1. Background
The genus Penicillium comprises a large number of ubiquitous filamentous fungi, of which some are involved in human infections, ranging from mild to severe infections, especially in patients infected with human immunodeficiency virus (HIV), patients with underlying diseases, and intravenous drug abusers (1). Moreover, they lead to many complications, such as mycotoxicosis, allergies, and fungal sinusitis, and are frequently isolated from soil, dead plant materials, rotten wood, decaying vegetables, and foods (2). Based on phenotypic data, the genus of Penicillium is regarded to be related to ascomycota, because of its sexual reproduction by means of ascospores (3, 4). Members of the genus are of commercial and industrial importance due to their use in production of antibiotics, anti-tumoral, anti-fungal, anti-insect, and anti-viral compounds, as well as extracellular enzymes (2).
To date, it has been revealed that various sections of Penicillium encompass more than 250 species (5). The most common species of Penicillium, P. chrysogenum, is found to be an agent of onychomycosis, keratomycosis, allergic bronchopulmonary mycosis, and asthma, which may be associated with indoor environments, deserts, dried foods, cheese, and is morphologically characterized by biverticillate, terverticillate, or quarter-verticillate conidiophores, floccose to velutinous in colony texture, and yellow exudate droplets (2, 6-8). However, these conventional methods were noted to be tedious, time consuming and unreliable for species delineation (2, 9, 10). Nevertheless, molecular-based methods, such as DNA sequencing, have provided powerful tools for precise identification of the Penicillium species.
Currently, species identification in this genus is based on polyphasic criteria consisting of morphological and biochemical traits and molecular data from internally transcribed spacer (ITS) regions of ribosomal DNA (rDNA) and the partial β-tubulin (BT2) sequences (2, 9, 11). While the ITS-rDNA regions have some limitations, such as less resolution in differentiation of closely related species, due to a better species-specific resolution provided by the partial BT2 sequencing, application of this locus has substantially increased and is now an excellent marker for differentiation of Penicillium spp (2, 9, 12).
2. Objectives
Given that there was no comprehensive evaluation about the biodiversity of this genus in Iran, this study was focused on determining the distribution profile of Penicillium spp. in two provinces of Iran (Mazandaran and Tehran).
3. Materials and Methods
3.1. Fungal Strains
In total, 400 isolates used in this study were obtained from soil, air, cereals, and decaying vegetables from different regions of Mazandaran and Tehran provinces (Table 1). Stock cultures were maintained on slants of 2% malt extract agar (Difco, Detroit, MI, USA) and incubated at 25°C for one week.
| Species Identification by Phenotypic Criteria | Number of Isolates | Source | Cultural Characteristics | Species Identification by Sequencing of B-Tubulin | Accession Number |
|---|---|---|---|---|---|
| P. chrysogenum | 6 | Soil, fruit, air | Floccose | P. chrysogenum | KP851943, KT285850, KT285859, KT285861, KT285865, KT285866 |
| P. polonicum | 3 | Wheat | Velutinous | P. polonicum | KT285848, KT285856, KT285864 |
| P. glabrum | 2 | Plant | Velutinous | P. glabrum | KT285852, KT285854 |
| P. palitans | 2 | Bread | Velutinous | P. palitans | KT285849, KT285851 |
| P. melanoconidium | 2 | Wheat | Velutinous | P. melanoconidium | KT285858, KT285860 |
| P. expansum | 1 | Fruit | Floccose | P. expansum | KT285855 |
| P. raistrickii | 1 | Soil | Velutinous | P. raistrickii | KT285857 |
| P. italicum | 1 | Fruit | Velutinous | P. italicum | KT285853 |
| P. griseofulvum | 1 | Soil | Velutinous | P. griseofulvum | KT285863 |
| P. canescens | 1 | Soil | Velutinous | P. canescens | KT285862 |
| Total | 20 |
3.2. Morphological Identification and Cultural Characterization
Strains were cultured on 2% malt extract agar (Difco, Detroit, MI, USA), Czapek yeast extract agar (Difco, Detroit, MI, USA), and yeast extract agar (Himedia, India) and incubated at 24°C in the dark for one week. Identification was performed primarily based on macroscopic and microscopic morphology. Microscopic observations were based on slide culture techniques using potato dextrose agar (PDA) (5). Mounts of four-day-old slide cultures were made in lactic acid or lactophenol cotton blue, and light micrographs were taken using a Nikon Eclipse 80i microscope (Nikon, Tokyo, Japan) equipped with a Nikon digital sight DS-Fi1 camera. Moreover, strains were evaluated based on phenotypic characters, i.e. color, texture, growth rate, pigmentation, conidiophores, conidia, metulae, and phialide morphology (2).
