1. Background
Tuberculosis (TB), a disease as old as human life, remains a serious worldwide health threat (1). Second-line drugs, which are expensive and toxic, are an inevitable treatment course for Mycobacterium tuberculosis isolates that are resistant to first-line medications. This form of M. tuberculosis is known as multidrug-resistant M. tuberculosis (MDR-M. tuberculosis) (2). Immediate and accurate treatment of TB is essential for controlling the disease; therefore, rapid and accurate detection of drug resistance is a key factor in the effort to control MDR-TB (3). The proportion method is a standard drug-susceptibility test for detection of drug resistance in M. tuberculosis (4). However, this test demands from eight days to more than one month for completion, a long time for a patient who is likely subjected to suboptimal treatment. Moreover, in most developing countries like Iran, drug-susceptibility tests are available only in reference laboratories, and because of this low availability, results are delayed (5).
According to broad analyses, mutations in the rpoB gene at the 81bp region are the cause of more than 98% of rifampicin (RMP) resistance (6), and a change in the katG 315 codon is the cause of more than 50% of isoniazid (INH) resistance (7). The world health organization (WHO) recommends the development and implementation of molecular approaches for detecting M. tuberculosis drug-susceptibility, especially in developing countries (5). Real-time polymerase chain reaction (PCR) is a widely used molecular technique, as it is a high-speed method with a low risk of contamination (8-10). Melting curve analysis is a simple approach for detecting mutations by real-time PCR (1).
2. Objectives
The purpose of this study was to assess the potential of real-time PCR melting curve analysis for the detection of mutations associated with INH and RMP resistance in M. tuberculosis clinical isolates.
3. Patients and Methods
In a cross-sectional study, 90 patients with pulmonary TB were randomly selected for analysis in 2014 from five border provinces of Iran, including east and west Azerbaijan, Ardebil, Kermanshah and Kurdistan. The university ethics committee code of study was 1391/6/18/5/4/5375.
3.1. Demographic Data and Analysis
Demographic and environmental data were collected using a questionnaire. Clinical characteristics were provided using the information mentioned in the surveying of medical records and with the assistance of the physician in charge of the treatment of TB; additional clinical details were obtained from personal interviews and home inspections. The validity and reliability of the questionnaire had been previously confirmed through a pilot study (Cronbach’s alpha = 0.98). Data was analyzed using univariate logistic regression in SPSS 18 (233 south Wacker drive, 11th floor, Chicago, Illinois, USA). The validity (sensitivity and specificity) test was done using the OpenEpi online program (open source epidemiologic statistics for public health, version. www.OpenEpi.com).
3.2. Inclusion Criteria
As determined by ELISA (enzyme-linked immune sorbent assay) test, all patients were HIV negative and received DOTS (directly observed therapy, short-course). The inclusion criteria of the study were smear-positive pulmonary TB with at least two initially positive sputum smears for acid-fast bacilli, or one positive sputum smear and radiographic abnormalities consistent with active pulmonary TB, or one positive smear and positive culture for M. tuberculosis (11). The M. tuberculosis complex was identified by culturing on Lowenstein-Jensen (LJ) medium containing p-nitro benzoic acid. The M. tuberculosis species was identified based on a niacin test and a nitratase test (a positive reaction of a pink color indicated nitrate had been reduced to nitrite (12).
3.3. Standard Proportion Test
A drug-susceptibility test was carried out in LJ medium according to the proportion method recommended by WHO/IUTLD (11) using INH: 0.2 mg/L, RMP: 40 mg/L. Incubation of the samples was continued for 43 days.
3.4. DNA Extraction
DNA was extracted from clinical isolates grown on LJ medium in the following process: at least two to three M. tuberculosis colony loops were suspended in 1X TE buffer, inactivated at 80°C for 20 minutes, washed three times with 1X TE, and incubated with lysozyme at 37°C for 24 hours. Afterward, proteinase K was added, and the samples were incubated at 65°C for another 24 hours. After adding CTAB/NaCl + 5M NaCl and chloroform/isoamyl alcohol, isopropanol was added to each sample and maintained in a -20°C freezer for 24 hours. Finally, after washing the DNA with 70% ethanol, the DNA was dissolved in 30 μL of distilled water and preserved at -20°C until use (13).
