1. Background
Cystic fibrosis (CF) is an autosomal recessive genetic disorder, caused by one of many different mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) protein, which regulates the movement of chloride and sodium ions across epithelial membranes (1). The lungs of patients with CF often become colonized by bacteria from an early age, which can lead to chronic infections.
Bacterial species, most commonly associated with respiratory tract infections in CF patients, include Staphylococcus aureus, Haemophilus influenzae, and most commonly, Pseudomonas aeruginosa, as a frequent cause of acute and chronic lung infections in these patients (2). Other Gram-negative non-fermenting organisms, such as Stenotrophomonas maltophilia, Burkholderia cepacia complex members, and Alcaligenes xylosoxidans, have been also reported to cause respiratory infections in CF patients (3).
Members of Enterobacteriaceae family are considered as transient colonizers of the airways in CF patients and seem to play a minor role in the development of severe respiratory diseases (4). Klebsiella pneumoniae has been shown to cause chronic obstructive pulmonary disease with symptoms such as recurrent cough and acute exacerbation (5). More recently, this organism has been isolated from respiratory infections in CF patients (6-9).
Cephalosporins are often used to treat respiratory tract infections caused by Gram-negative pathogens. However, it is difficult to eradicate these infections mostly due to multidrug-resistant strains, which produce Ambler class A extended-spectrum β-lactamases (ESBLs) (10). ESBLs comprise a rapidly growing group of β-lactamases, encoded by mobile genetic elements, which are capable of hydrolyzing penicillin, cephalosporins, and aztreonam.
The most frequently encountered ESBLs belong to TEM, SHV, and CTX-M families (11). Another class of enzymes, ie, class D β-lactamases (oxacillinases or OXA-type β-lactamases), was first shown to hydrolyze cloxacillin and oxacillin faster than benzyl penicillin and was later reported to hydrolyze amino- and carboxypenicillin (12, 13). Moreover, production of OXA-type enzymes, such as OXA-23, 40, 51, 58, and 48 groups, has been shown in K. pneumoniae (14).
2. Objectives
We aimed to investigate the antibiotic resistance profiles and prevalence of ESBL and OXA type β-lactamase production in K. pneumoniae isolates from pediatric CF patients in Tehran, Iran.
3. Methods
3.1. Ethics Statement
Ethical approval was obtained from the ethics committee of Shahid Beheshti University (grant No., D/600/3490).
3.2. Bacterial Isolates
Sixteen K. pneumoniae strains were identified among 98 bacterial isolates from the sputum samples of children with CF at Mofid children’s hospital in Tehran between July 2012 and February 2013. Among patients with K. pneumoniae colonization, 13 (81.25%) were male and 3 (18.75%) were female within the age range of 6 months to 8 years (median, 24 months).
Bacterial isolates were maintained in brain heart infusion broth (Oxoid, UK), containing 10% dimethyl sulfoxide at -20°C until further use. Klebsiella pneumoniae 7881 harboring blaTEM, blaSHV, and blaCTX-M genes, Pseudomonas aeruginosa KOAS carrying blaPER-1, P. aeruginosa 10.2 harboring blaVEB-1, and P. aeruginosa containing blaOXA were obtained from pasteur institute of Iran and used as the positive controls in polymerase chain reaction (PCR) experiments. Klebsiella pneumoniae ATCC 10031 was also used as the control in antibiotic susceptibility tests.
3.3. Antimicrobial Susceptibility
Antimicrobial susceptibility was determined by Kirby-Bauer disc diffusion method according to the clinical and laboratory standards institute (CLSI) guidelines (15). The following antibiotic discs (MAST Diagnostics, Merseyside, UK) were used in the present study: ceftazidime (CAZ, 30 μg), ceftriaxone (CRO, 30 μg), cefoxitin (FOX, 30 μg), cefotaxime (CTX, 30 μg), cefepime (CPM, 30 μg), imipenem (IMP, 10 μg), meropenem (MEM, 10 μg), ertapenem (ETP, 10 μg), aztreonam (ATM, 30 μg), piperacillin (PRL, 100 μg), carbenicillin (PY, 100 μg), ticarcillin (TC, 75 μg), amikacin (AK, 30 μg), tobramycin (TN, 10 μg), ciprofloxacin (CIP, 5 μg), co-amoxiclav (augmentin, AUG, 20/10 μg), ampicillin-sulbactam (SAM, 10/10 μg), and piperacillin/tazobactam (PTZ, 100/10 μg). The minimum inhibitory concentration (MIC) of ceftazidime was determined via broth microdilution.
