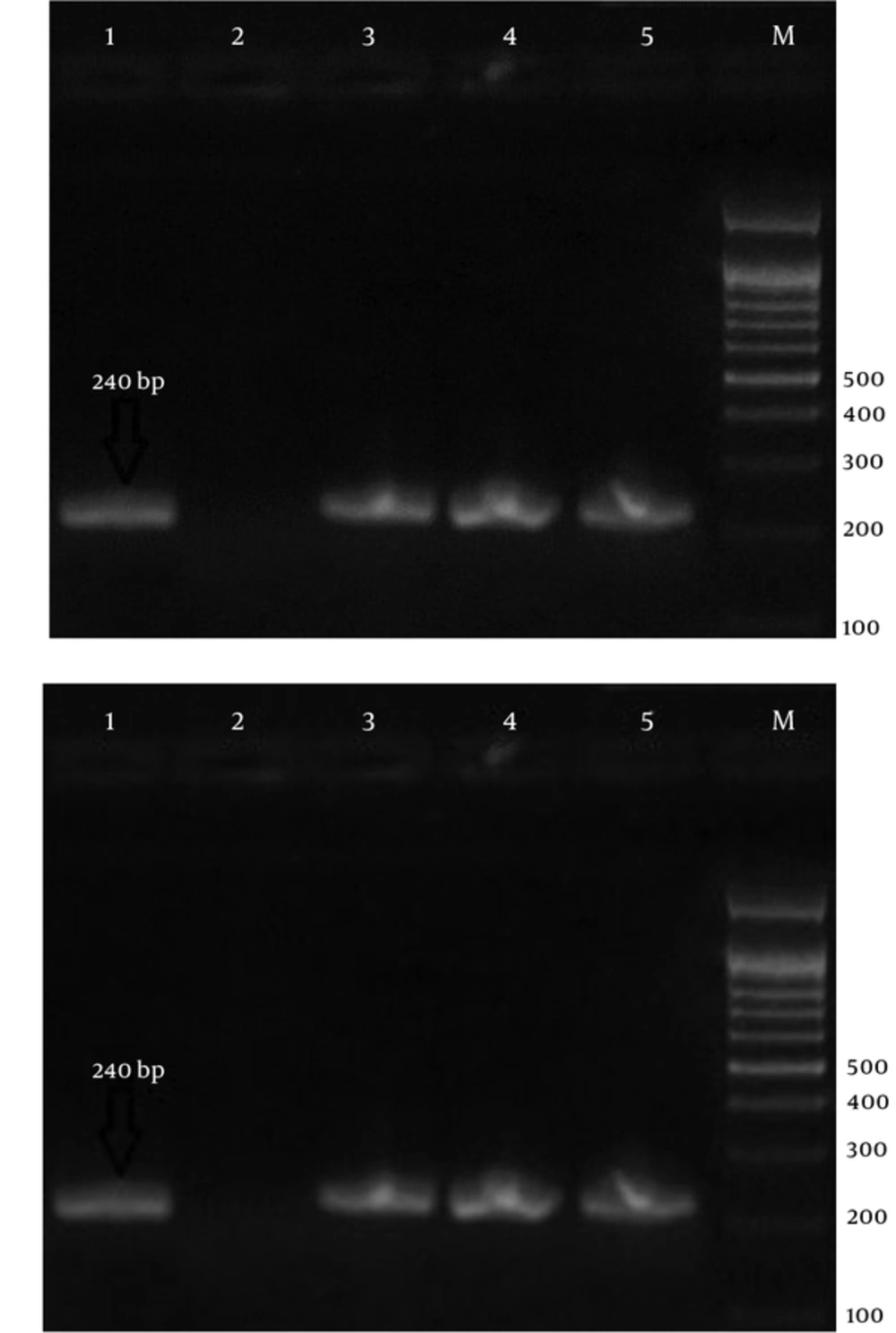
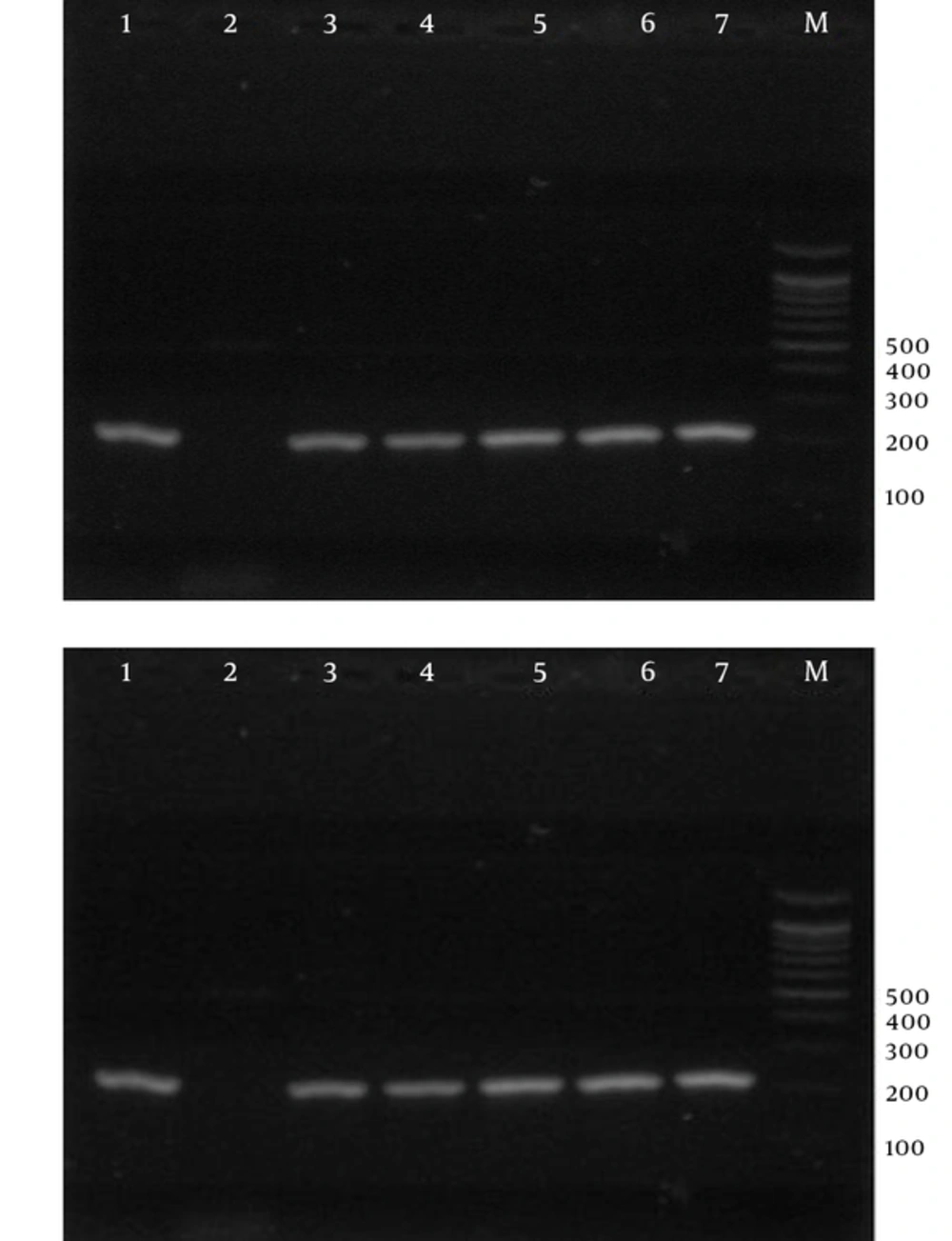
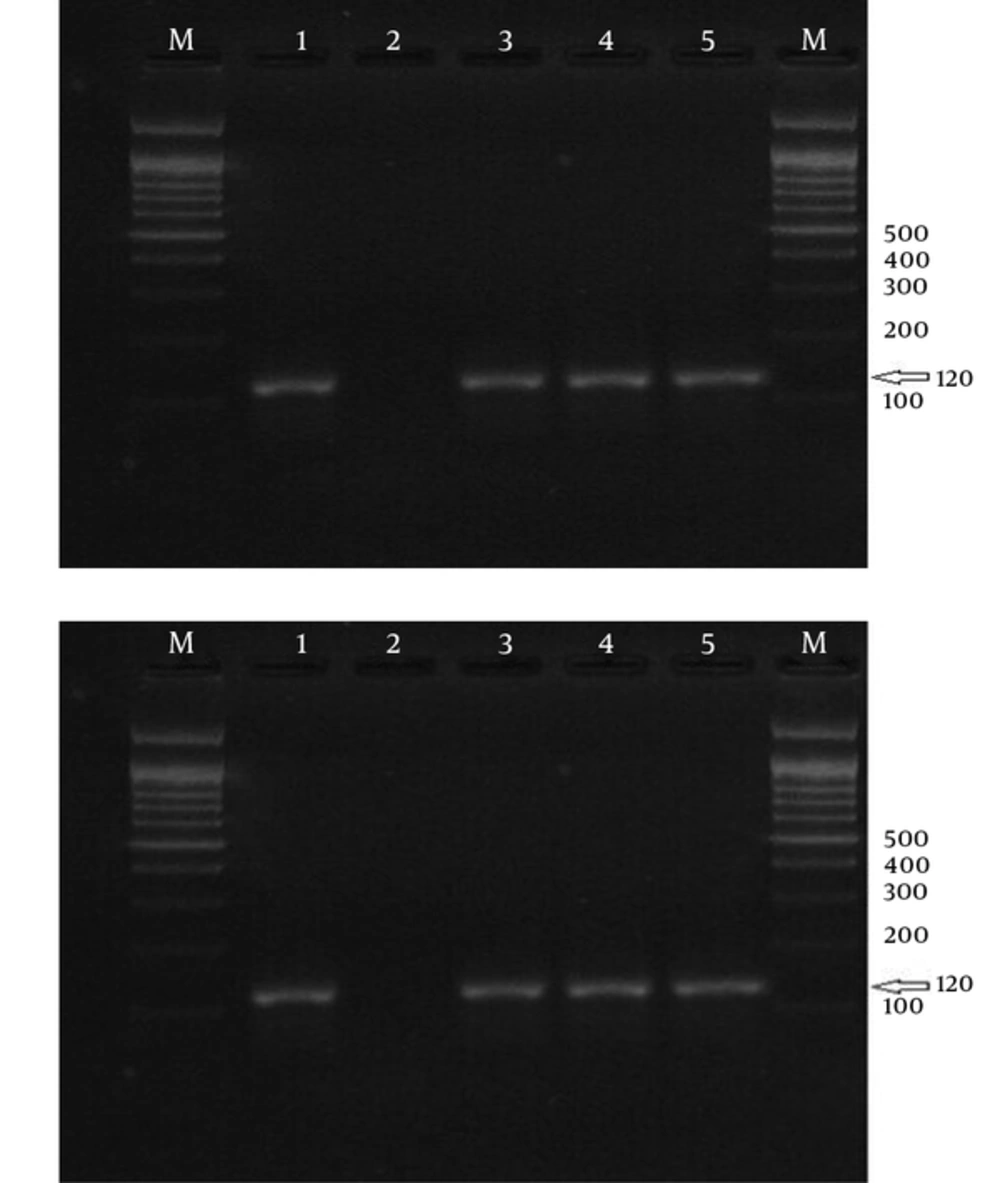
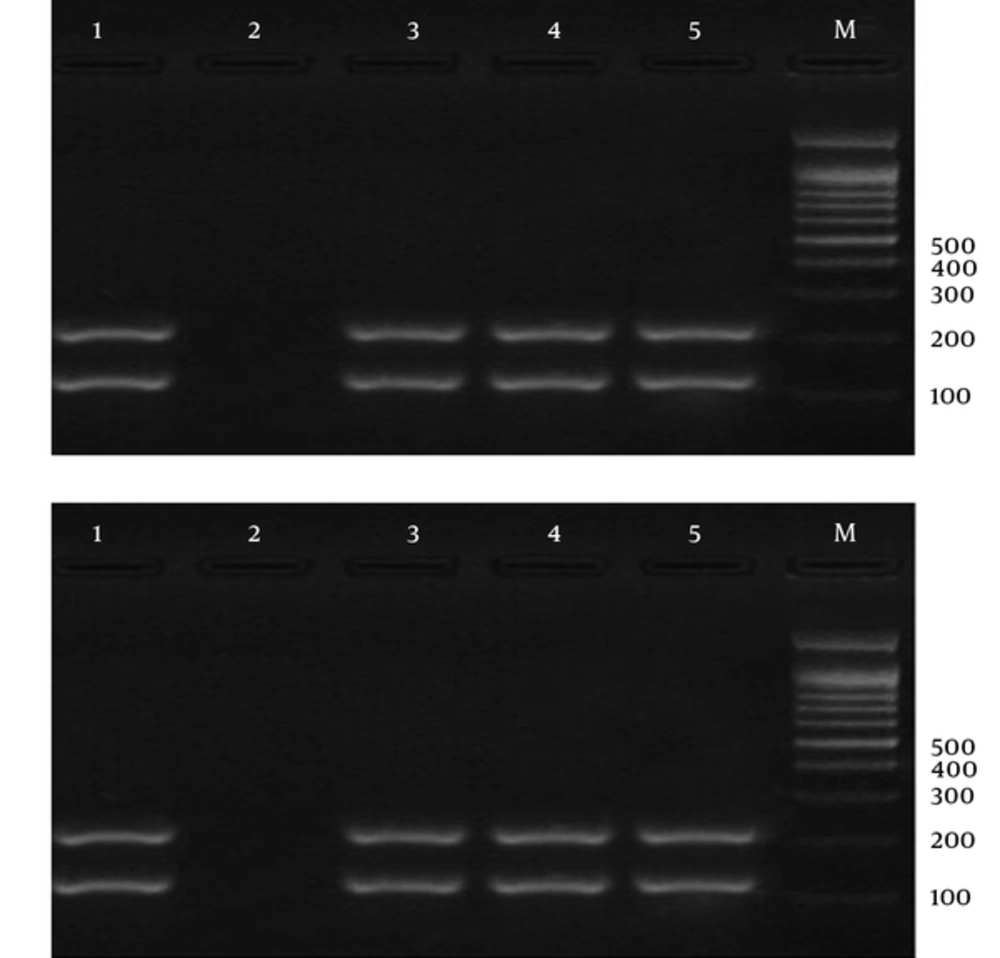
Laboratory diagnosis of malaria is currently performed to detect parasites by the light microscopy of Geimsa-stained thick and thin blood smears. This procedure is cheap and simple, but is a labor-intensive procedure, which requires well-trained personnel (
17). When parasitemia is very low, the data of microscopy diagnosis are limited, and in some cases biased by the inability to devote the necessary amount of time to the examination of blood smears (
17).
Greater sensitivity and specificity of PCR method in comparison with the thick blood films examination method are shown by many studies. The detection of low
P. vivax and
P. falciparum parasitemia by PCR, at levels undetectable by microscopy, was reported earlier (
14). The current study on microscopy and nested-PCR assay showed that the results obtained by PCR were equivalent or superior to those of the microscopy, in which all microscopy-positive samples were positive by PCR. In addition, the PCR test was able to detect mix infections missed by microscopy. This may be due to the tendency of 1 species to be dominant over other ones (
14,
18).
Given the progresses achieved over 5 decades of working on malaria eradication and also significant reduction in the incidence of the disease in the recent 10 years, complete elimination of malaria in the country is the main agenda of the joint direction of the Iranian ministry of health and medical education and WHO. Nowadays, the local distribution and transmission of malaria in Iran is limited to 28 cities, located in Sistan and Baluchistan, Hormozgan, and Kerman provinces. The ultimate goal of malaria elimination program in 2025 horizon is interrupting local transmission of the disease in Iran (
19-
23).
