1. Background
Since the early 1980s, fungi have been known as one of the important causes of human infection diseases, especially among immunocompromised individuals (1, 2). The morbidity and mortality associated with these infections are considerable (3, 4), so it is obvious that fungal diseases are important public health problems (5, 6).
Endoproteinases, carboxypeptidases and aminopeptidases are vacuolar proteases that have intracellular proteolytic activities in Candida albicans. Intracellular proteinase A is one of the most important enzymes and is produced by the APR1 gene in C. albicans (7). It is demonstrated that this enzyme has functions in the Candida genus, such as nutrition, help with penetration and invasion, the infection of host tissues and the suppression of the immune system (8, 9). In a study conducted by Mechler and Wolf, (10) the lack of proteinase A led to 30% and 40% reductions in protein degradation under sporulation conditions and sporulation frequency in mutant cells, respectively. Yeast aspartyl proteases are considered an important group of protease enzymes that catalyze the breakdown of peptide in yeast cells (7). In response to several types of stress, a group of genes are activated in C. albicans, where each type of stress is responded to by a particular set of genes (11). It is suggested that the increase of APR1 gene expression will impact C. albicans compatibility in the change of environmental conditions (12).
Multiple sclerosis (MS) is one of the most common chronic neurological disorders, especially in young adults. It is suggested that this disease may be caused by some non-genetic environmental factors, such as infectious agents (13-16). The medical importance of Candida species, such as C. albicans, as infectious agents can cause demyelinated lesions in MS patients (17, 18). The investigators suggested two hypotheses about fungal infections in MS patients: i) Fungal infections can play a major role in the development of MS, and ii) fungal infections in MS may be due to immune disorders. Candida species can have an unconstructive effect in the clinical course of MS even though they may not be the main etiology of the disease (13, 19). It is possible that the secretion of fungal proteases could damage the surrounding tissue, leading to inflammation. Thus, the continuous synthesis of proteolytic enzymes would account for the alterations observed in the CNS (20). Candida infection among MS patients has not been studied in depth. However, the possibility that MS is caused by an infective agent has been put forward.
2. Objectives
The aim of this study was to evaluate the expression of the APR1 gene in C. albicans isolates obtained from MS patients with immune disorders and from controls.
3. Patients and Methods
3.1. Patients and Controls
One hundred and thirty-five MS patients with fungal infections were included in this study. The samples were obtained from four subtypes of MS disease. Of 91 patients with the relapsing-remitting subtype (RR), 51 cases used MS drugs, while 40 cases did not receive any MS and antifungal drugs at the sampling time. A total of 44 patients belonged to the other MS subtypes, including: i) secondary progressive (SP), ii) primary progressive (PP) and iii) progressive-relapsing (PR) subtypes. All MS patients in these three subtypes consumed MS drugs without using any antifungal drugs.
One-hundred control samples were obtained from blood donors in the hospital and from volunteer students. According to the results of the laboratory analysis, the control samples exhibited no problems in the immune system, while MS patients had immune dysfunction. The immune dysfunction in the three subtypes of MS (PP, SP and PR) was lesser than that in the RR subtype. The control samples had a harmony in age and sex with MS patients (Table 1). The expanded disability status scale (EDSS) was described as ranging from 0 to 10, with normal neurologic examination and death due to MS in half-point increments, respectively (21). All of the C. albicans strains were isolated from the superficial and mucosal areas of MS patients, such as a nail, the skin and the vagina. The study protocol was approved by the Ethics Committee of the Tehran University of Medical Sciences and registered under Code Number 1562302. Informed written consent was obtained from all subjects prior to their entering the study.
| Clinical Information | MS Patients |
|---|---|
| Total individuals with MS | 135 |
| Female/male ratio | 2/1 |
| Age at onset in years, Mean ± SD | 21.8 ± 4.8 |
| Disease duration in years, Mean ± SD | 3.30 ± 2.2 |
| Disease course, No. (%) | |
| RR | 91 (67.4) |
| PP | 19 (14.1) |
| SP | 18 (13.3) |
| PR | 7 (5.2) |
| EDSS | |
| ≤ 3 | 113 (83.7) |
| 3.5 - 6 | 18 (13.3) |
| > 6 | 4 (3.0) |
Abbreviations: EDSS, expanded disability status scale; PP, primary progressive; PR, progressive-relapsing; RR, relapsing-remitting; SD, standard deviation; SP, secondary progressive.