3.5. Real-Time PCR
Three dually labeled probes and four primers were designed to detect two genes associated with drug resistance. Usually, more than four probes should be used to assess the 81 bp region of the rpoB gene (1, 2, 14). We designed two long probes to detect mutations at rpoB for decreasing workload and consuming costs. The sequence of primers (forward and in reverse) and probes in rpoB 511 to 519 and 524 to 533 codons, and 315 codons katG genes are described in Table 1 (1).
| Detection Target | Probes (5'-3') | Forward Primers (5'-3') | Reverse Primers (5'-3') |
|---|---|---|---|
| katG 315 | TCA CCA GCG GCA TCG | CGT ATG GCA CCG GAA CCG GTA A | GCT CCC ACT CGT AGC CCG TAC A |
| rpoB 511-519 | GGT TGT GGT CCA GAA TTG GCT CAG C | GCC GCG ATC AAG GAG TTC TTC | CGG CAC GCT CAC GTG ACA GAC |
| rpoB 524-533 | GCC CCA GCG TCG ACA GCG GTG CTT GTC GG |
The Sequence of Primers and Probes in rpoB and katG Codons (1)
The content of real-time PCR reaction in a thermal cycler (applied biosystems® thermal cyclers CA, USA) with a total volume of 20 μL was as follows: PCR master mix (premix Ex Taq, Takara Bio Inc., Japan): 10 μL, primer mix (Forward-Reverse): 0.25 μL, probe: 0.5 μL, Rox reference Dye (Takara Bio Inc., Japan): 1 μL, sterilized distilled water: 7.25 μL and extracted DNA with the maximum concentration of 100 ng: 1 μL. PCR conditions were as follows: primary denaturation at 95°C for 10 minutes followed by denaturation at 95°C, 10; annealing at 58°C for 30 seconds and extending 72°C for 15 seconds (40 cycle). Each probe had a different melting condition in PCR so that, for rpoΒ 511 to 519: 95°C at 15 seconds; 58°C at 15 seconds; 95°C at 15. For rpoΒ 524 to 533: 95°C at 15; 58°C at 15; 95°C at 15 and melt katG 315: 95°C at 15 seconds; 55°C at 15 seconds; 95°C at 15 seconds. The isolates with a melting temperature (Tm) difference of 1.5°C (compared to the Tm of sensitive isolates) were considered as mutant isolates (1, 2, 14).
3.6. Multiplex Allele-Specific PCR (MAS-PCR)
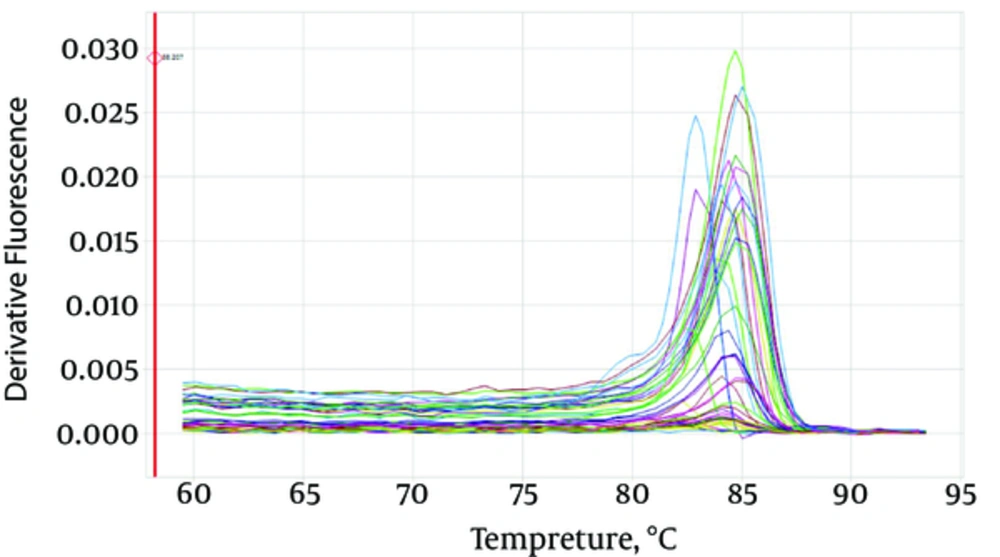
Using the MAS-PCR technique, codon 315 of the katG gene and codons 516, 526 and 531 of the rpoB gene were evaluated. Four allele-specific primers corresponding to the four target codons and two common primers were used in the MAS-PCR assay as described by Yang et al. (15). The MAS-PCR reaction was prepared in a 25 μL volume and contained 1 μL primer mix (6 primers: rpoB 516, rpoB 526, rpoB 531, RRm, katG FC, katG RC), 10 μL master mix (Ampliqon, Denmark), 1 μL DNA (with a concentration of at least 100 ng/µL) and 13 μL of distilled water. The thermal cycler (PeQ lab, primus 96, Germany) conditions followed were: initial denaturizing at 94°C for 7 minutes, 35 cycles of 94°C for 30 seconds, 68°C for 40 seconds and a final extension at 72°C for 5 minutes (Figure 1).
