1. Background
Amoxicillin-clavulanate (also known as augmentin) is a broad-spectrum antibacterial agent used in the treatment of community-acquired respiratory tract infections. An increasing prevalence of amoxicillin-clavulanate resistance has been reported, notably due to the continued spread of β-lactamases-mediated resistance. These enzymes act by cleaving the lactam ring of susceptible antibiotics via an irreversible hydroxylation of the amide bond. β-lactamase enzymes in bacteria are very diverse, and they mutate continuously in response to the heavy pressure of antibiotic use, leading to the development of extended spectrum β-lactamases (ESBLs). Most of the ESBLs are members of the TEM and SHV β-lactamase families, whereas other groups for example CTX-M, OXA, and PER β-lactamase have been discussed more recently (1). ESBLs are transferable, and they contain a number of mutations that allow them to hydrolyze expanded-spectrum β-lactam antibiotics, such as penicillin; first-, second-, and third generation cephalosporins; and aztreonam (2-4).
β-lactamases resistant to inhibition by clavulanic acid were discovered in the early 1990s; these enzymes are mostly variants of TEM-1 or TEM-2 β-lactamase (4). Inhibitor-resistant TEM β-lactamases have been found mainly in clinical isolates of Escherichia coli and in some strains of Klebsiella pneumoniae, K. oxytoca, Proteus mirabilis, and Citrobacter freundii (4). Amoxicillin-clavulanate resistance seems to be mainly associated with hyperproduction of plasmid-mediated TEM enzymes, production of oxacillinases, or due to co-expression of TEM enzymes with OXA-like and/or SHV β-lactamases (5, 6). A study in France showed that up to 41% of amoxicillin-clavulanate resistant Escherichia coli in a hospital were producers of inhibitor-resistant TEM variants (6). Hyperproduction of plasmid and chromosomal-mediated AmpC may also cause amoxicillin-clavulanate resistance. AmpC-type β-lactamases are clinically important cephalosporinases, and their overexpression confers resistance to broad-spectrum cephalosporins, but they are not inhibited by clavulanic acid (7, 8). AmpC enzymes are encoded by both chromosomal (blaAMPC) and plasmid genes (blaDHA and blaCMY) (7). There was also a significant association between the Sul gene and resistance to amoxicillin-clavulanate (9).
The three food categories most frequently associated with antibiotic resistant pathogens and foodborne outbreaks are dairy products, ground beef, and poultry (10, 11). Retail meat products can serve as important sources for the transfer of multidrug resistant bacteria from food-producing animals to the public. Foodborne bacteria often carry high levels of resistance due to the large amounts of antibiotics often given to food-producing animals in feed and water for growth promotion and other non-therapeutic purposes. Studies have shown that feeding low doses of antibiotics to large numbers of food animals over long periods of time leads to resistance (12). Recently, increasing prevalence of antibiotic resistance genes recovered from food sources like poultry has been reported; this is related to the use of antibiotics as growth promoters in livestock (13). Resistance towards amoxicillin-clavulanate in foodborne pathogens has also been reported in China and Nigeria (14, 15). In our previous study, blaCTXM-2, blaSHV, and blaTEM genes were discovered in bacteria isolated from retail sausages in Malaysia (16).
2. Objectives
The present study aimed to identify the gene determinants responsible for amoxicillin-clavulanate resistance in bacteria isolates obtained from retail sausages. Sausages as poultry products could be a source of multidrug-resistant bacteria, which may spread to and cause the development of antibiotic resistance in humans.
3. Methods
3.1. Bacterial Isolation and Identification
Seven different types of retail sausages were collected in Kampar, Perak between January and November, 2014. The selected sausages were chopped into small pieces and weighed to approximately 2 grams before they were transferred into sterile Luria-Bertani (LB) enrichment broths (Laboratories CONDA, Spain). These broths were incubated at 37°C for 16 to 18 hours with agitation. Afterwards, a loop full of bacteria from LB enrichment broth was streaked onto MacConkey agar petri dishes (Oxoid, England) and incubated at 37°C for 24 hours. MacConkey agar was used to selectively grow the Gram negative bacteria and differentiate the lactose fermentation. The purified isolates were preserved and suspended in 80% (v/v) glycerol and stored at -80°C. Further identification of the isolates was carried out using API strips (bioMerieux®, France).
