1. Background
Infertility is a disease of the reproductive system that affects one in seven couples all around the world. Thirty percent of all infertility cases are due to the malefactor, half of which have unknown causes; therefore, it is a great concern in public health (1). Urinary tract infection is one of the most important causes of infertility in males (2). According to the literature and World Health Organization (WHO) guidelines for semen analysis (2010), infection in infertile males is associated with changes in semen parameters (sperm count, sperm motility, or sperm morphology) and increased leukocytes count (3, 4). In general, the genital tract is prone to the growth of pathogenic and non-pathogenic microorganisms. Numerous studies have shown that semen bacterial infections may adversely affect male reproductive health, which in turn leads to the reduction of male fertility (1, 5).
Chlamydia trachomatis is a Gram-negative bacterium that is an obligate intracellular pathogen (6). Chlamydia infection is the most common infection in sexually transmitted diseases, affecting millions of men and women annually (7). According to the WHO report (2019), nearly 127 million cases of C. trachomatis genital infection occur each year. Meanwhile, it should be noted that about 50% of males and 75% of females infected with these bacteria are asymptomatic (8). Chlamydia has two forms of development: the elementary body, that is infectious but non-replicating and extracellular; and the reticulate body, that is non-infectious but replicating and intracellular (9). In most of the cases, such infections transmit from males to females through sexual intercourse, via free chlamydial elementary bodies in seminal plasma or the infected epithelial cells in the male genital tract. Untreated infections of C. trachomatis in males cause urethritis, prostatitis, epididymitis, and epididymo-orchitis and in females cause severe reproductive complications such as pelvic inflammatory disease, chronic pelvic pain, ectopic pregnancy, miscarriage, and tubal infertility (10, 11). Detecting asymptomatic infections in the male genital tract is very difficult. Cell culture cannot be used for C. trachomatis, and immunological techniques are also less sensitive, but the molecular method might be applicable (12).
Listeria monocytogenes is a Gram-positive bacillus, without spores. This bacterium is an intracellular and opportunistic pathogen that its role in human infections caused by food is well-studied. Over the past few decades, L. monocytogenes is considered as a pathogen that has caused numerous infections in Western countries and recently in African regions, which its pathogenic properties are studied along with simultaneous advances in biological sciences, particularly molecular biology (13). The identification of L. monocytogenes via molecular techniques dates back to the 1990s (14). Listeria monocytogenes causes neurological complications, and septicemia, as well as negatively affects the genital organs. Besides, it brings about abortion, premature birth, meningitis, and meningocele inflammation.
These problems can affect especially those patients with a background disease or severe conditions, and other high-risk individuals such as pregnant women, and unborn or newborn babies and immunocompromised persons. The incidence of listeriosis among pregnant females with a history of spontaneous abortion is 12 per 100,000, while its prevalence is reported as 0.7 per 100,000 in healthy individuals, and about one-third of reported human listeriosis cases are associated with recurrent abortion (15). Listeria monocytogenes can cause inflammation in the placenta or amniotic sac, fetal infection, and even abortion or stillbirth as the bacteria can cross the placenta barrier (16).
According to the previous studies, the semen analysis is the most reliable test to screen infertile males (1); the use of PCR is also recommended as it is highly sensitive and specific to C. trachomatis in infertile males and L. monocytogenes in females with recurrent abortion (13, 17-19). Detection of C. trachomatis in infertile males is associated with various challenges, but PCR can detect the bacteria in eight hours. For diagnosis of Listeria, although routine microbiological techniques are still common, but PCR can be helpful to diagnose this infection. Evidence about the association between infections with C. trachomatis and L. monocytogenes and male infertility can help promote public health; In this line, several studies in developed countries concluded that infection with these bacteria may lead to infertility (20, 21).
2. Objectives
The current study aimed to investigate the prevalence of C. trachomatis and L. monocytogenes in asymptomatic male patients with infertility using PCR, and to examine their effects on semen parameters, which can play an important role in the ART program (Assisted Reproductive Technique). Since the detection of L. monocytogenes was previously being conducted only in females with recurrent abortion, identification of these bacteria using PCR, as a sensitive technique for microbial screening of males with infertility, seems to be essential.
