1. Background
Whooping cough is caused by Bordetella pertussis, a Gram-negative bacterium. It is one of the main causes of infant death in the world, and it remains a public health problem even in regions with high coverage vaccination. Whole-cell pertussis (wP) vaccines have been encouraged and broadly employed for expanded programs on the immunization of children. They have been used in the world as a component of a combined diphtheria, tetanus, and pertussis (DTP) vaccine in national children vaccination plans for many years (1).
The relation of wP vaccine with cases of persistent cerebral injury has not be confirmed (2, 3), since it may be the result of fever, irritability, seizures, hypotension, or local side effects (4). However, the possible side effects resulting from the administration of wP vaccines has led to the development of acellular pertussis (aP) vaccines in some countries, though wP vaccines are still broadly manufactured and universally employed (5). In comparison to the aP vaccine, which needs vast purification procedures, the wP vaccine is more economical. Furthermore, the resurgence of whooping cough in nations where aP vaccination is high coverage has led to relevant research demonstrating that wP vaccines create superior and continuing immunity compared to aP vaccines. Therefore, it is predicted that wP vaccines will be used for the next years (6-9).
Suspensions of inactivated B. pertussis micro-organisms are the main immunizing ingredient in wP vaccines, which are produced by culturing suitable B. pertussis strains under states that provoke the expression of the bacteria phenotype of virulent Phase I. The bacteria culture in semi-synthetic or synthetic media has been employed in vaccine preparation for a number of years (10). The bacteria strains are chosen to contain agglutinogen serotypes 1, 2, and 3, which are at this time attributed to fimbriae serotypes 1, 2, and 3. Generally, one or more strain of each bacteria serotypes 1, 2 and 1, 3 are employed; however, some manufacturers use a single strain of serotype 1, 2, and 3 (11).
There is no doubt as to the efficacy of a whole cell pertussis vaccine, but it is necessary to improve the quality of the vaccine specially to decrease its toxicity (3, 12). In order to decrease the toxicity of wP vaccines, procedures of detoxifying all the toxins of B. pertussis organisms without destroying the immunogenicity have been considered (13). On this basis, Gupta et al. evaluated the effects of various chemical agents (such as glutaraldehyde, formaldehyde, acetone, and thimerosal) in different temperature conditions on the inactivation of B. pertussis cells (14). Moreover, some studies explained the preparation of a potent and safe pertussis vaccine with good stability using the glutaraldehyde inactivation process (15-19).
2. Objectives
In this study, to achieve optimal conditions for preparing a low toxic and highly potent B. pertussis whole-cell vaccine, the effects of different chemicals and temperatures were investigated for inactivation of B. pertussis micro-organisms. The samples were prepared by killing B. pertussis micro-organisms using various concentrations of chemicals with variable durations and temperatures, which were analyzed in mice for intra-cerebral potency and toxicity.
3. Materials and Methods
3.1. Materials
In this study, we used formaldehyde, glutaraldehyde, and thimerosal (Merck, Germany). The B. pertussis culture of strains 509 (batch number P93-19, optical density 15, 15 × 109 bacterial particle/mL) and 134 (batch number P93-20, optical density 15, 15 × 109 bacterial particle/mL) were obtained from the Razi Vaccine and Sera Research Institute (Alborz, Iran). Freeze-dried, mono-specific pertussis agglutinogen antiserum for factors 1, 2, and 3 was purchased from NIBSC (UK). All other chemicals were reagent grade.
3.2. Animals
Healthy Saurian strain mice < 5 weeks old from the same stock with weighing 10 to 18 g were used, and the difference in weight between the heaviest and the lightest was not greater than 5 g. They were used for evaluating the intracerebral potency of B. pertussis inactivated cells and mice with weight 14 to 16 g in toxicity test. The study was reviewed by the Pasteur institute of Iran, and all animals were handled in accordance with institutional guidelines.