Malaria elimination is defined as halting its transmission by anopheles mosquitoes in a specific geographical area to reach 0 incidence of the disease. Malaria eradication is defined as continuous and persistent reduction in the global incidence of malaria until reaching the rate of zero (
24). According to WHO reports, malaria is on the verge of elimination in Iran (
25). Iran reported only 330 cases of malaria in 2015, which is about 50% reduction compared with 2014 (
26). Malaria is a major health problem in the Southeastern regions of Iran and greatly inhibits the socioeconomic development of the region. In the current study, nested-PCR molecular technique was used, which has high sensitivity; it can detect mixed infectious agents as well as parasite species. Many studies showed the high sensitivity and specificity of PCR, compared with those of the microscopic methods (
27-
31).
Zakeri et al. investigated the malaria infection prevalence in the Southeastern areas of Iran using nested-PCR method. They tested 120 samples targeting ssrRNA gene using molecular methods. They identified
P. vivax in 59%,
P. falciparum in 10%, and a mix infection of
P. vivax and
P. falciparum in 28.4% of the samples (
15). Ebrahimzadeh et al. studied malaria strains by molecular methods. They analyzed 140 samples, 94.3% of which were positive; 51.4% were
P. vivax; 12.6%
P. falciparum and 29.3% were a mixed infection of
P. vivax and
P. falciparum (
32).
Plasmodium malaria was also reported (
33).
Zakeri et al. investigated the sensitivity of nested-PCR in the diagnosis of a mixed infection of
P. falciparum and
P. vivax in Pakistan, Iran, and Afghanistan. They studied 100
P. vivax-positive samples diagnosed by microscopic methods in Iran and mix-infections in 22% of the samples (
34). Different studies in Iran reported a reduction in malaria incidence in recent years (
35-
39). The current research investigated malaria species via molecular methods; the results revealed that out of 100 malaria-suspected samples, 84% were negative and 16% positive. The positive samples included 8%
P. vivax, 2%
P. falciparum, and 6% of positive samples were a mix-infection of
P. falciparum and
P. vivax. However,
P. vivax was the dominant species in the current study, which was consistent with previous studies. No
P. ovale or
P. malaria species was observed; as found by Ebrahimzadeh et al. (
40).
Microscopic methods are the golden standards for the simple and efficient identification of malaria, which conduct routinely in medical laboratories; but there is potential for error to identify parasite type and false negative reports in low-rate parasitemia (asymptomatic malaria). Moreover, in microscopic methods, there is no possibility to identify mixed plasmodium infection or diagnose the disease after drug consumption due to low parasite count. Furthermore, the presence of mixed-infection is an important factor for disease severity; the presence of P. vivax can have suppressing effects on P. falciparum infection, if it remains undiagnosed. On the other hand, considering the necessity of special treatment for P. falciparum infection, mixed infection including this species can lead to increase in P. falciparum infected cases that increases the number of parasite carriers in the population, and therefore, increases the incidence of the disease.
5.1.Conclusion
Correct diagnosis of malaria species can help proper treatment of the disease and reduce carriers in the general population, and accordingly, reduce the risk of malaria infection. According to the goal of malaria control program to minimize mortality rate from malaria and decrease the prevalence of the disease, a reliable laboratory test is necessary for the early diagnosis of the disease. The current study findings suggested that PCR diagnostic method was very helpful in the areas where the mixed infection of P. falciparum and P. vivax is common. It can contribute to precise determination of incidence rate of the disease and is also useful in follow-up endeavors, as a very useful supplement for microscopic diagnosis of malaria. Unfortunately, there is no facility or procurement to perform PCR tests in rural areas; therefore, every 5 years epidemiological molecular studies can narrow this gap. Moreover, considering the previous studies and the current study findings, it is apparent that malaria had a declining trend in recent years in Baluchistan. Thus, implementation of malaria elimination program is feasible and attainable in the province.