3. Methods
3.1. Clinical Samples
In total, 100 infertile males who were referred to the Infertility Clinic of the Sarem Women’s Hospital (Tehran, Iran) for infertility from December 2016 to June 2017 were included in the current study. First, the medical, sexual, and social profile and history of all participants were obtained and reviewed by a specialist physician. It was found that none of them had symptoms of urogenital tract infection (i.e., they were all healthy). The mean age of patients was 34.84 years (ranging from 21to 60). Semen specimens of these cases were collected in sterile sample cups, with a sexual abstinence duration of at least 48 hours and no antibiotic consumption for up to one week. These containers had no cytotoxic effects on human spermatozoa.
3.2. Semen Analysis
The semen analysis was carried out based on the WHO’s manual (3) for the determination of variables such as volume, sperm count, sperm motility, sperm morphology, and leukocyte count. The results were verified against the WHO criteria (2010), to check whether the sample is normal or abnormal.
3.3. Identification of Chlamydia trachomatis and Listeria monocytogenes in Semen Samples by PCR Test
Semen samples that their leukocyte level was higher than normal were kept under standard conditions at -20°C until assayed by the PCR test. The specimens were maintained in the freezing temperature in the special sample boxes, filled with ice, and were transferred from the hospital to the Iranian Gene Fanavar Institute (IGF) to perform PCR assay. The DNA extraction was performed on samples by using a standard phenol-chloroform method according to the following protocol (22).
The First 100 μL of the semen sample was transferred to a 1.5 microliter microtube, and distilled water was added so that the volume reached 500 μL. The same volume (500 μL) of phenol was added, then briefly vortexed for 1 minute, 10 times inverted, and then centrifuged at 13500 g for 5 minutes. The supernatant phase was slowly separated by a 100 μL sampler (without mixing with a low phase) and transferred to a new tube. The same volume (about 400 μL) of chloroform was added, 10 times inverted, and then centrifugation at 13500 g for 5 minutes. The supernatant fluid was slowly transferred to a new tube by a 100 μL sampler. 20 μL of 5-molar sodium acetate was added to the solution, and then 750 μL of absolute cold ethanol was added, 10 times inverted, and finally centrifuged for 10 minutes at 13500 g. The supernatant fluid was drained and 1000 μL of cold-70%-ethanol was added and 10 times inverted, and then centrifuged at 13500 g for 10 minutes. The supernatant fluid was drained and placed in a 65°C block heater (open door tube) to dry out. An amount of 40 microliters TE buffer was added and shorthand vortex blows dissolved the patient’s DNA, which was finally incubated at 65°C for 5 - 10 minutes. The final DNA solution was stored at -20°C to be used at the proper time.
The sequence primers used to detect the genome of the C. trachomatis bacterium for amplification of the region of 517 bp of the 16S rRNA gene (23), as well as those used to detect the L. monocytogenes genome, to amplify the area of 226 bp of the 16SrRNA gene of this bacterium (24), are described in Table 1. Steps conducted to optimize the PCR assay are described in the following (22). To carry out the PCR test, the Taq DNA Polymerase (1.5 units per μL), MgCl2 (1.5 mM), dNTP (0.2 mM), and primers (0.2 μM) were used.
| Species | Product Size(bp) | Target Gene | Primer Sequence (5’ - 3’) | Reference |
|---|---|---|---|---|
| Chlamydia trachomatis | 517 bp | 16S rRNA | Forward Primer: 5’-GGACCAAATCGTATCTCGG-3’ | (23) |
| Reverse Primer: 5’-GAAACCAACTCTACGCTG-3’ | ||||
| Listeria monocytogenes | 226 bp | 16S rRNA | Forward Primer: 5’-TGTTAATGAACCTACAGGACCTTC -3’ | (24) |
| Reverse Primer: 5’-TAGTTCTACATCACCTGAGACAGA-3’ |
3.4. Limited of Detection
The PCR test was optimized according to the standard strain to detect C. trachomatis and L. monocytogenes, and then specificity and limit of detection were evaluated. To determine the limit of detection of the optimized PCR assay and to specify the minimum number of C. trachomatis and L. monocytogenes provided dilutions of DNA with a specific number separately. Then, the PCR test of each obtained dilution was performed. The amount of limit of detection optimized PCR test on C. trachomatis and L. monocytogenes was determined separately, due to the concentration of DNA in the first tube and using the Genome Copy Number (GCN). Then, the samples were placed in the thermocycler unit (MyGene Brand MG96G model), which was according to the temperature and schedule, to achieve the desired DNA gene amplification.