3.3. Molecular Characterization
The fungal mycelia were grown on 2% MEA plates for four days at 24°C. Subsequently, a sterile blade was used to scrape off the mycelium from the surface of the plate. Genomic DNA was extracted using an ultra clean microbial DNA Isolation Kit (MoBio Inc. Solana Beach, CA, USA), according to the manufacturer’s instructions. DNA extracts were stored at −20°C prior to use.
The BT2 gene was partially amplified and sequenced using the universal fungal primers as follows: Bt2a (5’-GGTAACCAAATCGGTGCTGCTTTC-3’) and Bt2b (5’ACCCTCAGTGTAGTGACCCTTGGC-3’) (13, 14). PCR reactions were performed on a Corbett research thermal cycler, model CG1-96 (Sydney, Australia) in 50 μL volume containing 25 ng of template DNA, 5 μl reaction buffer (0.1 M Tris-HCl, pH 8.0, 0.5 M KCl, 15 mM MgCl2, 0.1% gelatine, 1% Triton X-100), 0.2 mM of each dNTP, and 2.0 units Taq DNA polymerase (ITK diagnostics, Leiden, The Netherlands). Amplification of BT2 was performed with one cycle of five minutes at 95°C for primary denaturation, followed by 35 cycles of 95°C for 45 seconds, 60°C for 120 seconds, and 72°C for 60 seconds, with a final extension of 72°C for seven minutes. Amplicons were purified using GFX PCR DNA and a gel band purification kit (GE healthcare, Buckinghamshire, UK). PCR-sequencing was performed as follows: 95°C for one minute, followed by 30 cycles consisting of 95°C for 10 seconds, 50°C for five seconds and 60°C for two minutes. Reactions were purified with Sephadex G-50 fine columns (GE healthcare bio-sciences AB, Uppsala, Sweden), and PCR products were sent to Bioneer company (Bioneer, Daejeon, South Korea) for bidirectional sequencing with the primers mentioned previously.
3.4. Alignment and Phylogenetic Reconstruction
Sequence data obtained in this study were imported to MEGA software version 5 (http://www.megasoftware.net) and adjusted using ClustalW. Ambiguous regions were excluded from the alignment, and then each trimmed sequence was exported to a BLAST search of the NCBI databases for species recognition. They were compared with Penicillium sequences, and query sequences with high similarity (≥ 99%) were considered as the same species.
The program, RAxML-VI-HPC v. 7.0.0, as implemented on the Cipres portal v. 1.10, was used for the tree search and the bootstrap analysis (GTRMIX model of molecular evolution and 500 bootstrap replicates). Bootstrap values equal to or greater than 70% were considered significant. Phylogenetic analyses were carried out based on the BT2 sequence in order to assess the phylogenetic placement of diverse Penicillium species, along with their relative sequences downloaded from GenBank, based on the Barreto et al. design (9). The phylogenetic tree was edited using Tree View v. 1.6.6. Paecilomyces variotii was used as the out-group.
4. Results
4.1. Morphological Identification and Cultural Characterization
All 400 Penicillium isolates were screened for phenotypic characters, and 20 strains representative of 10 morphological species, including P. chrysogenum, P. polonicum, P. canescens, P. griseofulvum, P. italicum, P. raistrickii, P. expansum, P. melanoconidium, P.palitans, and P. glabrum, were identified and selected for molecular analysis. In Figure 1A - J, the colony characteristics of 20 representative isolates on CYA and YES after seven days at 25°C were exemplified.