3.2. Antibiotic Susceptibility Test
The isolated bacteria from MacConkey agar was inoculated into 5 ml of Tryptic soy broth (TSB) (Merck, Germany) and incubated at 37°C. After 24 hours, the inoculum with turbidity equivalent to 0.5 McFarland standards were then inoculated evenly on Mueller-Hinton agar (Oxoid, England) using a sterile cotton swab. The antibiotics used were amoxicillin-clavulanate (AMC-30), aztreonam (ATM-30), cefotaxime (CTX-30), gentamicin (CN-10), ampicillin (AMP-10), tetracycline (TE-30) (all from BD BBLTM, United States), cefpodoxime (CPD-10), ceftazidime (CAZ-30), ceftriazone (CRO-30), imipenem (IMP-10), meropenem (MEM-10), norfloxacin (NOR-10), oxacillin (OX-1), penicillin (P-1), and piperacillin (PRL-75) (all from Oxoid, England). The discs were placed on the Mueller-Hinton agar (Oxoid, England) plate. The plates were then inverted and incubated aerobically at 37°C for 16 to 18 hours. In this study, indication of the bacteria as “susceptible,” “intermediate,” or “resistant” towards the antibiotic discs was based on the Zone Diameter Interpretative Standards of the Clinical Laboratory Standard Institute (CLSI) (17).
3.3. Double Disc Synergy Test
This test is a recommended routine practice to detect ESBL production in Gram negative bacilli. The test was performed by using an amoxicillin-clavulanate (AMC-30) (BD BBLTM, United States) disc. The disc was placed at the center of the Mueller-Hinton agar plate, and ceftazidime (CAZ-30) (Oxoid, England) and cefotaxime (CTX-30) (BD BBLTM, United States) were placed 15 mm away from the disc at the center. The plate was incubated overnight at 37°C and then examined. The presence of the keyhole phenomenon was considered a positive result for ESBL production (17).
3.4. Phenotypic Confirmatory Test
A sterile cotton swab was dipped into the organism suspension. The swab was then swabbed over the dry surface of a Mueller-Hinton agar plate to obtain a lawn culture. Discs containing cefotaxime, cefotaxime-clavulanic acid (BD BBLTM, United States), ceftazidime, or ceftazidime-clavulanic acid (Oxoid, England) were arranged so that the distance between them was approximately twice the radius of the inhibition zone produced by the cephalosporin. Based on CLSI guidelines, the isolates were classified as ESBL producers if there was more than (≥) a 5 mm increase in the zone diameter for either ceftazidime or cefotaxime with clavulanic acid versus its zone when tested alone (17).
3.5. Total and Plasmid DNA Extraction
Bacterial isolates were subjected to total DNA extraction using a fast-boiling method (18). Plasmid DNA was extracted using the DNA-spinTM Plasmid DNA Purification Kit (Intron, South Korea).