The thermal protocol for amplifying the desired genes (DNA amplification) in relation to C. trachomatis and L. monocytogenes was carried out under the following conditions: an initial denaturation cycle at 94ºC for 5 minutes for C. trachomatis and 3 minutes for L. monocytogenes, followed by 40 cycles for C. trachomatis and 45 cycles for L. monocytogenes of denaturation at 94ºC for 30 seconds, annealing at 54ºC for 45 seconds for C. trachomatis and at 62ºC for 30 seconds, and extension at 72ºC for 1 minute, with a final extension cycle at 72ºC for 5 minutes for C. trachomatis and at 7 minutes for L. monocytogenes. Positive controls were C. trachomatis and L. monocytogenes and negative control was distilled water (22).
3.5. Electrophoresis technique and the detection of the PCR Product
The amplified products were added to 1.5% agarose gel and then were electrophoresed (22). PCR yields were detected and DNA bands were photographed by UV-transilluminator (Spectroline Brand).
3.6. Statistical Analysis
Statistical analyses to assess the agreement between the detection of C. trachomatis and L. monocytogenes in semen specimens were performed by SPSS version 24. Semen parameters such as volume, sperm count, sperm motility, and sperm morphology of all samples (with or without C. trachomatis) were compared. Besides, the same was performed for semen specimens with or without L. monocytogenes. The statistical significance of the difference between age groups, and the differences between the groups of asthenoteratoleukocytospermia, teratoleukocytoseprmia, and asthenoteratooligoleukocytospermia were evaluated for the presence of bacteria
4. Results
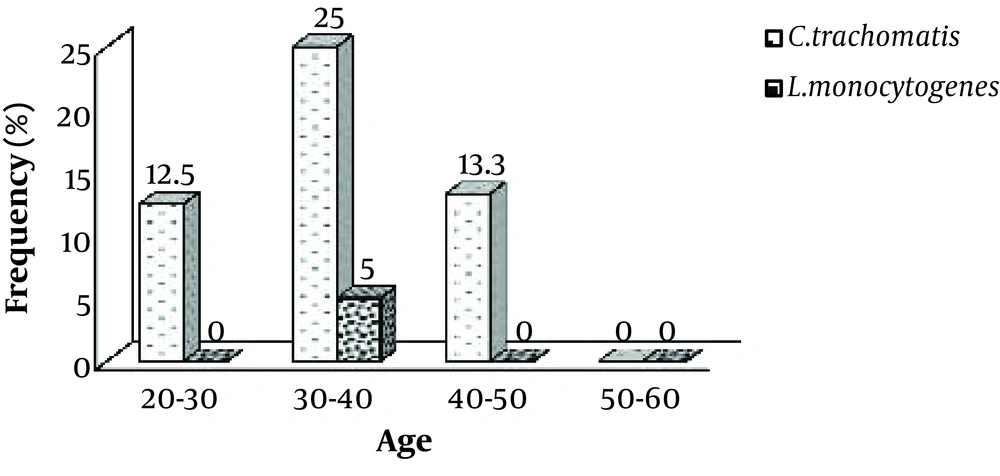
The minimum and maximum age of infertile men was 21 and 60 years, respectively. The mean age was 34.84 years. For those subjects who had C. trachomatis in their samples, the mean age was 35.25 ± 1.28 years. For those who were negative for C. trachomatis, the mean age was 34.44 ± 0.75 years (P = 0.75). For those who had L.monocytogenes in their specimens, the mean age was 36.33 ± 1.33 years. For those who were negative for L.monocytogenes, the mean age was 34.39 ± 0.67 years (P = 0.68). The prevalence of both bacteria was higher in infertile men aged 30 to 40 years than in other age groups (Figure 1).