Some species, such as P. chrysogenum and P. italicum, due to production of typical yellow and red exudates on CYA and YES, respectively, could easily be identified, while for others it was essential to apply three complementary specific media (MEA, YES, and CYA ) for exact identification (Figure 1A - G). Except for P. chrysogenum and P. expansum with floccose colony texture, others were velutinous (Table 1 and Figure 1). Upon microscopic examination, nearly all identified species were one- or two-staged branched (biverticillate, terverticillate), except for P. glabrum, which typically produces no branching in conidiophore (monoverticillate) (Table 2).
| Species Identification by Phenotypic Criteria | Conidia | Phialide | Conidiophore Branching Patterna | Metulae |
|---|---|---|---|---|
| P. polonicum | Smooth,globose, 3 μm | Cylindrical, 10 μm | Terverticillate, Biverticillate | Cylindrical, 10 μm |
| P. glabrum | Smooth, subglobose to elliptical, 3 μm | Cylindrical, 10 μm | Monoverticillate | not present |
| P. expansum | Smooth,ellipsoidal, 3 μm | Cylindrical, 8 μm | Terverticillate | Cylindrical, 11 μm |
| P. raistrickii | Smooth,globose, 2.5 μm | Cylindrical, 7-9 μm | Biverticillate | Cylindrical, 10-12 µm |
| P. italicum | Smooth, ellipsoidal, 3.5 μm | Cylindrical, 8-10 μm | Terverticillate | Cylindrical, 14 μm |
| P. melanoconidium | Smooth,globose, 3.5 μm | Cylindrical, 10-14 μm | Terverticillate | Cylindrical, 12 μm |
| P. griseofulvum | Smooth, ellipsoidal, 2 μm | Cylindrical, 4-5 μm | Terverticillate | Cylindrical, 9 μm |
| P. chrysogenum | Smooth, globose, 3 μm | Cylindrical, 7 μm | Bi, ter and quarterverticillate | Cylindrical, 10 μm |
| P. canescens | finely roughened, globose, 2 µm | Cylindrical, 8 µm | Biverticillate, Monoverticillate | Cylindrical, 10-16µm |
| P. palitans | Smooth, globose, 3 - 4 μm. | Cylindrical, 10 μm | Terverticillate | Cylindrical, 10-14 μm |
aDefinitions: Monoverticillate, conidiophore without branching; Biverticillate, conidiophore one-stage branching; Terverticillate, conidiophore two-stage branch.
The species isolated in this study had globose, smooth-wall conidia, cylindrical phialide, and metula in various sizes. Interestingly, the only observed finely roughened conidium was related to P. canescens, which had been isolated from soil.
4.2. Results of BT2 Amplification, Sequencing, and Phylogeny
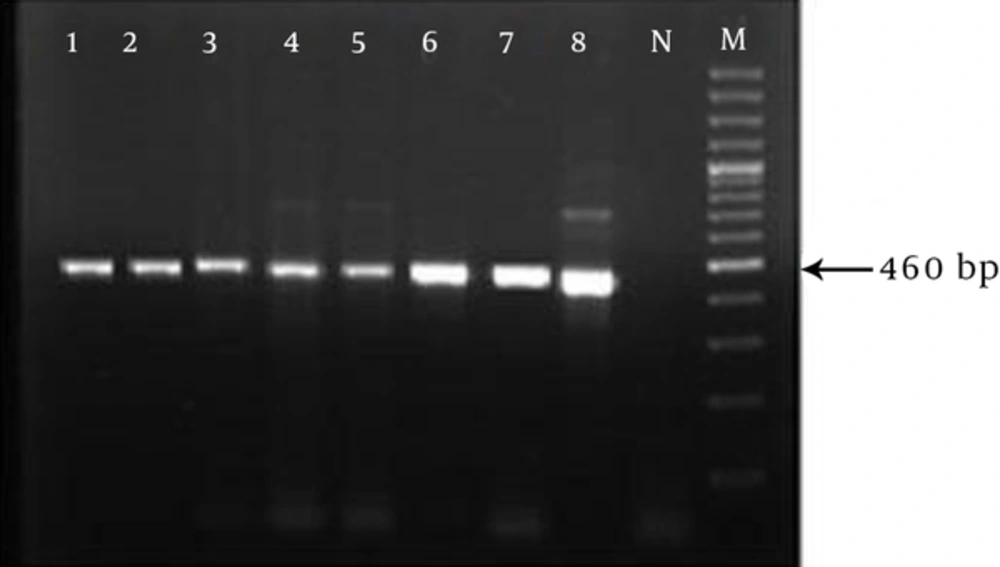
Partial amplification of BT2 was successful in all isolates and yielded a single PCR band of approximately 450 base pair (bp) in gel electrophoresis (Figure 2). Sequencing showed that the length of the isolates ranged between 445 to 460 bp.