3.6. Polymerase Chain Reaction (PCR)
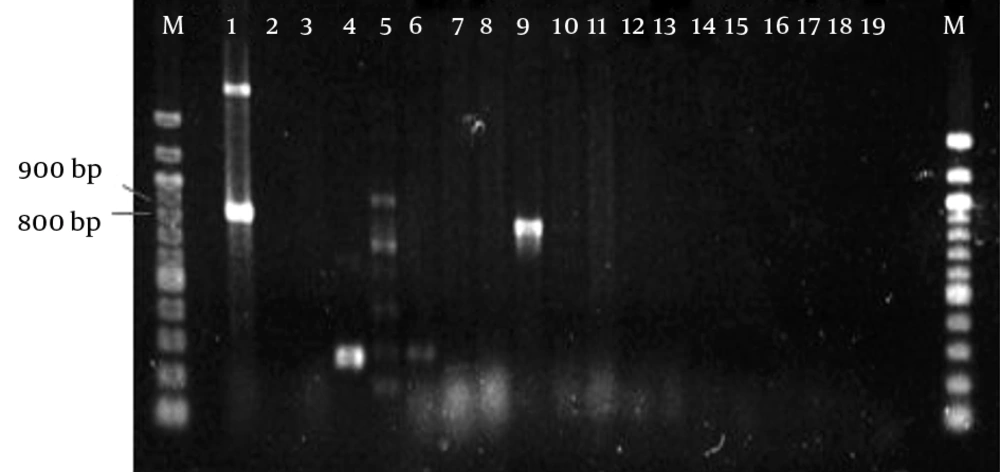
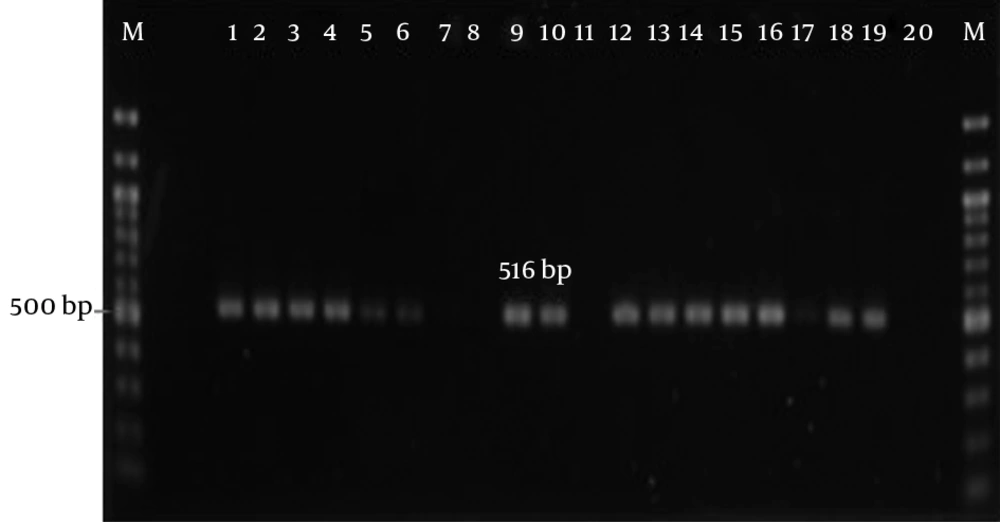
PCR was used to screen for the presence of resistant genes, such as blaOXA-1, blaOXA-10, plasmid-mediated AmpC (blaCMY and blaDHA), chromosomally-mediated blaAMPC, Sul1, blaTEM, and blaSHV. In order to detect these resistant genes, gene-specific forward and reverse primers (IDT, Singapore) were used to carry out the PCR reaction (Table 1). After the amplification process, the PCR products were subjected to 1.5% gel electrophoresis.
