a” determinant. The length of applicant was 482 bp and PCRs were performed with a forward primer (5’ CTA GAC TCG TGG TGG ACT TCT C -3’) and reverse primer (5’ AAG CCA NAC ART GGG GGA AAG C 3’), corresponding to nucleotide position 250 - 732 (3).
3.2. PCR, DNA Sequencing and Mutation Analysis
100 ng of DNA extraction was added to an amplification mixture including 1X PCR buffer, 2.5 Mm of MgCl2, 0.1 Mm dNTP mix, 2.5 U of TaqDNA polymerase (QIAGEN, Hamburg, Germany), and 0.2 pmol/μL of each primer in a total volume of 50 μL with distilled water. The PCR program consisted of 5 minutes denaturation at 94°C, 30 cycles of amplification including denaturation for 20 Sec at 94°C, primer annealing for 30 Seconds at 60°C, and extension for 40 Seconds at 72°C, with a final extension at 72°C for 5 minutes. PCR products were analyzed by 1.5% agarose gel electrophoresis and stained with ethidium bromide. Direct sequencing was performed to positive PCR products (Macro gene, South Korea) and surface gene was aligned using standard hepatitis B sequence [Accession number: AB033559 and GU938305] from the gene bank database. Amino acid mutations were determined using blast, gene runner, and chromas programs.
4. Results
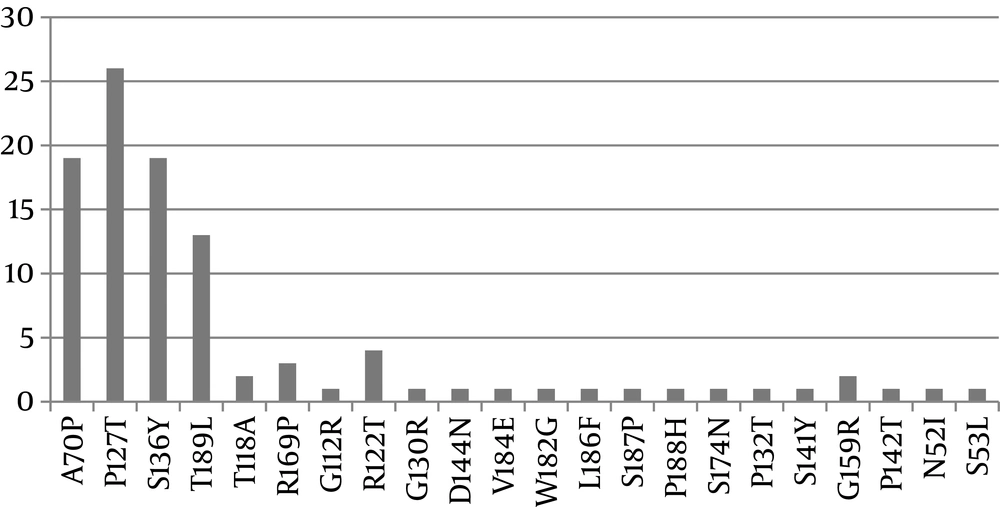
In this study, 8 (20%) participants were female, with a mean age of 33.88 years. A minority of the patients (10%) were HBeAg positive. The results of reference sequence revealed that all sequences belonged to genotype D, subgenotype D1, subtype ayw2. In the Amino acid mutation level, our results showed the occurrence of 50 point mutations composed of 23 mutations (46%) as missense (amino acid altering) and 27 mutations (%54) as silent. Our findings also showed that P127T and A70P were the most frequently occurring substitution mutations, as shown in Figure 1. Based on statistical analysis in SPSS, there was no significant association between the age and sex of patients and the patterns of amino acid substitution. P127T, P132T, G130R, and S136Y substitutions placed in the first loop of the “a” determinant and the other substitutions of P142T and D144N occurred in the second loop of “a” determinant.
| Variable | Total HIV/HBV Patients | Genotype |
|---|---|---|
| Patients, n (%) | 124 | D |
| Age, years, mean (IQR) | 33.88 | D |
| Male sex, n (%) | 80% | D |
| CD4+ T-cell count, cells/mm3, median (IQR) | 130 - 220 | D |
| HBeAg positive, n (%) | 10% | D |
| HBV DNA, log10IU/mL, median (IQR) | 5.7 (2 - 7.65) | D |
4.1. Amino Acid Substitutions Within the HBS Ag Immune Epitopes
Out of 23 amino acid mutations which occurred in different immune epitopes within the surface Antigen, 11 (47.82%) occurred in B cell epitope, 6 (26.08%) in T- helper, and 2 (%8.6) in CTL epitopes (Table 2).
| Mutation | HLA Restriction | Cell Subsets | Sequence |
|---|---|---|---|
| - | B | 100 - 160 | |
| P142T, G159R, G112R | Class II | Th (CD4) | 160 - 100 |
| C149S, P127 T,S136Y,S136Y | Class II | Th (CD4) | 100 - 160 |
| R122T,T118A,G130R | Class II | Th (CD4) | 100 - 160 |
| V176L,S174N | Class I | CTL | 171 - 179 |
| V176L, W182G,V184E | Class I HLA-A2 | 175 - 184 | |
| T189L,S139K, T189M,P188H | Class II | Th (CD4) | 197 - 186 |
The phylogenetic tree was drawn using the “maximum likelihood” method with bootstrap test (500 replicates). The strains were classified into two clusters A and B. The type A included 12 isolates with 78% similarity divided into two sub-groups I and II. Two isolates S151 F and S64 F are distant members of other isolates belonging to this group. The two species KU212155.1 and KJ810962.1 are sister taxa. There are two sub-groups in type B that were separated with a value of 49%. In sub-group II, s68F, s89F, and S98F had 79% similarity and more distant from other species.