Final identification of isolates was performed by comparing the obtained sequences with those of reference strains held in the open access validated CBS database for Penicillium (http://www.cbs.knaw.nl/penicillium/).
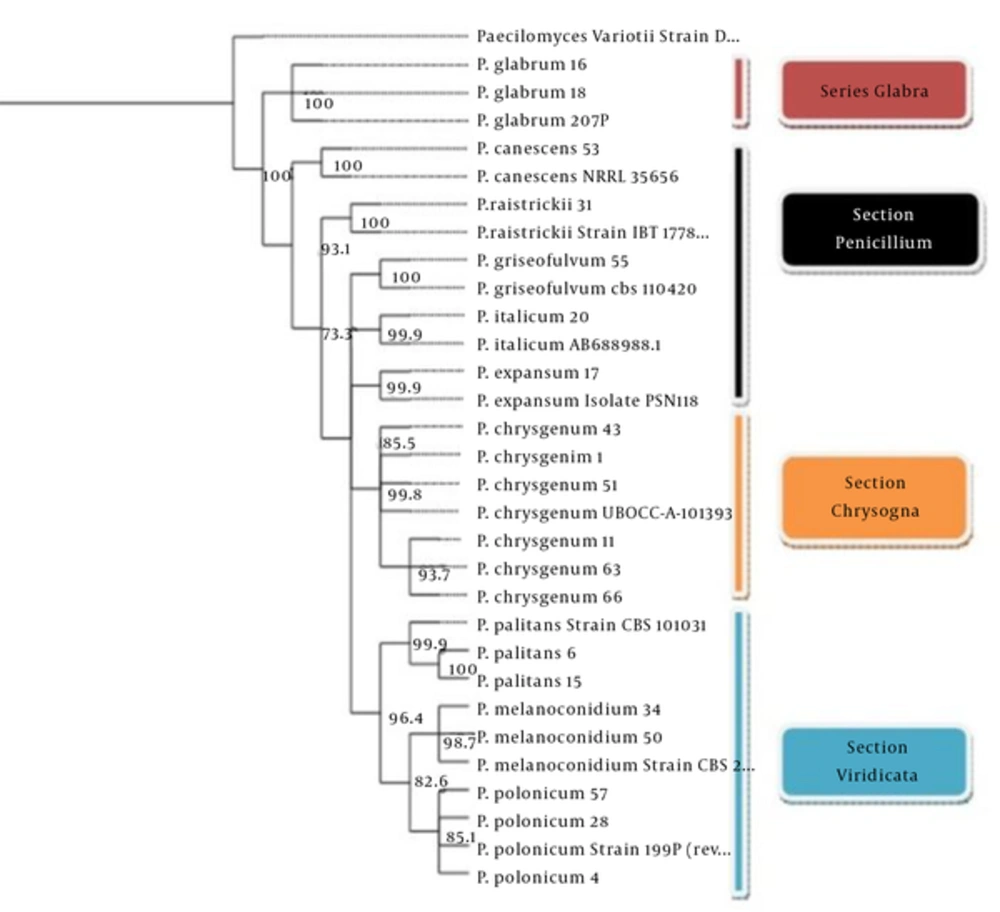
Sequences of all isolates were deposited in GenBank and the accession numbers of the sequences are given in Table 1. In Table 1 the results of sequence and phenotypic-based identification were compared. Figure 1 shows that phylogenetic reconstruction was successfully carried out for the BT2 region and represented the maximum parsimony phylogenetic tree reconstructed o then β-tubulin gene. The phylogenetic assessment showed that there were three major sections, consisting of Viridicata, Chrysogena, Penicillium, and one series, Glabra including P. glabrum.
4.3. Viridicata Section Dataset
The analysis showed that the environmental species including P. melanoconidium (represented by two strains), P. polonicum (represented by three strains), P. palitans (represented by two strains), and the related standard strains (represented by three strains) are monophyletic and well supported (96.4% bootstrap, Figure 3).