4.1. Identification of Chlamydia trachomatis and Listeria monocytogenes DNA in Semen Samples by PCR Assay
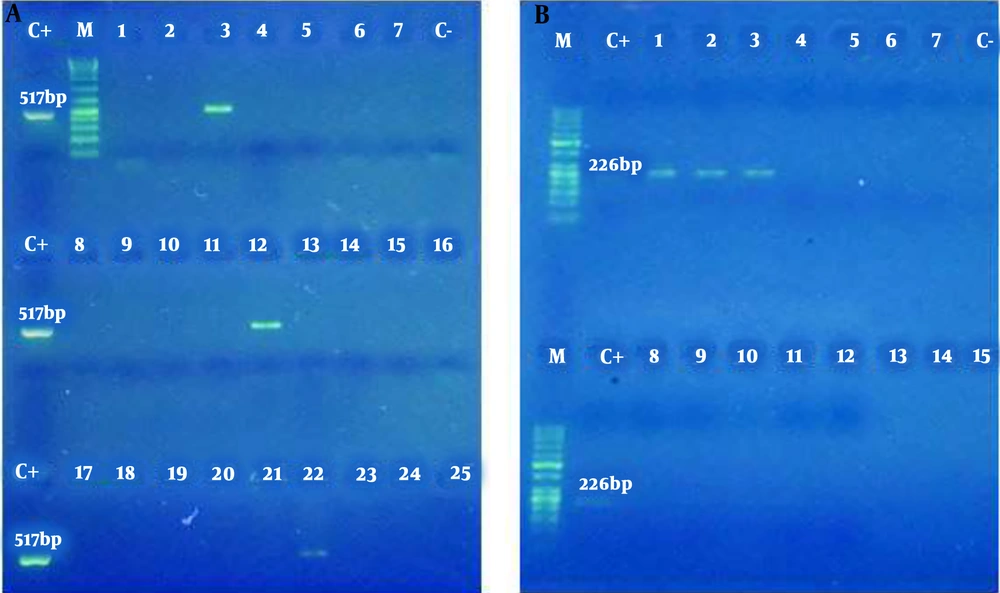
Limit of detection of PCR test of C. trachomatis was shown 100 copy/reaction. The same procedure was performed for L. monocytogenes. Out of 100 semen specimens of infertile men, 20 (20%) were positive for C. trachomatis, 3 (3%) were positive for L. monocytogenes (Figure 2), and 3 (3%) were positive for both bacteria (were co-infected).
The amplification product of Chlamydia trachomatis obtained from semen samples of infertile men. M: Size Marker 1 Kb DNA Ladder (Bioflux); C+: Positive Control; C-: Negative Control; Positive Samples: 3, 12, 22; Negative Samples: 1, 2, 4 - 11, 13 - 21, 23 – 25 (A). Amplification product of Listeria monocytogenes obtained from semen samples in infertile men. M: Size Marker 1 Kb DNA Ladder (Bioflux); C+: Positive Control; C-: Negative Control; Positive Samples: 1, 2, 3; Negative Samples: 4 – 15 (B).
4.2. Chlamydia trachomatis and Listeria monocytogenes Infections and Semen Quality
Sperm analysis revealed that all samples had abnormal results, at least for one parameter (leukocytes count, sperm count, sperm motility, or sperm morphology). According to the results of the spermiogram, for all samples leukocyte count was higher than normal (0-1 Mil/mL). The standard semen analysis (conducted following the World Health Organization Guideline (2010)) showed that only 3 (3%) of the specimens were azoospermic. The remaining samples were nonazoospermic (97%). The presence of C. trachomatis DNA in semen specimens of infertile men was significantly higher in azoospermic patients compared to nonazoospermic ones (66.67% vs. 18.26% respectively, P < 0.05). Also, the presence of L. monocytogenes DNA was considerably higher in nonazoospermic patients compared to azoospermic patients. There were no azoospermic patients infected with L. monocytogenes (P > 0.05).
Patients with parameters such as abnormal sperm motility and sperm morphology who were infected with Chlamydia and Listeria were categorized in the asthenoteratospermia group, which had no statistically significant difference with those who had Chlamydia and Listeria (P = 0.86 and P = 0.94 respectively). Patients who were only abnormal in morphology and were infected with Chlamydia and Listeria were categorized in the teratospermia group, which had no significant association with Chlamydia and Listeria (P = 0.14 and P = 0.94, respectively).
