1. Introduction
Brain abscess is a rare but life-threatening complication, described as focal suppurative infections (1). It begins with colonization of microorganisms into the brain parenchyma and develops to a well-vascularized, ring-enhancing capsule form surrounded with collection of pus (2). In developed countries, the incidence rate among all cranial lesions is about 1% - 2%, whereas in developing countries it increases up to 8%. The mortality rate associated with brain abscess have had a significant decrease in last years (0% - 24%) due to improved surgical techniques, effective antimicrobial treatments, and contemporary diagnosis methods (3). It is usually observed that the spread of the infection causesbrain abscess complications, e.g. maxillary sinusitis and tooth inflammations, middle ear infections and meningitis, which appear after trauma and surgical interventions (4, 5).
Prevotella is an obligate anaerobic pigmented and non-pigmented bacterium (6). Prevotella denticola is a pigmented bacterium of oral microbiota. As many anaerobic bacteria, P. denticola are natural members of the flora under appropriate conditions. Otherwise, they might cause periodontal diseases, severe septicemia, osteomyelitis, and deep tissue abscess.
Gram staining is crucial for the Prevotella spp. identification. The presence of polymorphonuclear leucocytes mixed with the bacteria in morphotype suggests anaerobic infection. Aerotolerance tests can reveal anaerobic nature of the bacteria and subsequently, different advanced diagnostic procedures might be performed, including automated systems based on biochemical tests, and matrix-assisted laser desorption ionization-time of flight (MALDI-TOF). DNA sequencing method is the gold-standard. The treatment of Prevotella spp. varies from β-lactamase to a combination of β-lactam-β-lactamase inhibitors, and carbapenems for more severe infections (7). The present study reports a brain abscess after chronic otitis media infection due to P. denticola, which is a rare cause of brain abscess.
2. Case Presentation
A 27-year-old male diagnosed with otitis media presenting purulent drainage from the left external ear tract underwent treatment with IV antibiotics at an external medical center. The brain imaging was done for otitis media and since abscess in the temporal lobe appeared, the patient was dispatched to our hospital and hospitalized for examination and follow-up. The patient history showed a yellow-green ear drainage that was intense enough to create stains on the pillow; the drainage stopped after a few days and restarted again. He had headache for 1 month and its severity had increased over time. Ten day before admission to our hospital, he had been hospitalized with the diagnosis of otitis media and took IV antibiotic treatment (ceftriaxone). Following the identification of temporal abscess at the external center, the patient rejected the treatment and came to our hospital.
At the time of admission, he was 37.4°C with stable vital parameters, presenting purulent drainage in his left external auditory canal, and granulation tissue in the external auditory canal. The evaluation of the left ear membrane was not possible due to the polypoid tissue covering it. He had facial paralysis in left and neck rigidity. Kernig’s and Brudzinski’s symptoms were negative. Treatment with vancomycin and meropenem was applied empirically. In the brain imaging, at the time of emergency admission, some findings which were concordant with abscess were identified.