4.4. Penicillium Section Dataset
The phylogenetic tree (Figure 3) shows that the Penicillium section is composed of five species, P. canescens, P. italicum, P. expansum, P. griseofulvum and P. raistrickii, which are phylogenetically distinct from other species and sister to each other and standard strains in this section. The species of the Penicillium section were strongly (93.1%) well supported by bootstrap.
4.5. Chrysogena Section and Glabra Series Dataset
Among accepted members of the Chrysogena section and the glabra series, only P. chrysogenum and P. glabrum, respectively, were isolated from all the examined isolates. The phylogenetic tree (Figure 3) shows that six P. chrysogenum and one standard strain (UBOCC-A 101393) formed a well-supported monophyletic group nested within this section of Penicillium.
5. Discussion
To evaluate biodiversity and phylogeny of the Penicillium species in Mazandaran and Tehran Provinces, the current study focused on morphology-based identification and sequencing of the β-tubulin gene. Except for the studies by Sabokbar et al. and Javadi et al. there has been no extensive attempt at this type of study, including reliable morphological and high-resolution molecular approaches for Penicillium identification, until now in Iran (15, 16). Over the past decades, focusing on macro- and microscopic characteristics of the colonies has been the main strategy for classification and identification of the Penicillium species (2, 17). In our study, which was morphologically based on three specific medium (CYA, MEA, and YES) (2), 10 species of Penicillium were identified, compared to Javadi et al. who did not focus on phenotypic characteristics and whose sampling was restricted, taken only from soil (16). Also, the study performed by Sabokbar et al. showed intra-species variation of Penicillium species isolated from air in Iran using RAPD-PCR, but did not consider identification of the Penicillium species (15).
In the present study, in descending arrangement of frequency, the isolates that were identified were: P. chrysogenum, P. polonicum, P. palitans, P. glabrum, P. melanoconidium, P. expansum, P. raistrickii, P. italicum, P. griseofulvum, and P. canescens. Contrary to ITS regions, the β-tubulin gene is known to have more variation in Penicillia and currently is considered a reliable alternative marker for differentiation of the Penicillium species (5). Since identification of all Penicillium species by morphology was stated to be very problematic and often impossible, relying on sequencing of the high resolution genetic marker, β–tubulin, allowed for accurate and rapid recognition of the isolates in our study (2, 14). Similar to Barreto et al. parsimony analysis was used in our study to determine the phylogeny of the isolates (9). The Viridicata section is substantially divided into five series including Viridicata, Camemberti, Corymbifera, Verrucosa, and Solita (9). In the current survey, only species related to two former series, such as P. melanoconidium, P. polonicum, and P. palitans, were isolated, of which P. melanoconidium and P. polonicum are regarded to be prone to cereals such as barley, wheat, rye, oats, rice, and cause mycotoxicosis symptoms in humans (2).
Also, P. palitans may produce some significant toxins such as cyclopiazonic acid and fumigaclavine on foods, and the former is proven to be an agent of organ damage in mammals (2). In the Penicillium section, P. griseofulvum, as the main producer of griseofulvin, which is placed in ser. Urticicolae alongside P. raistrickii and P. canescens, had completely identical sequences with the standard strain (100%). Penicillium expansum and P. italicum, two members of the Expansa and Italica series, respectively, had 99.9% identity to the standard sequences (PSN118and AB688988.1). Penicillium chrysogenum, the predominant isolate in our study, is assigned to section Chrysogena, ser. Chrysogena (9). The species belonging to this section vary from one geographic site to another. Whereas P. chrysogenum, P. spinulosum, and P. oxalicum ranked as the first infrequency in Kansas, P. citrinum was the main agent isolate from the Taipei area (18). Penicillium chrysogenum was the predominant agent among all assessed isolates in this study, and this finding was concordant with other reports from around the world (19). Extra evaluation, with emphasis on polyphasic procedures, not only phenotypic methods but also molecular approaches focusing on a combination of partial β-tubulin, calmodulin, and ITS sequence, are required to clarify the complete spectrum of the Penicillium species in Iran.
5.1. Conclusions
The current investigation found a high level of genetic variability in the β-tubulin gene in various Penicillium species isolated from soil, fruits, food, and grains of Mazandaran and Tehran Provinces. This study reconfirmed the view that BT2 PCR-sequencing is a reliable and applicable diagnostic tool for differentiating and the molecular taxonomy of closely related Penicillium species.