3.3. Bordetella Pertussis Strains
Bordetella pertussis strains 509 (serotypes 1 and 2) and 134 (serotypes 1 and 3) were obtained from the Human bacterial vaccine division of the Razi vaccine and serum research institute, and the strains were used for pertussis preparation. Bordetella pertussis strain 18323 (serotype 1, 2, and 3) and ATCC 9797 were obtained from the aforementioned institute and employed as a challenge bacteria in the mouse for an intracerebral protection test (MPT) as a current potency test for pertussis vaccines.
3.4. Harvesting
Culture suspensions of B. pertussis from the fermenter were transferred to the Huading separator. Harvesting was carried out at a separator flow rate of 250 to 300 L/h, 6000 rpm. The ejaculated micro-organism mass was collected in a sterile, non-pyrogen vessel; normal saline was added to adjust optical density (OD) of suspension in the 150 (150 × 109 bacteria particle/mL) to make up the required volume of a pellet.
3.5. Inactivation of B. Pertussis Organisms
The bacterial suspensions of B. pertussis strains 509 and 134 were homogenized by shaking at 50 rpm, in 5 ± 3°C for 1 hour; a 1000 mL sample was taken from each suspension. The samples were then homogenized by shaking at 50 rpm, in 5 ± 3°C for 1 hour. In this study, 40 mL of each of the 21 samples was prepared from the bacterial suspensions and labelled F1 to F21. Each part was treated using one of the specified inactivating conditions mentioned (Table 1). The inactivated bacterial suspensions were immediately cooled to below 25°C after completion of the inactivation process and sampled for tests of opacity, non-viability, agglutination, purity, and sterility.
| Formulation | Heat (°C) | F (%w/v) | G (%w/v) | T (%w/v) | Duration (min) | Shaking (rpm) |
|---|---|---|---|---|---|---|
| F1 | 56 | 10 | 50 | |||
| F2 | 56 | 20 | 50 | |||
| F3 | 56 | 30 | 50 | |||
| F4 | 37 | 0.05 | 24 h | 50 | ||
| F5 | 37 | 0.05 | 36 h | 50 | ||
| F6 | 37 | 0.10 | 24 h | 50 | ||
| F7 | 37 | 0.10 | 36 h | 50 | ||
| F8 | 37 | 0.15 | 24 h | 50 | ||
| F9 | 37 | 0.15 | 36 h | 50 | ||
| F10 | 22.5 | 0.025 | 10 | 50 | ||
| F11 | 22.5 | 0.025 | 15 | 50 | ||
| F12 | 22.5 | 0.05 | 10 | 50 | ||
| F13 | 22.5 | 0.05 | 15 | 50 | ||
| F14 | 22.5 | 0.075 | 10 | 50 | ||
| F15 | 22.5 | 0.075 | 15 | 50 | ||
| F16 | 37 | 0.01 | 24 h | 50 | ||
| F17 | 37 | 0.01 | 36 h | 50 | ||
| F18 | 37 | 0.02 | 24 h | 50 | ||
| F19 | 37 | 0.02 | 36 h | 50 | ||
| F20 | 37 | 0.03 | 24 h | 50 | ||
| F21 | 37 | 0.03 | 36 h | 50 |
Abbreviations: F, formaldehyde; G, glutaraldehyde; T, Thimerosal; min, minute.
3.6. Pooling of Inactivated Bordetella Pertussis Bacterial Suspensions
The inactivated suspensions of strains 509 and 134 were homogenized by shaking at 50 rpm, in 5 ± 3°C for 1 hour; thereafter, the same formulation samples were pooled together. The mixture of inactivated bacterial suspensions were homogenized by stirring at 50 rpm, in 5 ± 3°C for 3 hours and then sampled for sterility, agglutination, opacity, specific toxicity, and potency tests.
3.7. Test for Non-Viability
The samples of inactivated bacterial suspensions were tested for absence of B. Pertussis viability by inoculating Bordet-Gengou agar slope and incubating at 35°C for 7 days (20).