3.2. Candida albicans Isolation and Cultivation
The clinical and control isolates of C. albicans obtained from individuals were cultured onto sabouraud dextrose agar (SDA) (Merck, Germany) at 37°C for 24 hours. For isolating the yeast cells, Candida colonies were harvested from SDA (Merck, Germany) media and placed into a 1 mL microfuge tube with 0.5 mL distilled water. For equaling the cell number in different samples of Candida, the OD of the cells was read at 550 nm. Subsequently, Candida cell suspensions were heated at 100°C for 10 minutes and centrifuged at 4500 × g at 4°C for 15 minutes. Finally, the supernatants were used in the reverse transcriptase-polymerase chain reaction (RT-PCR) process.
3.3. RT-PCR Reaction
To evaluate the expression of the APR1 gene, the RT-PCR reaction was performed in three steps: i) ribonucleic acid (RNA) extraction, ii) RT reaction and iii) PCR reaction.
A) RNA extraction: Total RNA was extracted using RNX-plus buffer (Cinagen, Iran). Briefly, about 2 × 106 cells were transferred to 1 mL of RNX-plus buffer (Cinagen, Iran) in an RNase-free microtube, mixed thoroughly and left at room temperature for 5 minutes. A volume of 200 µL of chloroform (Merck, Germany) was added to the slurry and was mixed gently. The mixture was centrifuged at 13,200 × g at 4°C for 15 minutes; the supernatant was transferred to a new tube and was precipitated with an equal volume of isopropanol (Merck, Germany) for 15 minutes on ice. The RNA pellet was washed using 75% ethanol (Merck, Germany), briefly dried and resuspended in 15 µL of RNase free water. The purified total RNA was quantified by a NanoDrop ND 1,000 spectrophotometer (Wilmington, USA).
B) RT reaction: A sample (0.005 mg) of RNA was used for first-strand cDNA synthesis, using 100 pmol oligo-dT (18 mer) (Fermentas, Germany), 15 pmol dNTPs (Fermentas, Germany), a 20 U RNase inhibitor (Fermentas, Germany), and a 200 U M-Mulv reverse transcriptase (Fermentas, Germany) in a 0.02 mL final volume. Twenty µL of solution were obtained and maintained at room temperature for 10 minutes. These solutions were incubated at 42°C for 60 minutes in a heat block (Clifton, USA) and then at 70°C for 10 minutes.
C) PCR reaction: Two-pair primers (Macrogen, South Korea) including APR1-f: 5ʹ-TCCACCAATCTACAATGCCA-3ʹ and APR1-r: 5ʹ-ATTTCAGCCAATGAGGATGG-3ʹ as the proteinase A gene, and 18S rRNA-f: 5ʹ-GCCAGCGAGTATTAACCTTG-3ʹ and 18S rRNA-r: 5ʹ-ATTTCAGCCAATGAGGATGG-3ʹ as the housekeeping gene were selected for this study (22). PCR amplification was carried out in a final volume of 25 μL containing PCR buffer, MgCl2, dNTP, reverse primer, forward primer, template cDNA (0.1 ng), Taq polymerase (0.2 units) and distilled water (Amplicon, Denmark). The best annealing temperature was considered 55.5°C for both APR1 and 18S rRNA genes.
3.4. Agarose Gel Electrophoresis
To show the cDNA bands of APR1 and 18S rRNA genes, the PCR products were run on 1.5% (w/v) agarose gel (Merck, Germany). A 1 kb DNA ladder (Fermentas, Germany) was used as a marker at this stage.
3.5. Statistics
The statistical analysis of the expression of the APR1 gene in C. albicans strains isolated from MS patients and from controls was performed using the independent T-test (SPSS, version 17, Chicago, USA) method. The differences were considered to be significant at P < 0.05. The correlation between APR1 gene expression with age at onset in years and EDSS was investigated using the Pearson correlation test, and the correlation between the APR1 gene with MS subtypes and sex was detected using the Chi-square (X2) test in this software program.