4. Results
4.1. Descriptive Results
The studied isolates were collected from 90 patients with an average age of 54.35 years (ranging from 15 - 83); 44 cases (48.9%) were male and 46 cases (51.1%) female. The content of sampling in different provinces was as follows: 28 cases (31.1%) were from east Azerbaijan, 25 cases (27.8%) from West Azerbaijan, 9 cases (10%) from Ardebil, 16 cases (17.8%) from Kermanshah and 12 cases (13.3%) from Kurdistan. TB category II or patients who are either diagnosed with a previous infection of TB or who have received the first line drugs but have faced failure, relapse or drug resistance during the treatment, had a shortage of access to drugs, or have encountered faults in receiving the treatment plan from health care providers, was detected in 9 (10%) patients. Seventy-seven percent of patients in category II had mutations in their katG and rpoB genes.
4.2. Real-Time PCR Results
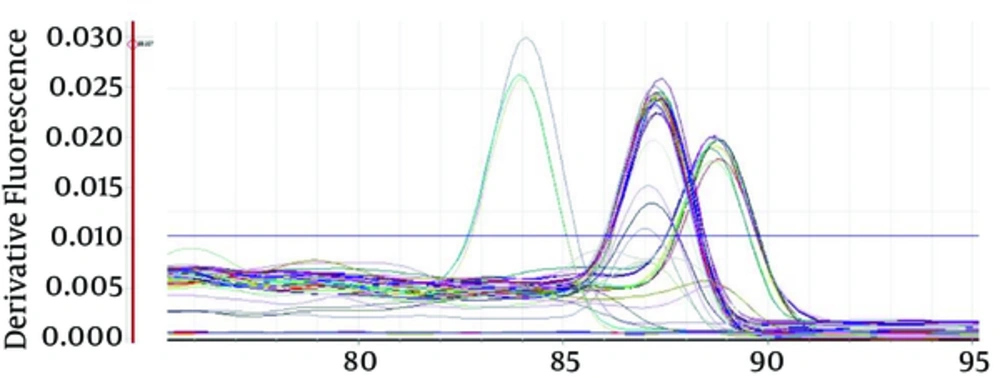
Melting curve method was used to study mutations associated with drug resistance. The mutations were detected by comparing the Tm of amplicons of resistant isolates with that of susceptible isolates. The frequency of all mutations was 23, where the frequency of mutations was nine for rpoB P1, 7 for rpoB P2 and 7 for katG 315. The frequency of isolates with at least one mutation in rpoB or katG was 11 cases. The frequency of mutations at katG 315 and rpoB P1 or rpoB P2 (in the likelihood of MDR-TB) was three cases (Figure 2). The average Tm and standard deviation (SD) of susceptible isolates was as follows: 315 katG codon 85.4, 0.18; rpoΒ P1 sequence 84.6, 0.6 and rpoΒ P2 sequence 83.4, 0.18 (Table 2)
| Type Mutation | TM, °C (SD) Susceptible Isolates | TM, °C (SD) Mutant Isolates | Mean Difference, °C |
|---|---|---|---|
| katG 315 | 85.4 (0.18) | 87.54 (0.62) | 2.14 |
| rpoB P1 | 84.6 (0.61) | 82.9 (0.38) | -1.7 |
| rpoB P2 | 83.37 (0.18) | 85.27 (0.19) | 1.9 |
Melt Temperature Results in katG 315, rpoB P1 and rpoB P2 Sequences
The average Tm of rpoB P1 in mutant isolates was 82.9 (SD: 0.38). In other words, mutant isolates in rpoB 511 to 519 codons were accompanied by a Tm decrease of 1.7°C (Table 2). The Tm of rpoB P2 in mutant isolates was 85.3 (SD: 0.19). This implies that mutations in rpoB 524 to 533 codons were accompanied by a Tm increase (1.9 °C). Therefore, a difference of more than two standard deviations from the rpoB P1 Tm and a difference of 10 standard deviations from the rpoB P2 Tm significantly accompanies mutation isolates. The average Tm in katG 315 in mutant isolates was 87.54°C (SD: 0.62) which was increased by 2.1°C compared with 315 katG Tm in susceptible isolates (Table 2). It should be noted that three isolates displayed a 3.42°C decrease in the katG Tm, and were removed from analysis (Figure 2). The distribution of frequency of katG and rpoB mutations based on provinces is shown in Table 3.