| Primer | Sequence | Expected Size (bp) | Source |
|---|---|---|---|
| 16S rDNA | F: AGA GTT TGA TCC TGG CTC AG | 1492 | (19) |
| R: GGT TAC CTT GTT ACG ACT T | |||
| blaCMY | F: ATGATGAAAAAATCGTTATGC | 1200 | (20) |
| R: TTGCAGCTTTTCAAGAATGCGC | |||
| blaDHA | F: TGATGGCACAGCAGGATATTC | 997 | (21) |
| R: GCTTTGACTCTTTCGGTATTCG | |||
| Chromosome-mediated AmpC | F: TGCTATTTCAAAGGAACCTTCA | 147 | (22) |
| R: TTAATGCGCTCTTCATTTGG | |||
| blaOXA-1 | F: GTGCGTCAACGGATATCTCT | 736 | (23) |
| R: GTGATCGCATTTTTCTTGGC | |||
| blaOXA-10 | F: GTCTTTCGAGTACGGCATTA | 720 | (24) |
| R: ATTTTCTTAGCGGCAACTTA | |||
| Sul1 | F: ATGGTGACGGTGTTCGGCAT | 840 | (25) |
| R: CTAGGCATGATCTAACCCTC | |||
| blaTEM | F: ATCAGCAATAAACCAGC | 516 | (26) |
| R: CCCCGAAGAACGTTTTC | |||
| blaSHV | F: TGGTTATGCGTTATATTCCCC | 868 | (16) |
| R: GGTTAGCGTTGCCAGTGCT |
4. Results
In this study, a total of 18 strains of bacteria resistant to amoxicillin-clavulanate were successfully isolated from a variety of retail sausages. Half of the isolates (D1, H3, J2, J6, J7, J8, K1, L1, and L2) were identified as members of Enterobacteriaceae by using an API 20E kit, whereas the remaining (C1, E1, E2, E3, H2, J1, J3, J4, J5) isolates were identified as non-Enterobacteriaceae Gram-negative bacteria. The most prominent Enterobacteriaceae isolated from the retail sausages were Klebsiella spp. (33.33%), followed by Enterobacter spp. (49.01%).
The antibiotic susceptibility and interpretation of bacteria isolates isolated from a variety of sausage brands towards the antibiotic agents are summarized in Table 2. Based on the results, all the isolated strains (100%) were resistant towards amoxicillin-clavulanate, penicillin, and oxacillin. Furthermore, the most common resistances were observed against cefotaxime (71.4%), cefpodoxime (66.7%), and ampicillin (83.3%), whereas the least frequent resistances were against piperacillin, imipenem, and meropenem (5.6%) as well as gentamicin (11.1%). Fifty percent of the isolates showed resistance to aztreonam, whereas 33.3% were resistant towards ceftriazone, and 16.7% were resistant to ceftazidime and tetracycline. None of the isolates showed resistance to norfloxacin. The results for the double disc synergy test and phenotypic confirmatory test are summarized in Table 3. In this study, only one (isolate E3) exhibited the keyhole phenomenon on MH agar after incubation and demonstrated a positive phenotypic result. The only ESBL producer in this study was identified to be Chryseobacterium meningosepticum, a non-Enterobacteriaceae bacteria.
| Sample in Assigned Code | Diameter of Zone of Inhibition (mm) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMC-30 | ATM-30 | CTX-30 | CPD-10 | CRO-30 | CAZ-30 | AMP-10 | P-1 | OX-1 | PRL-75 | IPM-10 | MEM-10 | CN-10 | NOR-10 | TE-30 | |
| C1 | R (0) | R (23) | R (19) | R (0) | R (21) | S (25) | R (0) | R (0) | R (0) | S (26) | S (34) | S (36) | S (20) | S (26) | R (11) |
| D1 | R (9) | R (26) | R (22) | R (0) | R (22) | S (24) | R (0) | R (0) | R (0) | S (21) | S (26) | S (30) | S (16) | S (23) | S (19) |
| E1 | R (0) | R (10) | R (21) | R (0) | R (22) | S (25) | R (9) | R (0) | R (0) | S (28) | S (34) | S (42) | S (25) | S (34) | S (20) |
| E2 | R (0) | R (0) | R (0) | R (0) | R (0) | R (0) | R (8) | R (0) | R (0) | R (0) | R (10) | R (14) | R (11) | S (19) | R (10) |
| E3 | R (0) | R (0) | R (21) | R (14) | S (26) | R (0) | R (0) | R (0) | R (0) | S (29) | S (32) | S (36) | S (30) | S (20) | S (34) |