4. Results
The data analysis of sex, age, disease duration, EDSS and different subtypes of MS are shown in Table 1. A total of 135 MS patients and 100 healthy controls participated in this study; 41 of the former were male, and 94 were female. MS was observed in patients ranging from 17 to 42 years (mean age: 25 ± 6.4 years). The mean disease duration ranged from 1 to 11 years (mean duration: 3.3 ± 2 years). The mean EDSS ranged from 0.5 to 7 (mean value: 2.25 ± 1.5), and in most of the patients, the EDSS was less than 3 (67.4%). Ninety-one (67.4 %) of the MS patients were in the subtype RR, 19 (14.1%) in PP, 18 (13.3%) in SP and 7 (5.2%) in PR.
As shown in Table 2, the correlations between APR1 gene expression and clinical variables were investigated. There were statistically significant correlations between APR1 gene expression and age at disease onset as well as EDSS (P = 0.000), whereas no significant correlation was found between the gene expression and MS subtypes as well as sex (P > 0.05). Because of some limitations in doing laboratory examinations, among the clinical features of MS patients, only the EDSS of the MS patients was investigated after 9 months of the treatment of these patients with antifungal drugs. The results showed that the mean values of EDSS were 1.6074 ± 0.1081 after antifungal treatment and 2.2519 ± 0.1323 before antifungal treatment, representing a statistically significant difference between what occurred before and after treatment with antifungal drugs (P = 0.000). Our findings demonstrated that the progression of MS decreased after the treatment of patients with antifungal drugs.
aNo significant.
Abbreviations: EDSS, expanded disability status scale; SD, standard deviation.