3.4. Screening for ESBL Production
According to the CLSI recommendations, isolates with reduced susceptibility to 1 or more oxyimino-cephalosporins, including ceftazidime (≤ 22 mm), cefotaxime (≤ 22 mm), ceftriaxone (≤ 25 mm), and aztreonam (≤ 27 mm), were selected as probable ESBL producers (15). These organisms were then subjected to the phenotypic confirmatory test, using discs containing cefotaxime (30 μg), cefotaxime plus clavulanic acid (30/10 μg), ceftazidime (30 μg), and ceftazidime plus clavulanic acid (30/10 μg). An increase of ≥ 5 mm in the zone diameter for either antibiotic in combination with clavulanic acid or antibiotic alone confirmed the isolate as an ESBL producer.
3.5. PCR Amplification of β-lactamase Genes
DNA extraction was carried out by boiling. Briefly, a loopful of bacterial colonies was suspended in 500 μL of sterile double-distilled water. The suspensions were heated at 100°C for 10 minutes, and cellular debris was removed by centrifugation at 13000 × g for 10 minutes. The supernatant was used as the DNA template for PCR amplification of ESBL genes, using the primers listed in Table 1.
| Primer | Sequence (5’ to 3’) | Annealing Temperature (°C) | Product Size (bp) | Reference No. |
|---|---|---|---|---|
| TEM-F | GAGTATTCAACATTTCCGTGTC | 45 | 889 | (16) |
| TEM-R | TAATCAGTGAGGCACCTATCTC | |||
| SHV-F | ATGCGTTATATTCGCCTGTG | 58 | 862 | (17) |
| SHV-R | AGCGTTGCCAGTGCTCGATC | |||
| CTX-M-F | CGCTTTGCGATGTGCAG | 63 | 550 | (18) |
| CTX-M-R | ACCGCGATATCGTTGGT | |||
| PER-1-F | ATGAATGTCATTATAAAAGC | 43 | 920 | (18) |
| PER-1-R | AATTTGGGCTTAGGGCAGAA | |||
| VEB-1-F | CGACTTCCATTTCCCGATGC | 55 | 643 | (19) |
| VEB-1-R | GGACTCTGCAACAAATACGC | |||
| OXA-F | ACACAATACATATCAACTTCGC | 60 | 813 | (20) |
| OXA-R | AGTGTGTTTAGAATGGTGATC |
The PCR experiments were carried out in reaction mixture volume of 25 μL, containing 10 pM of each primer, 0.2 mM of dNTPs, 0.7 mM of MgCl2, 1 μL of crude DNA template (including the positive controls), and 1 U of Taq polymerase in the reaction buffer, provided by the manufacturer (CinnaGen, Tehran, Iran). Amplifications were carried out in a thermocycler (Techne TC-3000G, UK), using the following program: 5 minutes of denaturation at 94°C, followed by 30 cycles of 95°C for 1 minute, annealing temperature for 1 minute, and 72°C for 1 minute, with a final extension at 72°C for 10 minutes.
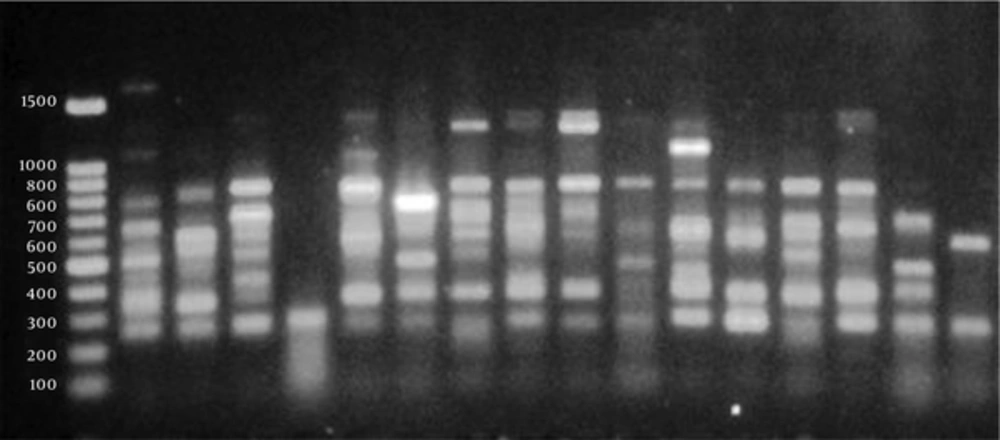
The PCR products were separated via electrophoresis on 1.2% Agarose gel, stained with RedSafe (Intron Bio, Korea), and visualized using an image analysis system (UVLtec, St John’s innovation centre). The nucleotide sequences were analyzed by searching the GenBank using BLAST (http://www.ncbi.nlm.nih.gov/blast/).