| Provinces (Number) | Frequency of katG Mutation | Frequency of rpoB Mutation | Frequency of Both katG and rpoB Mutation |
|---|---|---|---|
| East Azerbaijan (28)b | 1 (4.0) | 4 (14.3) | 1 (4.0) |
| West Azerbaijan (25) | 1 (4.0) | 4 (16.0) | 0 (0.0) |
| Ardabil (9) | 3 (33.3) | 1 (11.1) | 1 (11.1) |
| Kermanshah (16) | 1 (6.25) | 4 (25.0) | 0 (0) |
| Kurdistan (12) | 3 (25.0) | 1 (8.3) | 1 (8.3) |
| Total (90) | 9 (8.0) | 14 (15.6) | 3 (3.3) |
4.3. Comparison of Real-Time PCR Results and Standard Proportion Test
In this study, eight and 12 cases had resistance to INH and RMP, respectively. However, 4.4% (four cases) were MDR, i.e., they showed resistance to both INH and RMP. The sensitivity and specificity of real-time PCR in the detection of INH resistance were 75% (CI 95%; 40.93, 92.85) and 97.47%, respectively (CI 95%; 91.23, 99.3); in the detection of RMP resistance, its sensitivity and specificity were 83.3% (CI 95%; 55.2, 95.3) and 98.72% (CI 95%; 93.09, 99.77) (Table 4).
| Real Time-PCR Test | Proportion Test | Sensitivity (CI 95%) | Specificity (CI 95%) | |
|---|---|---|---|---|
| katG | Resistance to INH | Sensitive to INH | 75% (40.93, 92.85) | 97.47% (91.23, 99.3) |
| katG 315 mutation | 6 | 2 | ||
| Without mutation in katG 315 | 2 | 77 | ||
| rpoB | Resistance to RMP | Sensitive to RMP | 83.3% (55.29, 95.3) | 98.72% (93.72, 99.77) |
| rpoB codons(P1 and P2) mutation | 10 | 1 | ||
| Without mutation in rpoB codons (P1 and P2) | 2 | 77 | ||
Diagnostic of INH and RMP Resistance Test Evaluation of MTB Tuberculosis Patients in Real-Time PCR Results
4.4. Comparison of Real-Time PCR Results With MAS-PCR
The sensitivity and specificity of real-time PCR technique in identifying katG mutation by MAS- PCR were 100.0% (CI 95%; 51.0, 100.0) and 95.18% (CI 95%; 88.25, 98.11), respectively; in detection of mutation in rpoB codons (516, 526 and 531) its sensitivity and specificity were 80.0% (CI 95%; 49.02, 94.33) and 96.25% (CI 95%; 89.55, 98.72), respectively (Table 5).
| Real Time-PCR Test | MAS-PCR Test | Sensitivity (CI 95%) | Specificity (CI 95%) | |
|---|---|---|---|---|
| katG | KatG 315 mutation | Without mutation in KatG 315 | 100% (51.01, 100) | 95.18% (88.25, 98.11) |
| katG 315 mutation | 4 | 4 | ||
| Without mutation in katG 315 | 0 | 79 | ||
| rpoB | mutation in rpoB (516,526 and 531) codons | Without mutation in rpoB (516,526 and 531) codons | 80.0% (49.02, 94.33) | 96.25% (89.55, 98.72) |
| rpoB codons (P1 and P2) mutation | 8 | 3 | ||
| Without mutation in rpoB codons (P1 and P2) | 2 | 77 | ||
Diagnostic of katG 315 and rpoB (codons 516, 526 and 531) Mutation Test Based MAS-PCR Evaluation of MTB Tuberculosis Patients in Real-Time PCR Results
Studies of the relationship between age and mutation odds in the rpoB gene revealed a significant statistical relationship (β = 0.85 and P = 0.046). It was observed, however, that a previous history of using TB drugs increased mutation odds at rpoB (OR = 16 and P = 0.036) while gender, household size number, immigration history and smoking did not (P > 0.005). There was also no significant relationship between the above-mentioned variables and mutation odds in katG 315 (P > 0.005).