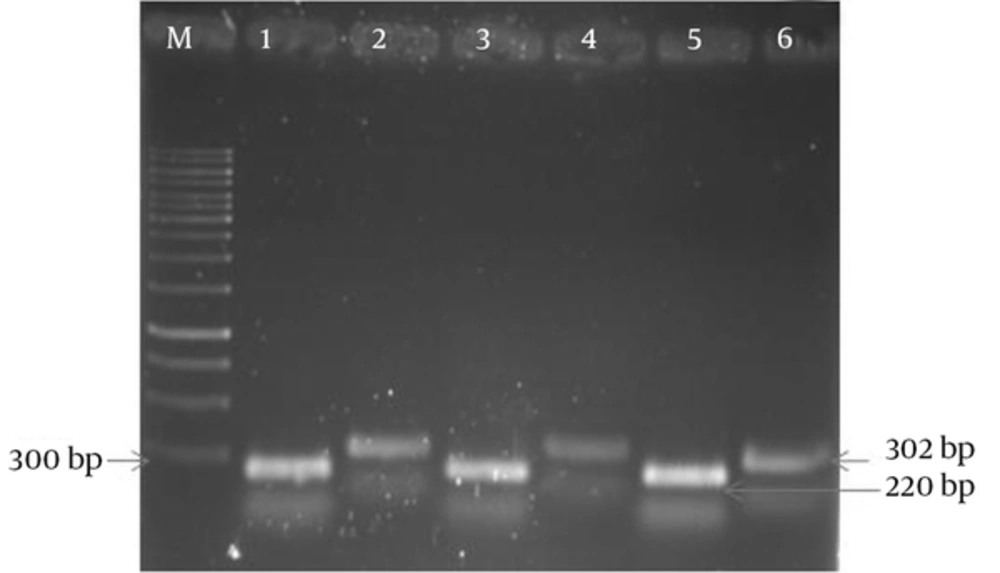
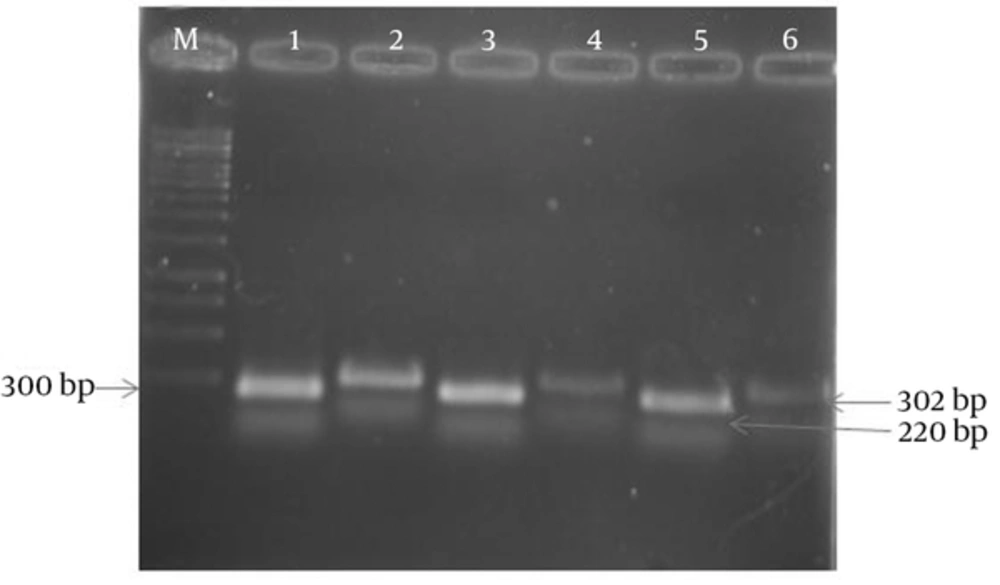
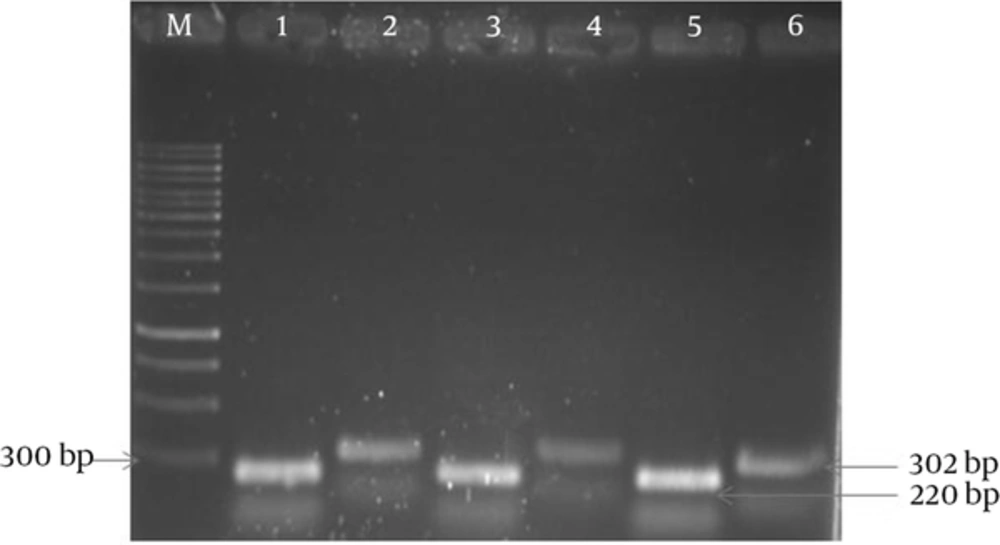
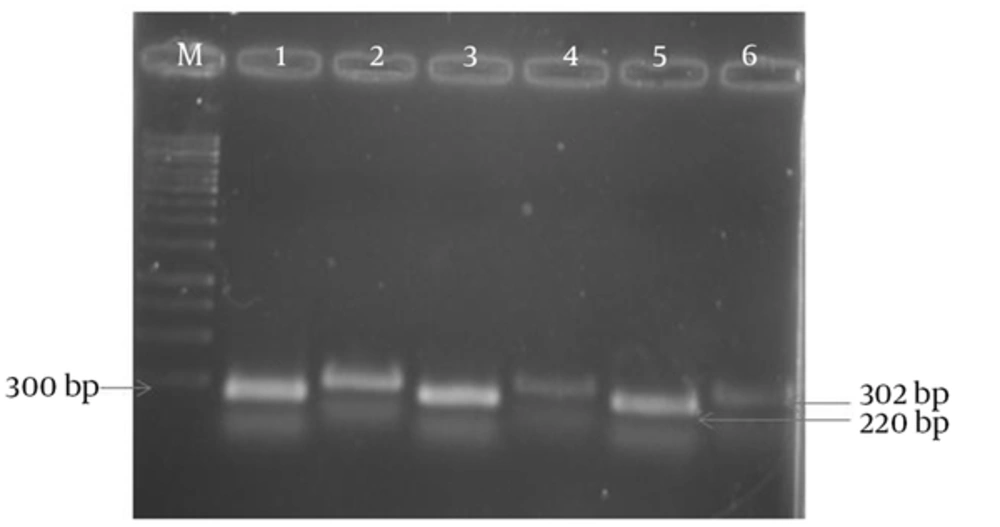
In this study, APR1 gene expression was investigated in C. albicans strains isolated from 135 MS patients in four subtypes (RR, PP, SP and PR) (Table 3). According to the data, there were no statistically significant differences in the mean ratio of APR1 to 18S rRNA genes in C. albicans strains isolated from four subtypes of MS patients with candidiasis (RR = 0.5467 ± 0.08185 unit/mg, PP = 0.5115 ± 0.07375 unit/mg, SP = 0.4929 ± 0.08588 unit/mg and PR = 0.1737 ± 0.05063 unit/mg). In contrast, the mean ratio in C. albicans strains isolated from four subtypes of MS patients (0.5208 ± 0.11518 unit/mg) was significantly less than that of the control group (0.7603 ± 0.11405 unit/mg) (P = 0.000).
| Variable | Ratio of APR1/18S rRNA, Mean ± SD, unit/mg | ||||
|---|---|---|---|---|---|
| RR | PP | SP | PR | Total Samples | |
| MS patients, No. 135 | 0.5467 ± 0.08185 | 0.5115 ± 0.07375 | 0.4929 ± 0.08588 | 0.1737 ± 0.05063 | 0.5208 ± 0.11518 |
| Controls, No. 100 | - | - | - | - | 0.7603 ± 0.11405 |
| P value | - | - | - | - | 0.000 |
Abbreviations: PP, primary progressive; PR, progressive-relapsing; RR, relapsing-remitting SD, standard deviation; SP, secondary progressive.
The results of APR1 and 18S rRNA gene expressions showed different expressions among various subtypes of MS patients (Figures 1 and 2) and also between MS patients and controls (Figures 3 and 4). As observed, the APR1 and 18S rRNA bands appeared on the agarose gel after the electrophoresis of the clinical isolates