| H2 | R (0) | R (0) | S (28) | R (17) | S (26) | S (25) | R (0) | R (0) | R (0) | S (25) | S (25) | S (31) | S (16) | S (26) | S (16) |
| H3 | R (0) | R (0) | S (27) | R (15) | S (25) | S (25) | R (0) | R (0) | R (0) | S (26) | S (25) | S (30) | S (15) | S (25) | S (15) |
| J1 | R (7) | S (30) | I (25) | I (18) | I (22) | S (22) | S (18) | R (0) | R (0) | S (28) | S (30) | S (34) | I (14) | S (32) | S (24) |
| J2 | R (6) | S (27) | R (22) | S (23) | S (28) | S (28) | I (14) | R (0) | R (0) | S (24) | S (26) | S (33) | S (20) | S (22) | S (18) |
| J3 | R (6) | S (23) | R (20) | R (0) | R (17) | S (23) | R (0) | R (0) | R (0) | S (25) | S (24) | S (32) | S (20) | S (28) | R (8) |
| J4 | R (0) | S (24) | R (21) | R (0) | I (20) | S (26) | R (0) | R (0) | R (0) | S (26) | S (27) | S (32) | S (21) | S (28) | I (13) |
| J5 | R (0) | R (0) | R (0) | R (0) | R (0) | R (0) | S (17) | R (0) | R (0) | S (24) | S (29) | S (30) | S (18) | S (28) | S (26) |
| J6 | R (6) | S (30) | S (26) | S (26) | S (26) | S (28) | R (0) | R (0) | R (0) | S (23) | S (28) | S (28) | S (17) | S (28) | S (26) |
| J7 | R (13) | S (32) | S (30) | S (27) | S (29) | S (27) | R (0) | R (0) | R (0) | S (23) | S (25) | S (22) | S (17) | S (24) | S (24) |
| J8 | R (0) | S (28) | I (25) | R (17) | S (23) | S (24) | R (0) | R (0) | R (0) | S (26) | S (23) | S (29) | R (12) | S (30) | S (20) |
| K1 | R (0) | R (0) | R (22) | R (14) | S (32) | S (34) | R (12) | R (0) | R (0) | S (25) | S (25) | S (29) | S (17) | S (30) | S (20) |
| L1 | R (13) | S (27) | S (29) | S (30) | S (26) | S (28) | R (7) | R (0) | R (0) | S (22) | S (28) | S (30) | S (16) | S (25) | S (23) |
| L2 | R (11) | S (25) | I (24) | I (19) | S (25) | S (24) | R (7) | R (0) | R (0) | S (24) | S (29) | S (30) | S (17) | S (26) | S (24) |
| No of resistant isolates | (18/18), 100% | (9/18), 50% | (10/18), 71.4% | (12/18), 66.7% | (6/18), 33.3% | (3/18), 16.7% | (15/18), 83.3% | (18/18), 100% | (18/18), 100% | (1/18), 5.6% | (1/18), 5.6% | (1/18), 5.6% | (2/18), 11.1% | (18/18), 0% | (3/18), 16.7% |
| Sample in Assigned Code | Double Disc Synergy Test | Phenotypic Confirmatory Test | Significant Taxa and % ID |
|---|---|---|---|
| C1 | Negative | Negative | Pseudomonas aeruginosa (76.4%) |
| D1 | Negative | Negative | Enterobacter cancerogenus (99.9%) |
| E1 | Negative | Negative | Pseudomonas luteola (98.8%) |
| E2 | Negative | Negative | Pasteurella pneumotropica (99.9%) |
| E3 | Positive | Positive | Chryseobacterium meningosepticum (87.5%) |
| H2 | Negative | Negative | Pantoea spp. 3 (92.2%) |
| H3 | Negative | Negative | Klebsiella oxytoca (97.4%) |
| J1 | Negative | Negative | Rahnella aquatilis (63.6%) |
| J2 | Negative | Negative | Enterobacter cloacae (95.1%) |
| J3 | Negative | Negative | Pseudomonas fluorescens / putida (75.5%) |
| J4 | Negative | Negative | Pseudomonas fluorescens / putida (75.5%) |
| J5 | Negative | Negative | Pseudomonas luteola (95.4%) |
| J6 | Negative | Negative | Klebsiella pneumoniae spp. pneumoniae (97.6%) |
| J7 | Negative | Negative | Klebsiella pneumoniae spp. pneumoniae (97.6%) |
| J8 | Negative | Negative | Enterobacter cloacae (95.1%) |
| K1 | Negative | Negative | Enterobacter cloacae (92%) |
| L1 | Negative | Negative | Klebsiella pneumoniae spp. pneumoniae (66.4%) |
| L2 | Negative | Negative | Klebsiella pneumoniae spp. pneumoniae (97.6%) |