Patients who were abnormal regarding their sperm motility, sperm morphology, and sperm count and were infected with Chlamydia and Listeria were categorized in the asthenoteratooligospermia group, where there was no statistically significant difference between them and those who had coinfection of both bacteria (P = 0.76 and P = 0.14, respectively). A summary of the abnormal semen parameters of infertile azoo and nonazoospermic men with C. trachomatis and L. monocytogenes DNA is provided in Table 2. The mean values of volume of the sperm, sperm count, rapid progressive sperm motility, and sperm morphology in cases with and without C. trachomatis and L. monocytogenes DNA in semen specimens are described in Table 3.
| Pathogens | Azoospermic vs. Nonazoospermic Patients | Abnormalities in Seminal Fluid of Nonazoospermic Patients (N = 97) | ||||||
|---|---|---|---|---|---|---|---|---|
| Azoo (N = 3) | N.Azoo (N = 97) | Asthenoteratospermia | Teratospermia | Asthenoteratooligospermia | ||||
| Asthenoterato (N = 33) | N.Asthenoterato (N = 64) | Terato (N = 34) | N.Terato (N = 63) | Asthenoteratooligo (N = 9) | N. Asthenoteratooligo (N = 88) | |||
| Chlamydia trachomatis-positive | 2 (66.67) | 18 (18.56) | 6 (18.2) | 12 (18.8) | 9 (26.5) | 9 (14.3) | 2 (22.2) | 16 (18.18) |
| P Value | 0.04 | 0.946 | 0.141 | 0.766 | ||||
| Listeria monocytogenes-positive | 0 (0) | 3 (100) | 1 (3) | 2 (3.1) | 1 (2.9) | 2 (3.2) | 1 (11.1) | 2 (2.3) |
| P Value | 0.757 | 0.98 | 0.949 | 0.145 | ||||
Abbreviations: Asthenoteratospermia, abnormalities in sperm motility, sperm morphology; Asthenoteratooligospermia, abnormality in sperm motility, sperm morphology, sperm count; Teratospermia, abnormality in sperm morphology
aAll studied samples were reported to be higher than normal (0 - 1 Mil/mL) for leukocyte count
| Pathogens | Volume (mL) | Sperm count ×106 /mL | Total Progressive Motility, % | Normal Morphology, % |
|---|---|---|---|---|
| Chlamydia trachomatis | ||||
| Positive (n = 20) | 4.7 ± 1.89 | 37.05 ± 23.56 | 34.1 ± 24.11 | 12.7 ± 6.83 |
| Negative (n = 80) | 4.58 ± 1.62 | 44.33 ± 22 | 34.04 ± 24.97 | 14.36 ± 6.09 |
| P value | 0.766 | 0.195 | 0.992 | 0.289 |
| Listeria monocytogenes | ||||
| Positive (n = 3) | 5.17 ± 2.25 | 36.67 ± 26.31 | 18.67 ± 22.81 | 10.33 ± 4.73 |
| Negative (n = 97) | 4.58 ± 1.66 | 43.06 ± 22.39 | 34.53 ± 24.69 | 14.14 ± 6.27 |
| P value | 0.553 | 0.628 | 0.275 | 0.3 |
5. Discussion
According to WHO, infertility is a disease that affects the reproductive system and is defined as the failure to achieve a clinical pregnancy after having 12 months (or more) of regular unprotected sexual intercourse (1). One of the crucial causes of male infertility is urogenital infections and it is estimated that about 15% of male infertilities are caused by such infections (25). Chlamydia trachomatis and L. monocytogenes are among the serious pathogens that cause sexually transmitted diseases all around the world (26). In most cases, the infection is asymptomatic, so that about 70% to 80% of infected females and 50% of infected males present no symptom of infection (27). Acute chlamydial infection in males causes urethritis, epididymitis, and prostatitis. Epididymitis is an inflammation that can cause infertility by preventing sperms from getting into the ejaculate, particularly when both testicles are involved (28). Studies showed that C. trachomatis can bind to sperm cell surfaces through its lipopolysaccharides and induce apoptosis (29). Infection with C. trachomatis directly affects semen parameters or indirectly changes the sperm quality via tissue inflammation, and ultimately causes male fertility (30).