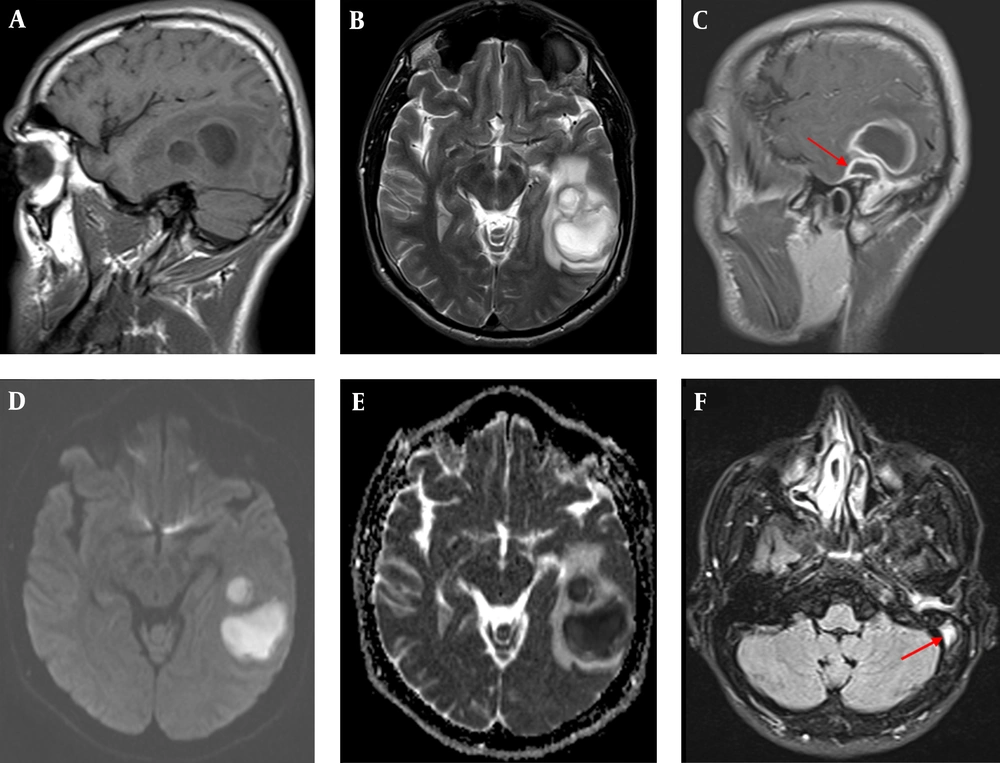
Magnetic resonance imaging of brain, sagittal T1W, and axial T2W images showed T1-hypointense and T2-hyperintense cystic lesions with peripheral vasogenic edema in the left temporal lobe. Postcontrast sagittal T1W image revealed ring-like thick contrast enhancement in the lesions, as well as, demonstrated the epidural abscess adjecent to parenchymal lesions. The lesions had high DWI signal and low signal intensity on ADC map representing diffusion restriction. Axial FLAIR image showed luminal hyperintensity within the left sigmoid sinus suggesting sinus thrombosis (Figure 1). On day 2 of hospitalization, mastoidectomy and temporal abscess drainage were performed.
Contrast-Enhanced Cranial Magnetic Resonance Imaging. (A) the Sagittal T1-Weighted Image Showing Two Adjacent Circular Hypo-Intense Lesions and Their Mild T1 Hyper-Intense Walls At Left Temporal Lobe; (B) the Axial T2A Image Indicating the Walls of the Lesions Are Mildly Hypotensive and Surrounded by Vasogenic Edema; (C) Contrast-Enhanced Sagittal T1A Image Showing Smooth Thick Contrast at the Walls of the Lesions Which Is Accompanied By Epidural Abscess (Arrow) in the Floor of the Left Temporal Lobe, Adjacent to Intra-Parenchymal Lesions; (D,E) Diffusion-Weight Imaging Demonstrating Limitation of Homogenous Diffusion, Typical for Abscess, at the Central to the Left Temporal Lesions; (F) the Axial FLAIR Image Monitoring the Intraluminal Hyper Intensity of the Left Sigmoidal Sinuses, on Behalf of Thrombosis.
Gram-negative coccobacillus were observed at the microbial investigation of abscess material. The abscess material was inoculated to the “eosin methylene blue” (EMB) agar and chocolate agar and incubated at 37°C for 48 hours to identify aerobic and anaerobic bacteria. Aerobic incubation resulted negative but the anaerobic incubation to the Schaedler Agar and thioglycollate broth at 37°C for 48 hours resulted dark pigmentated colonies on Schaedler agar, representing small and faintly stained gram-negative coccobacillus. In the analysis using half-automated system BBL crystal (Becton, Dickinson and company U.S.A) Prevotella spp. were identified. Thereupon 16S analysis revealed P. denticola causing the brain abscess. At the control MRG after drainage, two intact abscesses were observed but one of them was already drained. During the second week of the treatment, the patient got better in terms of neurological symptoms. Since monoplegia had been developed in the patient’s right lower extremity, brain imaging was repeated; however, no acute pathology was observed.