3.8. Estimation of Opacity Unit
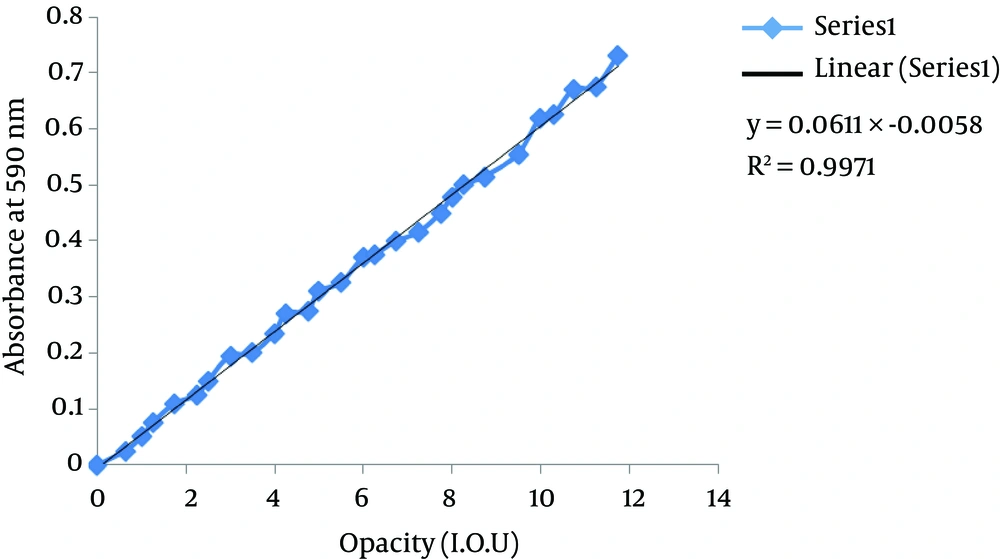
The B. pertussis bacterial suspensions’ opacity was measured both by visual comparison with the samples of the WHO’s 5th International Standard of Opacity (20, 21), which is equivalent to 10 IU of opacity (10 × 109 organisms per mL) (22), and the spectrophotometric method, which evaluates the sample at 590 nm (23). Briefly, serial dilutions of the bacterial suspension were prepared in saline; then, the opacity of the samples were standardized based on the international reference for opacity. The absorbance of each dilution was measured at 590 nm, and the results were drawn to obtain a calibration curve (Figure 1).
3.9. Estimation of Purity
Before they were killed, the samples of B. pertussis bacterial suspensions were analyzed for purity by microscopic evaluation of Gram stained smears and by culture into nutrient agar, Bordet-Gengou agar, and nutrient broth cultures media (24).
3.10. Test for Agglutinin Production
To study the expression of serotype specific fimbriae by B. pertussis bacteria and the effects of inactivation conditions on agglutinins, bacterial suspensions were examined before and after inactivation using the slide agglutination assay method (24). Briefly, 1 mL of the B. pertussis bacterial suspension was centrifuged, and the sediment was resuspended in 0.1 mL normal saline and homogenized. Then, 10 µL of the reconstituted sample was mixed with 10 µL of monospecific antiserum of agglutinins 1, 2 and, 3 separately. The slide gently rotated for 2 to 3 minutes, and agglutination was observed.
3.11. Test for Toxicity
The overall toxicity of the inactivated B. pertussis suspensions was performed by a mouse weight gain test (MWGT). Briefly, samples of the inactivated bacteria were diluted by normal saline to 40 opacity unit/mL, and each mouse was injected intraperitoneally with 0.5 mL of the samples. The average weight gain of the animals in the test groups was compared with the control group for 7 days (25, 26).
3.12. Potency Measurement
The potency of inactivated B. pertussis bacterial suspensions were measured by the intracerebral mouse protection method. The ED50 quantity for the test samples were estimated by probit analysis (24, 25, 27).
4. Results
4.1. Bacterial Harvest Characteristics
The bacterial harvest of strains 509 and 134 showed purity for cultural staining (a Mercury drop-like appearance on BG slants and no growth in nutrient agar and broth) and morphological specifications of B. pertussis (Gram negative, coccobacilli). The bacterial suspension of strain 509 contained agglutinogens 1 and 2, and strain 134 contained agglutinogens 1 and 3. The opacity unit of organisms in the harvests were 150 × 109/mL for each batch.