3.6. RAPD-PCR Fingerprinting
The clonality of K. pneumoniae isolates was determined, using random amplified polymorphic DNA (RAPD)-PCR fingerprinting. The PCR reaction mixture contained 1 µM of primer 640 (5’- CGTGGGGCCT-3’; Takapouzist, Tehran, Iran), 1 U of Taq DNA polymerase, 4 mM of MgCl2, 0.4 mM of dNTPs (CinnaGen, Tehran, Iran), and 90 ng of DNA template in a final volume of 25 µL (21).
The following cycling program was applied using a Bioer TC25/HH thermal cycler (Bioer Technology, China): 5 minutes of denaturation at 94°C, followed by 45 cycles of 30 seconds at 94°C, 30 seconds of annealing at 34°C, 30 seconds of extension at 7°C, and a final extension for 10 minutes at 72°C. The PCR products were separated via gel electrophoresis and then stained and visualized as mentioned before. The RAPD-PCR of polymorphic DNA fingerprints was analyzed using NTSYSpc version 2.02 (Setauket, NY, USA).3.7. Statistical Analysis
Chi square test was used for the comparison of antibiotic resistance profiles. Nonparametric two-tailed Mann-Whitney U and Kruskal-Wallis tests were used for comparing the differences in age, gender, and colonization, using SPSS version 20. P value ≤ 0.05 was considered significant.
4. Results
4.1. Antimicrobial Susceptibility
The antibiotic resistance profiles of the isolates are shown in Table 2. Overall, the resistance rates were as follows: ticarcillin (93.8%); ceftazidime, cefotaxime, and carbenicillin (75%); ceftriaxone and piperacillin (68.8%); aztreonam and cotrimoxazole (62.5%); tobramycin (50%); cefepime and co-amoxiclav (37.5%); amikacin and ampicillin-sulbactam (31.3%); cefoxitin and ciprofloxacin (18.8%); and ertapenem (6.3%). All the isolates were susceptible to imipenem, meropenem, and piperacillin/tazobactam. The MIC for ceftazidime ranged from 2 to 128 µg/mL (Table 2).
| Isolate and Gender | Age (Months) | Antibiotic Resistance Profile | CAZ MIC (µg/mL) | PCT | β-lactamase Type | RAPD Cluster |
|---|---|---|---|---|---|---|
| 1 M | 96 | CAZ, FOX, CTX, TC, AUG, TS | 16 | - | None | 1 |
| 2 M | 84 | CAZ, CRO, FOX, CTX, ETP, ATM, PY, TC | 16 | - | None | 2 |
| 3 M | 48 | PY, TC | 4 | - | None | 3 |
| 4 F | 8 | AUG, SAM, TS | 4 | - | None | 2 |
| 5 M | 12 | CAZ, CRO, CTX, CPM, ATM, PRL, PY, TC, AK, TN, CIP, AUG, SAM | 16 | - | None | 4 |
| 6 M | 60 | CAZ, FOX, CPM, TC, SAM, AUG, TS | 16 | - | None | 2 |
| 7 M | 7 | PRL, PY, TC, TS | 2 | + | TEM-1; SHV-11 | 4 |
| 8 M | 24 | CAZ, CRO, CTX, CPM, ATM, PRL, PY, TC, AK, TN, TS | 128 | + | TEM-1; SHV-11; CTX-M-15; OXA-1 | 4 |
| 9 M | 11 | CAZ, CRO, CTX, ATM, PRL, PY, TC, AK, TN, SAM, TS | 128 | + | TEM-1; SHV-11; CTX-M-15; OXA-1 | 4 |
| 10 M | 8 | CRO, CTX, ATM, PRL, PY, TC, TN, TS | 32 | + | TEM-1; SHV-11; CTX-M-15; OXA-1 | 2 |