5. Discussion
The global incidence of TB drug resistance is very high in TB patients with a recurring illness. The recurrence rate in low- and middle-income countries is estimated to be approximately 10 - 20% (16). In 2004, approximately 6.8% of the TB patients in Iran were in the recurrent group (17). We found that 7.2% of the patients in our previous study had recurrent TB (17). In the present study, 10% of patients were diagnosed with recurrent TB. The higher frequency of mutations (25%) in the present study might be due to a higher proportion of recurrent TB in isolates in this study (10%).
Mohajeri et al. (2015) used a density array (LCD-array) to detect mutations within the 90-bp rpoB region in the sputum of TB patients from western Iran. Of the 125 M. tuberculosis isolates, 35 subjects (28%) were found to be rifampicin resistant (18). This result had c very close to consistent with our study (25%).
In this study, we performed an inexpensive and rapid test using a real-time PCR approach, employing melting curve analysis of dually labeled probes in order to detect current mutations leading to INH and RMP resistance. A number of similar studies have used melting curve analysis based on FRET (fluorescence resonance energy transfer) probes to detect current mutations in TB (1, 10, 14, 15). In our current study, the maximum mean SD of Tm in susceptible isolates was 0.61°C. On the other hand, the minimum distance between Tm in susceptible isolates and mutant isolates was above 1.7°C (related to rpoB). This implies that the detection of mutations using the melting analysis technique provided a higher differentiating ability. In some previous studies (8, 14, 19), Tm of katG and rpoB in mutant isolates differed more than two standard deviations from the average Tm of susceptible species. Torres et al. (19) studied 30 RMP-resistant isolates using two pairs of primers. They showed a decrease by 6°C in Tm of mutant isolates in rpoB 513 to 516 codons, but an increase by 2°C in Tm of mutant isolates in rpoB 526 to 531 codons (19). In Garcia de Viedma et al. study (20), the isolates with mutation in rpoB 515 and 516 codons showed a decrease of 2.48 - 8.39°C in Tm, while codons 526 and 531 showed a decrease of 6.92°C, which is inconsistent with our results.
Torres et al. showed that the average Tm of katG 315 in susceptible species was 72.8°C (19). Also, Marin et al. estimated an average of 70.85°C of Tm in the 315 katG codon in susceptible species (14). According to our study's estimations, the average Tm of katG of mutated species was 87.54°C. In our current study, 83.3% of RMP-resistant isolates were detected by real-time PCR technique using two probes that included codons 513 to 516 and codons 526 to 531. However, 75% of INH-resistant isolates were detected by this technique. It should be noted that the result of the real-time PCR test was blind to the proportional test result.
In this study, 80.0% of rpoB codon mutations in MAS-PCR were detected by real-time PCR technique using two probes including codons 513 to 516 and codons 526 to 531. However, 100% of katG mutations were detected by this technique. In the study by Feizabadi et al. (2009 - 2012) on M. tuberculosis clinical isolates, the sensitivity and specificity of RMP resistance detection by high-resolution melting curve (HRM) analysis of the 129-bp fragment of rpoB were estimated at 95% and 100%, respectively. Also, sensitivity and specificity of INH resistance detection by HRM analysis were reported at 85.7% and 100%, respectively (21).
Sheikholslami et al. (2011) calculated the validity of single-strand confirmation polymorphism to detect drug resistance compared to DNA sequencing. The sensitivity and specificity of this assay in detection of the rpoB gene were 70.8% and 88%, respectively (22). It is clear from our research that, previous history of TB treatment and aging increased the odds of RMP resistance. A great number of studies, however, have found a significant relationship between previous history of TB treatment and drug resistance (20, 23, 24), while there are contradictory findings regarding the relationship between age and mutation odds (20, 23). The limitation of this study was its low budget, so it was not possible to perform the study on a higher sample size. Also, the role of mutation of inhA, ahp and oxyR genes in the creation of INH resistance was not identified.
In conclusion, the detection of drug-resistant mutations through melting analysis technique had a high differentiating ability. It is essential to monitor older patients and those with a previous history of TB treatment using rapid and accurate techniques t of determining their drug resistance odds.