4.2. Estimation of Opacity
In this work, the spectrophotometric method for the measurement of the B. pertussis bacterial suspensions’ opacity was distinguished to be straightforward and was favored; this was because it allowed the estimation of slight variances in opacity, which were indistinguishable by the optical opacity method (Figure 1). The B. pertussis suspensions’ opacity in various stages (before inactivation, after inactivation, and after pooling of inactivated bacterial suspensions) of work was measured (Table 2).
| Before Inactivation | Inactivation Status | After Inactivation | |||
|---|---|---|---|---|---|
| Strains | Opacity (Organism/mL) | Strain 509 (Organism/mL) | Strain 134 (Organism/mL) | Pooled Suspensions (organism/mL) | |
| 509 | 150 × 109/ mL | F1 | 139 × 109 | 138 × 109 | 139 × 109 |
| F2 | 138 × 109 | 138 × 109 | 138 × 109 | ||
| F3 | 133 × 109 | 132 × 109 | 132 × 109 | ||
| F4 | 142 × 109 | 144 × 109 | 141 × 109 | ||
| F5 | 141 × 109 | 141 × 109 | 141 × 109 | ||
| F6 | 141 × 109 | 142 × 109 | 141 × 109 | ||
| F7 | 140 × 109 | 140 × 109 | 140 × 109 | ||
| F8 | 140 × 109 | 141 × 109 | 141 × 109 | ||
| F9 | 138 × 109 | 138 × 109 | 138 × 109 | ||
| F10 | 141 × 109 | 142 × 109 | Exa | ||
| 134 | 150 × 109/ mL | F11 | 139 × 109 | 138 × 109 | 139 × 109 |
| F12 | 140 × 109 | 139 × 109 | 139 × 109 | ||
| F13 | 139 × 109 | 138 × 109 | 139 × 109 | ||
| F14 | 138 × 109 | 139 × 109 | 139 × 109 | ||
| F15 | 138 × 109 | 137 × 109 | 138× 109 | ||
| F16 | 143 × 109 | 142 × 109 | Exa | ||
| F17 | 141 × 109 | 141 × 109 | 141 × 109 | ||
| F18 | 141 × 109 | 140 × 109 | 141 × 109 | ||
| F19 | 139 × 109 | 139 × 109 | 139 × 109 | ||
| F20 | 138 × 109 | 139 × 109 | 139 × 109 | ||
| F21 | 138 × 109 | 138 × 109 | 138 × 109 | ||
aExcluded from study.
4.3. Test for Non-Viability
The non-viability test on B. pertussis organisms after inactivation showed that the micro-organisms of strains 509 and 134 treated under F10 and F16 conditions were not inactivated, while every other inactivation condition killed bacterial organisms. Therefore, bacterial suspensions of F10 and F16 were eliminated from the study, and other bacterial suspensions of both strains were pooled together for further evaluation and tests.
4.4. Test for Agglutinin
In this study, the expression of serotype specific fimbriae by B. pertussis bacteria was examined by slide agglutination assay. In order to evaluate the effects of inactivation conditions on agglutinins, bacterial suspensions were examined after inactivation and pooling for agglutinins (Table 3).