All the profiles of the 18 isolated strains are summarized in Table 4, and the gel electrophoresis for the PCR amplifications are presented in Figure 1 through 3. In this study, none of the bacterial isolates carried any chromosome-mediated blaAMPC, blaCMY, blaDHA, blaOXA-1, or blaOXA-10 genes. Sul1 genes were detected in Pseudomonas aeruginosa (C1) and Enterobacter cloacae (J2), a member of Enterobacteriaceae. In total, a high prevalence of blaTEM was detected in 15 (83.3%) isolates out of 18 isolates that harbored blaTEM in the total DNA or plasmid DNA. For Enterobacteriaceae, all were screened as positive for the blaTEM gene, 14.3% were detected in chromosomal DNA, and the remaining 85.7% were plasmid-encoded. On the other hand, out of the nine non-Enterobacteriaceae, only P. fluorescens/putida (J4) carried chromosomal-encoded blaTEM, whereas the remaining (88.9%) contained plasmid-encoded blaTEM. The only ESBL-producer (E3) screened positive for the presence of plasmid-encoded blaTEM. Additionally, chromosomal-encoded SHV was detected in the four Klebsiella spp. J6, J7, L1, and L2. Although all of the Enterobacteriaceae harbored at least one β-lactamase gene (either blaSHV or blaTEM) in the total DNA or plasmid DNA, none of these was an ESBL-producer.
| Bacterial Strain | Total DNA | Plasmid DNA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| blaCMY | blaDHA | blaAMPC | blaOXA-1 | blaOXA-10 | Sul1 | blaTEM | blaSHV | blaCMY | blaDHA | blaTEM | blaSHV | |
| C1 (Pseudomonas aeruginosa) | – | – | – | – | – | + | + | – | – | – | + | – |
| D1 (Enterobacter cancerogenus) | – | – | – | – | – | – | – | – | – | – | + | – |
| E1 (Pseudomonas luteola) | – | – | – | – | – | – | + | – | – | – | + | – |
| E2 (Pasteurella pneumotropica) | – | – | – | – | – | – | + | – | – | – | + | – |
| E3 (Chryseobacterium meningosepticum) | – | – | – | – | – | – | + | – | – | – | + | – |
| H2 (Pantoea spp.) | – | – | – | – | – | – | + | – | – | – | + | – |
| H3 (Klebsiella oxytoca) | – | – | – | – | – | – | + | – | – | – | – | – |
| J1 (Rahnella aquatilis) | – | – | – | – | – | – | – | – | – | – | – | – |
| J2 (Enterobacter cloacae) | – | – | – | – | – | + | + | – | – | – | + | – |
| J3 (Pseudomonas fluorescens / putida) | – | – | – | – | – | – | + | – | – | – | + | – |
| J4 (Pseudomonas fluorescens / putida) | – | – | – | – | – | – | + | – | – | – | – | – |
| J5 (Pseudomonas luteola) | – | – | – | – | – | – | + | – | – | – | + | – |
| J6 (Klebsiella pneumoniae) | – | – | – | – | – | – | + | + | – | – | + | – |
| J7 (Klebsiella pneumoniae) | – | – | – | – | – | – | – | + | – | – | + | – |
| J8 (Enterobacter cloacae) | – | – | – | – | – | – | + | – | – | – | + | – |
| K1 (Enterobacter cloacae) | – | – | – | – | – | – | + | – | – | – | + | – |
| L1 (Klebsiella pneumoniae) | – | – | – | – | – | – | + | + | – | – | + | – |
| L2 (Klebsiella pneumoniae) | – | – | – | – | – | – | + | + | – | – | + | – |
5. Discussion
The present investigation showed multidrug resistance among all the isolates used in this study. In this study, nine (50%) members of Enterobacteriaceae were isolated, but none of these Enterobacteriaceae harbored ESBLs. This is surprising because there have been numerous reports of ESBL-producing Enterobacteriaceae in food sources, especially poultry products (27-29). It is also known that resistant Enterobacteriaceae (i.e., salmonellosis) in food can be transmitted within the community (30, 31). The only one ESBL-producing bacteria detected in this study was Chryseobacterium meningosepticum (E3), a non-Enterobacteriaceae that is known to cause meningitis and pneumonia in patients with compromised immune systems (32, 33). These bacteria are opportunistic human pathogens (33), and they have been isolated from raw and fresh milk (34); however, there have been no reports of their isolation from sausages or poultry products. Here, the isolates exhibited resistance to third generation cephalosporins, including cefotaxime, cefpodoxime, and ceftazidime. This could indicate an increased resistance to β-lactam antibiotics, causing reduction of the efficacy of antibiotic therapy.