During follow-up, except left facial paralysis, neurological symptoms recovered within three days. The recovery of facial paralysis was determined on 5th day of follow-up. New MRG results showed temporal epidural hematoma and therefore, the patient was submitted to a new surgery for hematoma drainage. No growth was detected in the drainage culture of 10cc necrotic material. Thrombosis was detected on MRG images at left transverse and sigmoidal sinuses on the following days after hematoma drainage. Enoxaparin was used for anticoagulation and continued as empirical treatment for six weeks. At the time of discharge, the patient was healthy.
3. Discussion
Prevotella spp. are considered as normal flora of the oral cavity; but in case of low oxygen concentration, trauma, pyogenic infections, etc., they can cause severe inflammation especially when accompanied by predisposing factors such as pulmonary abscess, bronchiectasis, empyema, otitis media, or mastoiditis. Prevotella spp. may also cause brain abscess (8). Bacteria in oral cavity have more tolerance to oxygen in nature compared to other bacteria; this is a virulence factor for them.
The determinants of brain abscess differ with the time, geographical distribution, drug usage, surgical intervention, and infection type. Different bacteria, fungi, and parasites may cause brain abscess. Streptococcus spp. and Staphylococcus aureus have been frequently isolated from bacterial communities. Enteric gram negative bacillus (Proteus spp., Escherichia coli, Klebsiella spp., Morganella Morganii, Pseudomonas spp.) are generally isolated as a part of mixed cultures in the case of otitis media infection or suppressed immune system in the ratio of 23% - 33% (9).
Several studies have reported that the growth ratio of anaerobic bacteria is nearly 25-30% in the case of brain abscess, and Bacteroides and Prevotella species were the most frequently isolated bacteria (10, 11). The cases in which anaerobic bacteria are accounted for the development of brain abscess generally occur after middle ear and mastoid infection (12, 13). In a study by Menon et al., 75 brain abscess cases were reviewed in a 5-year period, and it was determined that chronic otitis media is the most frequent risk factor. In two of these cases, P. denticola was detected (14). In the present report, a long-term ear infection was present as well. In a study, two cases of brain abscess were reported from our country that one of them was associated with anaerobic agents and presented with middle ear and mastoid infections, whereas the other one was atrributed to head trauma followed by meningitis (15).
The recognized treatment method of brain abscess is ponction and aspiration of abscess followed by medical treatment towards the active microorganism agent (16). After surgical intervention, patient should be treated for 6 - 8 weeks, according to the agent determined in microbial investigations. Thrombosis of septic transverse sinus is always observed as a suppurative complication of otitis media and mastoiditis. As shown, Proteus spp., E.coli, S. aureus, and anaerobes are agents that cause transverse sinus thrombosis (17, 18). Most of thepatients may develop facial paralysis and intracranial pressure symptoms (19). In case of sagittal sinus thrombosis, fluctuation in mental level, motor changes, papillary stasis, and neck stiffness may be observed and they can become severe enough to threaten the life. Some coagulation defects such as lack of clotting factors might be observed, although they are general complications in the course of bacterial meningitis (20). Anticoagulant treatment for the septic sinus thrombosis is contradictive. In this study, the patient developed sinus thrombosis despite of active antibiotic treatment application. Even though it was not possible to detect any underlying coagulation defect, anticoagulant treatment was initiated due to the development of thrombosis during the antimicrobial treatment process.
In this way, considering irresponsiveness to the previous antibiotic treatment before admission to our hospital, a broad-spectrum antibiotic treatment was applied and even though the patient underwent surgical drainage in a short span of time, intracranial septic thrombophlebitis was developed during the follow-up. Consequently, development of severe complications such as brain abscess and septic intracranial thrombophlebitis should be considered after long-term otitis media infection. The main way to manage the chronic otitis media is early diagnosis and surgical intervention. Microbial pathogens facilitate the invasion and consequently complications. In addition, given the fact that anaerobic bacteria are responsible for etiology of brain abscess, appropriate diagnosis and treatment planning are required. For the prompt diagnosis of the brain abscess due to anaerobic agents, a multidisciplinary approach including neurosurgery, neurology, and microbiology disciplines is necessary.