| Before Inactivation | Inactivation Status | After Inactivation | |||
|---|---|---|---|---|---|
| Strains | Agglutinins | Strain 509 | Strain 134 | Pooled Suspensions | |
| Agglutinins | |||||
| 509 | 1, 2 | F1 | 1, 2 | 1, 3 | 1, 2, 3 |
| F2 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F3 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F4 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F5 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F6 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F7 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F8 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F9 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F10 | 1, 2 | 1, 3 | - | ||
| 134 | 1, 3 | F11 | 1, 2 | 1, 3 | 1, 2, 3 |
| F12 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F13 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F14 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F15 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F16 | 1, 2 | 1, 3 | - | ||
| F17 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F18 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F19 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F20 | 1, 2 | 1, 3 | 1, 2, 3 | ||
| F21 | 1, 2 | 1, 3 | 1, 2, 3 | ||
4.5. Test for Toxicity
The results of the MWGT overall toxicity assessment on the inactivated B. pertussis suspensions are shown in Table 4. This test demonstrated acceptable outcomes for bacterial suspensions inactivated under conditions of F2 (56°C, 20 minutes and 50 rpm), F4 (formaldehyde 0.05% w/v, 37°C, 24 hours, 50 rpm), F8 (formaldehyde 0.15% w/v, 37°C, 24 hours, 50 rpm), F12 (glutaraldehyde 0.05% w/v, 22.5°C, 10 minutes, 50 rpm ), F15 (glutaraldehyde 0.075% w/v, 22.5°C, 15 minutes, 50 rpm), and F17 (thimerosal 0.01% w/v, 37°C, 36 hours, 50 rpm).
| Pooled Killed B. pertussis Bacterial Suspensions | Average Weight Gain (g) Per Mouse | Death Number and Days | ||
|---|---|---|---|---|
| Day 3 | Day 7 | Percent Weight Gain Relative to the Control | ||
| Toxicity (MWGT) Test | ||||
| F1 | 0.8 | 0.3 | 26.1 | 1 (day 4) |
| F2 | 0.8 | 1.1 | 95.6 | - |
| F3 | 1.15 | 0.1 | 8.7 | 1 (day 4), 1 (day 5) |
| F4 | 0.3 | 0.8 | 69.6 | - |
| F5 | 0.3 | 0.6 | 52.2 | 2 (day 4) |
| F6 | 0.8 | 0.6 | 52.2 | 2 (day 5) |
| F7 | 0.95 | 0.3 | 26.1 | 1 (day 2), 1 (day 6), 1 (day 7) |
| F8 | 0.8 | 0.75 | 65.2 | - |
| F9 | 0.9 | - 0.3 | -26.1 | 1 (day 4), 1 (day 6) |
| F11 | - 0.75 | 0.95 | 82.6 | 1 (day 5) |
| F12 | 0.0 | 0.75 | 65.2 | - |
| F13 | - 0.5 | 0.4 | 34.8 | 1 (day 4), 1 (day 6) |
| F14 | - 0.45 | 0.5 | 43.5 | 1 (day 3) |
| F15 | 0.0 | 1.25 | 108.7 | - |
| F17 | 0.9 | 0.75 | 65.2 | - |
| F18 | 0.8 | -0.5 | -43.5 | 2 (day 4),1 (day 6) |
| F19 | 0.2 | 0.1 | 8.7 | 1 (day 2), 1 (day 4), 1 (day 5) |
| F20 | 0.25 | 0.25 | 21.7 | 1 (day 3) |
| F21 | - 0.25 | 0.05 | 4.3 | 1 (day 4) |
| Control | 1.25 | 1.15 | - | - |
| Pooled Killed B. pertussis Bacterial Suspensions | Dose (mL) | Challenge Dose (LD50) | ED50 (Human dose) | |
| Potency Test | ||||
| F2 | 0.5 | 100 | 1:62 | |
| F4 | 0.5 | 100 | 1:83 | |
| F8 | 0.5 | 100 | 1:100 | |
| F12 | 0.5 | 100 | 1:200 | |
| F15 | 0.5 | 100 | 1:83 | |
| F17 | 0.5 | 100 | 1:333 | |
4.6. Test for Potency
The in vivo potency test was performed on the pooled B. pertussis bacterial suspensions F2, F4, F8, F12, F15, and F17. Table 4 shows that the ED50 of these bacterial suspensions were calculated. The potency analysis of the aforementioned suspensions showed their ED50 order as follows: F17 > F12 > F8 > F15, F4 > F2. The bacterial suspension of inactivation condition F17 indicated a higher ED50 (1:333 of a human dose) compared to other inactivation conditions.