Listeria monocytogenes, an intracellular and opportunistic pathogen, enter the body through contaminated water and food and cause a systemic infection called listeriosis, which in turn causes meningoencephalitis in pregnant women, the elderly, and those with immunodeficiency (13). The clinical spectrum of listeriosis contains two specific targets; the placental-maternal unit and the central nervous system (15). Listeriosis in pregnant women is asymptomatic or has mild flu-like symptoms. Besides, after crossing the blood-brain barrier, the bacteria causes neonatal septicemia and meningitis, abortion, and preterm labor. Listeria monocytogenes phospholipases play an essential role in its pathogenesis (31).
According to the literature, results about the effects of Chlamydia infection on semen parameters are controversial. Moazenchi and colleagues showed that 11% of those infected with C. trachomatis had sperm abnormalities (32). In the current study, only azoospermic specimens infected with C. trachomatis had a significant association with sperm abnormalities (P < 0.05), and no significant association with nonazoospermic samples was found (P > 0.05). The study conducted by Gdoura and colleagues on 104 asymptomatic male patients with infertility in Tunisia reported that the detection of C. trachomatis in semen specimens had no significant association with abnormalities in semen parameters (P > 0.05) (12). Zhu and colleagues reported a significant correlation between chronic chlamydial infection and infertility in males and females. In a five-year investigation (2010 to 2014), they found an increasing prevalence of C. trachomatis infection (by about 3.15%) and reported no significant effect of the infection on semen parameters (P > 0.05) (33).
In the present study, no significant association was observed between abnormalities in semen parameters and the detection of L. monocytogenes in the specimens (P > 0.05). To the best of knowledge of authors, no study is conducted thus far on L. monocytogenes and its bacteriospermia effects on males with infertility. Because PCR diagnostic method is the most sensitive test for the detection of C. trachomatis and L. monocytogenes, in the current study the presence of these two bacteria in semen specimens of infertile men was assessed using this method. In a study conducted by Samplaski and colleagues on infertile men using the PCR assay at Toronto’s Mount Sinai Hospital, the presence of C. trachomatis was estimated to be about 0.3% (34).
The prevalence of C. trachomatis reported in the aforementioned studies is lower than the current study. In a research done by Sellami and colleagues on the semen of infertile men using the PCR molecular method, about 15.2% of 85 specimens were positive for the presence of C. trachomatis (17). In a study conducted by Sadrpour and colleagues on 120 semen samples from infertile men in the Avicenna Infertility Treatment Center in Tehran (Iran), C. trachomatis was identified in 23.3% of the specimens (35). These results are consistent with the results of the current study. Hence, it seems that the screening of men to find out C. trachomatis infection, as one of the major causes of infertility, is of crucial importance.
Eslami and colleagues used the PCR method and reported that about 16% of 96 women with recurrent abortions were infected with L. monocytogenes (18). Goudarzi and colleagues detected seven cases of L. monocytogenes positive (among 87 vaginal swab specimens from women with abortion) using the PCR assay (36). In a molecular assessment performed by Le Monnier and colleagues to detect L. monocytogenes, molecular techniques, including PCR, were introduced as rapid and reliable assays to detect the bacteria in clinical samples (37). Lotfollahi and colleagues showed that 9 (out of 100) vaginal samples with a history of abortion, were detected to be infected with L. monocytogenes (38). These results were achieved on women with recurrent abortion with a similar method (PCR) that we used on infertile men for detecting L. monocytogenes. By comparing the results of the current study with those of other studies, it can be concluded that differences in the ratio of these bacteria can be attributed to factors such as the selection and gender of the patients, the number of samples under study, the use of different diagnostic methods, the difference in the geographical distribution of populations, the rate and severity of bacterial colonization in the urogenital tract, and the sensitivity of the primers used in the test as well as sample volumes.
5.1. Conclusions
According to the findings, accurate sperm analysis to diagnose changes in semen parameters, as well as the application of PCR as a rapid and sensitive technique to detect the bacteria, are essential for all infertile men, particularly for asymptomatic patients. Also, the promotion of public education and raising the awareness of males about sexual health and the necessity of preconception screenings can be great preventive steps against infertility.