of C. albicans obtained from the MS patients and controls. The locations of the APR1 and 18S rRNA gene bands were detected about 220 and 302 bp, respectively. This change in density may not be distinguishable through agarose gel electrophoresis, but it can be shown in the calculation by the lab works 4.6 Acquisition and analysis program CD software program (Table 3). The comparison of APR1 gene expression via this software showed that the gene expression increased in C. albicans strains obtained from controls when compared to MS patients.
5. Discussion
MS is characterized by multifocal spots of demyelination, and its severity is influenced by many factors. The aim of this study was to evaluate APR1 gene expression producing proteinase A in C. albicans strains isolated from MS patients. To the best of our knowledge, there is no information about APR1 gene expression in C. albicans isolated from MS patients, so this study can be considered a novel research study. Previous studies indicated the effect of environmental factors (non-genetic), especially infectious agents, on MS (15, 18). There is a hypothesis about the association between some pathogenic yeasts, such as C. albicans, C. famata, C. parapsilosis and C. glabrata, and MS (17). In MS patients with candidiasis, demyelinated lesions have been shown in the central nervous system (23). Intracellular proteinase A is one of the most important enzymes in C. albicans that is expressed by the APR1 gene. This enzyme has an important role in cell survival under stress conditions in C. albicans, and the amount of this enzyme increases in the first few hours of nitrogen starvation when compared to mycelium growth (24, 25).