5. Discussion
Bacterial suspensions should not be utilized in the manufacturing of vaccines if impurity has been observed at any step in their preparation. From this point of view, bacterial characteristics such as morphology, purity, and appearance of bacterial suspensions should be controlled in bacterial harvest. The opacity of harvested bacterial suspensions should be monitored in order to estimate bacteria count per volume (5). The harvests of B. pertussis bacterial strains 509 and 134 showed purity by Gram staining, cultural evaluation, and from the point of appearance.
In this study, the bacterial count in suspensions of B. pertussis before and after inactivation and after pooling of inactivated bacterial suspension was measured. Table 2 shows that the average opacity of suspensions was decreased under different treatment conditions from 8% to 12% by heat, 6% to 8% in formaldehyde, 6% to 8% in glutaraldehyde, and 5% to 8% in glutaraldehyde. Various inactivating factors lyse B. pertussis cells with different intensities and decrease the opacity of bacteria suspension. These results are in agreement with other reports (19, 28).
The protective role of anti-agglutinogens (1, 2, and 3) is important for fighting whooping cough (5, 29). As is shown in Table 3, the outcomes of this research indicated that the bacterial harvests of strain 509 contained agglutinins 1 and 2, and strain 134 contained agglutinins 1 and 3. A test for agglutinins on bacterial suspensions 509 and 134 after inactivation under various conditions and pooled inactivated bacterial suspensions (Table 3) showed inactivation conditions due to heat, formaldehyde, and glutaraldehyde not degraded agglutinins of both B. pertussis strains. There are other reports with similar findings to our results, showing that inactivated agents such as formaldehyde, glutaraldehyde, and heat do not destroy agglutinins of B. pertussis during the inactivation process (18, 19, 30). This work also revealed that thimerosal does not change agglutinins of B. pertussis during the inactivation conditions of F16 to F21.
The overall toxicity test on inactivated bacterial suspension pools by the MWGT method after the inactivation process showed that various inactivation condition parameters had different influence patterns on the toxicity of bacterial suspensions (Table 4). Previous studies showed that in the MWGT, the early deaths of mice is a sign of the dermonecrotic toxin (DNT) content of the vaccine; the endotoxin content is responsible for the weight loss at 24 hours, and the leucocytosis promoting factor (LPF) content is the reason for the reduced rate of late weight gain (13, 31). Results of this work revealed that inactivated bacterial suspensions F5, F11, F13, F14, and F21 resulted in early weight loss while F1, F3, F7, F9, F18, and F19 caused a reduced rate of late weight gain. The bacterial suspensions inactivated under the conditions of F2, F4, F8, F12, F15, and F17 were passed through the toxicity test from the points of weight gain pattern and death of the mice. In all of the other formulations, except to these six selected bacterial suspensions that passed toxicity test, either deaths or undesirable weight gain pattern were observed.
For a long time, manufacturers have used the Kendrick test for potency analysis of B. pertussis cellular vaccines (1, 32, 33). The six selected inactive bacterial suspension pools (F2, F4, F8, F12, F15 and F17) that passed the toxicity test were comforted under potency estimation. Table 4 shows the ED50 of these bacterial suspensions calculated. The results presented in this table showed that the ED50 of selected formulations had the following order: F17 > F12 > F8 > F15, F4 > F2, and the bacterial suspension pool F17 indicated a higher ED50 (1:333 of a human dose) compared to other formulations.
Improving the presently accessible wP vaccine is necessary, especially in regard to decreasing toxicity while preserving the vaccine’s efficacy. This aim is achievable if attention is given to procedures used in detoxifying all the toxins of B. pertussis, without destroying its immunogenicity (12, 34). The results obtained in this study revealed high immunogenicity for an inactivated bacterial suspension pool in F17, which had the smallest bacteria content. It seems that the inactivation condition of F17 is able to detoxify B. pertussis toxins without destroying the potency of the vaccine.
It can be concluded that the inactivating condition of F17 successfully inactivates B. pertussis suspension. This formulation also showed desirable outcomes in the overall toxicity test and good immunogenicity with low bacteria content. Therefore, less adverse effects and better immunogenicity are foreseeable for vaccine preparation using this method.