| 11 F | 18 | CAZ, CRO, CTX, ATM, PRL, PY, TC, TS | 16 | + | TEM-1; SHV-11 | 2 |
| 12 M | 24 | CAZ, CRO, CTX, ATM, PRL, PY, TC, AK, TN, CIP | 32 | + | TEM-1; SHV-11; CTX-M-15; OXA-1 | 4 |
| 13 M | 24 | CAZ, CRO, CTX, PRL, PY, TC, TN, TS | 16 | + | TEM-1; CTX-M-15 | 3 |
| 14 M | 6 | CAZ, CRO, CTX, CPM, ATM, PRL, PY, TC, TN, AUG | 128 | + | TEM-1; SHV-11; CTX-M-15; OXA-1 | 4 |
| 15 M | 36 | CAZ, CRO, CTX, CPM, ATM, PY, PRL, TC, AK, TN, CIP, AUG, SAM | 16 | + | TEM-1; CTX-M-15, OXA-1 | 2 |
| 16 F | 36 | CAZ, CRO, CTX, CPM, ATM, PRL, TC, TS | 4 | + | TEM-1 | 2 |
Abbreviations: AK, amikacin; ATM, aztreonam; AUG, co-amoxiclav; CAZ, ceftazidime; CIP, ciprofloxacin; CPM, cefepime; CRO, ceftriaxone; CTX, cefotaxime; ETP, ertapenem; F, female; FOX, cefoxitin; M, male; PCT, phenotypic confirmatory test; PRL, piperacillin; PY, carbenicillin; SAM, ampicillin-sulbactam; TC, ticarcillin; TN, tobramycin; TS, cotrimoxazole.
4.2. ESBL Production
The phenotypic test results showed ESBL production in 10 out of 16 K. pneumoniae CF isolates (62.5%). The type of produced β-lactamase, as well as the antibiotic resistance profile of the isolates, is shown in Table 2. No significant differences were observed in the antibiotic resistance profiles between ESBL-producing and non-ESBL strains. As shown in Table 2, ten ESBL-positive strains carried blaTEM-1 gene, one of which harbored blaTEM-1 alone and 2 harbored blaTEM-1 plus blaSHV-11 (both non-ESBL β-lactamases). The remaining 7 isolates harbored the ESBL-encoding blaCTX-M-15 gene, five of which also harbored blaSHV-11, blaTEM-1, and blaOXA-1 genes; also, one isolate carried blaTEM-1 and one harbored blaTEM-1 plus blaOXA-1 genes. However, PER-1 and VEB-1 β-lactamase genes were not detected. Production of ESBL by K. pneumoniae isolates was only observed among patients under 3 years, compared to older patients (Kruskal-Wallis test, P < 0.05).
4.3. RAPD-PCR Fingerprinting
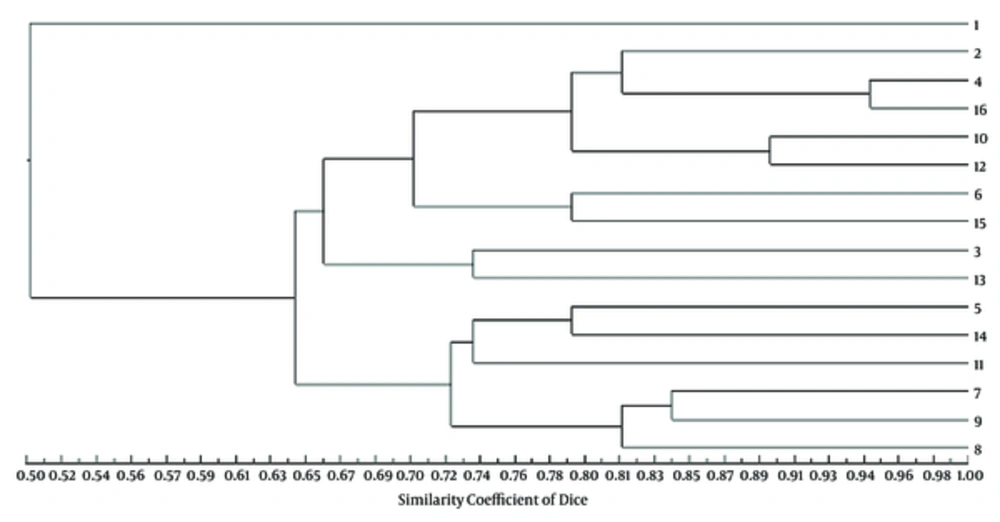
The DNA profiles generated by RAPD-PCR showed 19 bands within the range of 200 - 2000 bp (Figure 1). Four major clusters were observed at a similarity level of 70%, and 14 different groups were at a similarity level of 85%, indicating heterogeneity among the isolates (Figure 2). Among 10 ESBL phenotypes, four belonged to cluster 2 (isolates 10, 11, 15, and 16), one belonged to cluster 3 (isolate 13), and five were found in cluster 4 (isolates 7, 8, 9, 11, and 14). Among ESBL-negative isolates, one belonged to cluster 1 (isolate 1), three were in cluster 2 (isolates 2, 4, and 6), and one (isolate 5) was in cluster 4. The results showed no correlation between β-lactamase/ESBL production and specific genetic fingerprints of the isolates.