The present study showed that the expression of the APR1 gene in controls was more than that of MS patients. This difference could be related to both the situation of yeast cells in the human body and the existence of immune disorders. The immune system is involved in infectious diseases, such as candidiasis, and also in MS. In Candida infection, monocytes/macrophages synthesize chemokines and cytokines, leading to increased immune responses (26) and interleukin-10 (IL-10) and IL-12 production (27). The main protective mechanism in candidiasis is innate immunity, and T-cell response plays an important role in the control of infection through adaptive immunity (28). In MS, the activation of T cells is important in the first step of this disease and also in the progressive steps (29). Fradin et al. (30) indicated that environmental conditions, such as external pH, accessible nutrients, oxygen supply or the challenge of particular host defenses, such as antimicrobial peptides or leukocytes, can impact a range of differential gene expressions. In fact, C. albicans has to express the necessary genes for optimal growth or viability in immunocompetent hosts, whereas yeast cells are easily adapted to hosts with immune disorders and are disseminated into the body. It is suggested that the level of immune function has a direct relationship with APR1 gene expression in C. albicans. Therefore, the expression of the APR1 gene in MS patients with immune disorders was less than that of the control group with normal immune systems.
There are serious immune or metabolic deficiencies in patients with opportunistic fungal infections, and these patients are also under immunosuppressive therapy (31). The corticosteroids are one of the immunosuppressive drugs in MS patients that can reduce inflammation and cause the progression of the disease (32). In addition, immunosuppressive drugs decrease immune functions in MS patients, thus leading to the dissemination of fungal infections.
The present study showed that all MS patients in the relapsing condition (RR subtype, 91 cases) were recently involved in Candida infection and did not use any antifungal drugs. Some of these patients were treated by MS drugs (51 cases) and have immune dysfunction. The other patients (40 cases) did not receive any immunosuppressive drugs, and the immune systems of these patients were weak because of MS. All of the patients with the RR subtype were treated with antifungal drugs after sampling of the disease. No relapsing and progression were observed in MS after 9 months, and the EDSS of these patients was higher for after-antifungal treatment than for before treatment. In accordance with our findings, Pisa et al. (28) showed that no relapse and progression was observed in MS patients receiving antifungal drugs and those without any immunosuppressive therapy.
In this study, 44 patients were in the PP, SP and PR subtypes of MS with different immune disorders. They were under immunosuppressive drug treatment without using any antifungal drugs. These patients were treated with antifungal drugs after sampling, and the decrease of EDSS in these patients showed the low progression of MS after antifungal treatment. In addition, the immune response in the PR subtype of MS patients was stronger than that of the RR, SP and PP subtypes, so the adaptation of C. albicans and the expression of the APR1 gene in PR subtype were more than those of other subtypes of MS. It is suggested that Candida cells have an urgent need to increase protein production to supply nitrogen to the cells for adapting to normal immune conditions. Considering the role of proteinase A in Candida cells in the production of amino acids, which are substrates for new protein synthesis, it is logical for the amount of this enzyme to increase. In this situation, the APR1 gene is increased and, together with the increased production of messenger RNA (mRNA), creates an increase in the amount of proteinase A production.
It was detected that there are correlations between Candida infection and the increase of MS progression. Regarding the significant role of proteinase A in the production of new proteins in C. albicans, the APR1 gene was found to be an important gene in the adaptation of this yeast in different situations. It was also found that there is a correlation between MS patients with fungal infections and the etiology of MS with fungal agents (18, 33). In conclusion, the expression of the APR1 gene in C. albicans was higher in controls than in MS patients using the RT-PCR technique. Our findings showed that the over-expression of the APR1 gene can be an important factor in the development of candidiasis caused by C. albicans. In addition, the results here will be useful precursors for achieving the future goal of further research of intracellular proteinases in Candida species because of their importance in human pathogenesis. The results of this study propose that Candida infection may be associated with the increased progression of MS. Therefore, it is suggested that future studies should make clear how Candida species act in the pathogenesis of MS.