As indicated in Table 4, nine of the isolates identified as Enterobacteriaceae did carry either blaSHV or blaTEM genes, but not blaAMPC or blaOXA. blaTEM has been shown to be the most frequently detected amoxicillin-clavulanate resistant Enterobacteriaceae (35). This is further supported by other studies showing that resistance to amoxicillin-clavulanate in E. coli isolates from this area was mainly associated with presumptive overproduction of TEM-1, TEM-2, or SHV-1 β-lactamases or of AmpC cephalosporinase (36-38). However, none of our isolates carried blaAMPC. The carriage of blaSHV was usually found on the chromosomal DNA of K. pneumoniae isolates in our study. This can be supported by studies reporting that genes encoding SHV-1 were naturally found in K. pneumoniae chromosomes (4, 39). Therefore, blaSHV detection in this species is expected and predicted.
Most isolates showed negative results in a phenotypic confirmatory test for ESBL, even though the isolates carried blaTEM and blaSHV genes. This could be due to the hyperproduction of either TEM or SHV β-lactamases in ESBLs, which has been previously reported to cause a false negative phenotypic confirmatory test (40). Moreover, certain ESBL-producers could contain other β-lactamases, including inhibitor-resistant TEMs (IRT), which can mask the production of ESBL in the phenotypic test, leading to a false-negative result (4, 41). Although the inhibitor-resistant TEM variants show clinical resistance to amoxicillin-clavulanate, they remain susceptible to inhibition by tazobactam and subsequently to the combination of piperacillin and tazobactam (4). To confirm whether the isolates are of the ESBL or IRT phenotype, further tests with these antibiotic combinations may be performed in the future.
It is interesting to note that P. aeruginosa (C1) and E. cloacae (J2) co-harbored Sul1 and blaTEM genes in this study. This observation was in agreement with the previous studies, whereby the Sul1 gene was highly prevalent in relation with integron 1 in P. aeruginosa (18, 42). The dissemination of Sul1 may be a result of the horizontal transfer of antibiotic resistance gene cassettes (42). A study in Jordan showed that 53.9% of urinary E. coli isolates were positive for one or more of the Sul genes, and significant resistance was observed towards amoxicillin-clavulanate (9). Interestingly, amoxicillin-clavulanate resistance was also correlated with the presence of Sul in Salmonella enterica isolates in Spain (43). However, all these studies were performed using isolates obtained from humans and not food sources, suggesting that the results obtained in this study are novel.
In conclusion, the fact that amoxicillin-clavulanate resistant isolates were isolated from retail sausages demonstrates the risk of transmission to humans via these sausages if they are not handled or cooked properly prior to consumption. Thus, good practices in hygiene procedures and food preparation are necessary to control and reduce biological risks.