5. Discussion
The comparison of sequences found in this study with the gene bank standard sequence [Accession number: AB033559] and achieved Iranian sequences [Accession number: GU938305] showed all isolates belonged to the genotype D, sub-genotype D1, and subtype ayw2. Based on the results, our study is comparable with other studies conducted in Iran that found Genotype D, sub-genotype D1, and subtype ayw2 are accounted for 100% of isolates (Hamkar et al., (11) Norouzi et al. (12).
We found a few mutations at codons 122 (15%) and 160 (0%), which are known to dictate the d/y and w/r subtypic determinants. This is similar to the reports that indicate subtype-specific positions are supposed to be tolerant to random mutations (13). Some substitutions are able to defect the binding of neutralizing antibodies to the HBV surface antigen especially in “a” determinant region. Mutations that occur in this area include P127T, S136Y, P142T; D144N causes structural changes in the antigenicity of the surface gene and affects the affinity of antibodies. Viruses carrying mutations in T-cell epitopes cannot be recognized by specific T-cells of an individual, and anti-HBs production will not enhance in this group of patients (14). The mutation of Th and CTL epitopes in these patients indicates an ineffective T cell immune response.
In comparison with other immune epitope mutations in this study which distributed in different residues, the mutations of 2 CTL epitope altered in only 2 amino acid residues, suggesting a poor centralization of immune selection pressure at a hot spot position in this selection. It has already been shown that these responses are weak and sometimes the immune system is weakened during co-infection of HIV/HBV due to the absence of selective pressure that causes the virus to undergo a mutation (10). In a previous study, Huang et al. (2004) (15) found that mutations at amino acid positions 135, 126, 136, 145, and 129 of “a” - determinant occur in conjunction with the absence of detectable anti HBsAg in samples from Qidong in China (1).
Some studies have shown co-infection with HBV may be associated with a more progression course of HIV disease. Brown et al. (16) showed mutations within the “a” determinant during the natural history of infection are significantly observed within the first loop (aa 107 to 138), whereas those induced under immune pressure due to active and /or passive immunization are more frequently observed within the second loop (aa 139 to 147).
In our study, mutations of the P127T, G130R, P132R, and S136Y in the first loop and mutations of P142T and D144N in the second loop were observed (2). Similar mutations have been reported in the study of Kamili (17). G145R mutation was detected neither in our study nor in the study of Julie Sheldon in 2007 (4). However, we detected substitution A70P, P127T, and S136Y in the current study more frequently in HIV co-infected patients with genotype D. We compared the results of mutations in the S gene of HBV between co-infected HIV/HBV and mono-infected HBV chronic patients. The comparison showed that the co-infections of HIV/HBV had fewer mutations than mono-infections. This may be due to the weakness of the immune system and absence of selective antibody pressure in these patients. Several studies have shown that some genotypes of hepatitis related to severe hepatic diseases. Hepatitis B virus genotypes A and D are prevalent in chronic liver disease patients of Indian origin; and HBV genotype D is associated with more severe diseases and may predict the occurrence of HCC in young patients. Future research should focus on determining the clinical significance of the different HBV genotypes and mutations found in this population.
| Region | Common Amino Acid Substitutions | Co Infection | Mono Infection |
|---|---|---|---|
| B cell | N131T, I133M, H134Y, R159G, N140T, I117S, S120P, T122R, F114S, R109L, L110I, R112G | R159G, ,T122R,R112G | N131T, I133M, H134Y, R159G, N140T, I117S, S120P, T122R, F114S, R109L, L110I, R112G |
| TH cell | L62P, S65C, L80F C83F, C85F, M87L I89L, T92I, A96V, R186L, L189T G190V, A191W Y192L | L189T | L62P, S65C, L80F, C83F, C85F, M87L, I89L, T92I, A96V, R186L, L189T, G190V, A191W, Y192L |
| T CELL | N174S, L177V | N174S | N174S, L177V |
| Adeterminant | N131T, R133M, I133M, H134Y, N140T | S136Y, D144N ,G130R, P132T, P142 ,P127T | N131T, R133M, I133M, H134Y, N140T |
| MHR | Y100F, L109R, I110L, G112R, S117I, P120S/T, R122K/T, M133I, Y134H, T140I, S143L, G145R, G159R, E164V | G112R,R122T, G159R | Y100F, L109R, I110L, G112R, S117I, P120S/T, R122K/T, M133I, Y134H, T140I, S143L, G145R, G159R, E164V |
5.1. Conclusion
Mutations in the HBsAg of HBV/HIV co-infected patients presumably may be an ordinary exhibition, which might have some implications, eventually leading to the progression of chronicity, cirrhosis, and hepatocellular carcinoma in hepatitis B virus infection. The global frequency of MHR mutations is 27.8%. The frequency of MHR mutations for chronic carriers was 24% in Japan, up to 28% in Taiwan (18), close to 40% in Spain (19), and 50% in Korea (20). This frequency was 21.73% in our study compared to a global range of mutation in the MHR region of the HBV S gene. This study showed the frequency of mutations in MHR region similar to the prevalence of MHR mutations reported from other parts of the world. Our results showed that HBV surface gene sequencing and genotyping may be promising to treat patients effectively with antiviral chemotherapy for chronic hepatitis B in patients co-infected with HIV.