5. Discussion
Members of the Enterobacteriaceae family are considered as transient respiratory colonizers, which are not associated with severe diseases in CF patients. Limited reports have shown K. pneumoniae isolation from CF patients in Iran and other countries (6-9). In this regard, a study on 129 Iranian pediatric CF patients showed that 11.6% of the isolates were K. pneumoniae, although ESBL production by the isolates was not investigated (7).
Another study performed in Iran showed that 4% of Gram-negative bacterial isolates from 52 CF patients were K. pneumoniae, none of which produced ESBL (6). In the present study, we found that 16 out of 98 CF isolates were K. pneumoniae (16.3%), 10 of which showed the ESBL phenotype. However, 7 strains harbored the ESBL blaCTX-M-15 gene, along with non-ESBL β-lactamase genes. In this regard, Canton et al. reported colonization in a CF patient with a K. pneumoniae strain, capable of producing β-lactamase SHV-5 gene (22). Moreover, Pollini et al. showed that CTX-M group I was predominant in ESBL-producing Escherichia coli and K. pneumoniae isolates from CF patients (8). Leao et al. also reported 2 cases of K. pneumoniae carbapenemase 2-producing isolates from CF patients (4).
Emergence of OXA β-lactamases in the Enterobacteriaceae family, particularly Klebsiella species, is of major significance, as these bacteria are true pathogens capable of infecting immunocompromised individuals. The first sequenced blaOXA-1 gene was part of a transposon, carried by plasmid RGN238 (14). Coproduction of CTX-M and OXA enzymes by K. pneumoniae enhances resistance to β-lactamase inhibitors, presumably explaining their nonsusceptibility to amoxicillin/clavulanate (23). All our ESBL-positive isolates carried the non-ESBL TEM-1 gene, and the majority harbored the SHV-11 gene along with ESBL blaCTX-M-15 gene.
TEM-1 hydrolyzes ampicillin at a greater rate than carbenicillin, oxacillin, or cephalothin and shows negligible activity against extended-spectrum cephalosporins (24). Also, K. pneumoniae strains naturally possess a single chromosomal copy of blaSHV-1 or its derivative blaSHV-11 (25). We showed that 6 isolates carried blaSHV-11, 5 of which also harbored blaTEM-1, blaCTX-M-15, and blaOXA-1 genes. Finally, using RAPD-PCR, we found distinct heterogeneity among K. pneumoniae isolates (both non-ESBL and ESBL producers), and no association was observed between ESBL production and genetic fingerprints.
Colonization of CF patients by multidrug-resistant ESBL-producing K. pneumoniae, whether transient or not, can be important in the development of chronic airway inflammation and pulmonary exacerbation. Since β-lactam antibiotics (eg, ceftazidime and cefepime), used for the treatment of CF airway infections, are ineffective against ESBL-producing Enterobacteriaceae, persistence of these pathogens can complicate the diagnostic procedures and majorly influence the selection of therapeutic regimens.
6. Conclusion
The results of the present research are alarming, as multidrug-resistant pathogens can provide a reservoir for dissemination of drug-resistant genes among other bacterial species.