1. Background
Breast cancer is the most common and emotionally traumatic type of cancer among women (1-3). More than 120,000 women in the UK and more than 40,000 women in the United States die from breast cancer every year. In Iran, breast cancer accounts for 25% of all cancers and is the most common cancer among women (3).
Numerous studies have shown that metabolic dysfunction, hormones, and inflammatory mechanisms affect the progression of breast cancer (4-7). Evidence suggests that stressors in various forms can trigger physical stimulation and changes in the immune system (8). Epidemiological studies have shown a connection between obesity and increased risk of developing different subtypes of breast cancer, including triple-negative breast cancer, a particularly aggressive form of breast cancer with poor outcome and few therapeutic options (8).
Depression and sleep disturbance occur in patients with breast cancer; 20% to 30% of breast cancer patients suffer from depression and anxiety, due to the negative consequences of a cancer diagnosis and the sequelae of cancer-related treatments (9). Also, chemotherapy followed by breast cancer can have a major impact on patients’ quality of life, disrupting their physical, mental, social and spiritual well-being, and in other words, reducing their quality of life indicators (10). The depression and anxiety symptoms can persist for several years, leading to adverse effects on the patient’s quality of life, compliance to medical treatment, recurrence, survival and also recovery from surgery during hospital stay (11). Metastatic breast cancer and its treatment have a major impact on the survivor’s quality of life (8-11). Following a diagnosis of breast cancer, an assessment of QOL is an important clinical outcome measure, because patients have to face major stressors that are likely to deteriorate their QOL, not only just after diagnosis but also in the long term (e.g., facing a life-threatening illness, painful and impairing treatments, significant role changes, and issues about body image) (8-11).
Exercise therapy has been studied in a variety of populations with cancer to counteract the side effects of treatments (12). Concurrent endurance and strength training are particularly relevant to improving overall health as they combine the benefits of each single exercise modality to bring about local. Therefore, aerobic resistance training is more effective than aerobic exercise in improving body composition, quality of life, and cardiovascular strength and fitness (12).
Hojan et al. (13) showed that mixed type physical activity (aerobic training, AT, and resistance training, RT) during endocrine therapy for breast cancer could prevent negative changes of the treatments, in body build in premenopausal women. Battaglini et al. (14) suggest that exercise with an emphasis on resistance training promotes positive changes in body composition and strength in breast cancer patients undergoing treatment. Sprod et al. (15) showed that four weeks of moderate home-based aerobic exercise and resistance training can be helpful for patients with breast cancer under radiotherapy and sleep quality was higher in the training group but not significantly different. Ohira et al. (16) showed that QOL scores improved in the training group after 6-months of weight-loss training in women who survived from breast cancer, however, the previous research literature on the effects of this type of exercise training on body composition improvement and quality of life indicators in postmenopausal women with cancer is limited.
2. Objectives
The present study aimed to determine the effects of eight weeks of concurrent training on body composition, quality of life, and sleep quality in postmenopausal women with breast cancer.
3. Methods
This study was conducted on 42 postmenopausal women from Kermanshah city with breast cancer were randomly selected and divided into the control group and the training groups. Entry requirements for subjects were determined by an oncologist. All of them were at stage one of the disease. They were diagnosed postmenopausal by a gynaecologist. The inclusion criteria were having at least 30 years of age at the start of adjuvant tamoxifen or Letrozole therapy, not having menstruation or having no exercises in the last 6-months. Before starting the training protocol, the purpose of the study was explained to the subjects. The control group did not perform a training program and they just did daily chores. They did not perform training protocols subsequently the written and oral consent of which was obtained from the subjects. This study was conducted based on ethical principles in the research of the Helsinki Statement. One week before starting the main training protocol, anthropometric indicators including age, weight, body mass index (BMI), waist-to-hip ratio (WHR), %fat, and waist circumference were measured.
The one-repetition maximum (1RM) test was measured from subjects by using the Brzysky method (Equation 1 indirects estimation formula for one repetition maximum).
3.1. Training Protocol
The training protocol included eight weeks of concurrent training, three-days per week. Each section included 10 minutes of warm-up and cool-down with 40% maxHR and the main part of the training was divided into resistance and aerobic training (17-19). The motion resistance training included barbell chest press, barbell curl, lying barbell triceps, behind-the-neck lat pulldowns and lower body movements, including smith machine squat, leg extensions, lying leg curls, and standing calf raises. The subjects had active rests between one to three minutes for each movement and set depending on weeks of the training protocol. Also, during aerobic training (walking and running), the heart rate of the subjects was evaluated by counting the pulse of the subjects in 15 seconds and calculated for one minute every 10 - 15 minutes. Also, the Borg RPE scale was used to control the intensity of aerobic training (12 - 14 degrees) (18, 19). The intensity and duration of each training section are shown in Table 1.
| Weeks | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Resistance training intensity | 2 (50/18) | 2 (55/16) | 2 (60/14) | 2 (60/14) | 3 (65/12) | 3 (65/12) | 3 (70/10) | 3 (70/10) |
| Resistance training time, min | 20 | 25 | 30 | 30 | 35 | 35 | 40 | 40 |
| Aerobic training intensity (max HR), % | 50 | 55 | 60 | 60 | 65 | 65 | 70 | 70 |
| Borg scale (aerobic training) | 12 | 12 | 12 | 13 | 13 | 13 | 14 | 14 |
| Aerobic training time, min | 20 | 25 | 30 | 30 | 35 | 35 | 40 | 40 |
Anthropometric characteristics were measured by using body composition, ZEUS 9.9. All measurements were carried out in the same conditions between 8 AM and 11 AM. The sleep quality was estimated using the Pittsburg sleep quality index (PSQI) questionnaire. It is a self-completed questionnaire evaluating the quality of sleep during the last month, and includes 19 questions in 7 parts (mental sleep quality, late sleep, sleep sufficiency, sleep period, sleep disorder, use of soporific drugs and defective function during the day). Each part can be scored from ‘0’ to ‘3’ The total score of PSQI ranges between ‘0’ and ‘21,’ with higher scores indicating the lower quality of sleep. A score higher than ‘5’ indicates low-quality sleep and many problems in at least two of three dimensions of this index (20). The quality of life was also measured by the McGill quality of life (MQOL) questionnaire. This questionnaire consists of 17 self-report items whose validity and reliability have been confirmed in cancer patients. This questionnaire includes three dimensions of quality of life, physical and psychological dimensions (21).
3.2. Statistical Analysis
To describe the characteristics of the subjects, the descriptive statistic was used. Kolmogorov-Smirnov test was used for data normality and Levene’s test was used for data homogeneity. Two-way repeated measures ANOVA was used to compare the difference between the variables. To examine significant differences, the Bonferroni test was used. The significance level was P < 0.05 in all tests. Finally, SPSS 24 was used for data analysis.
4. Results
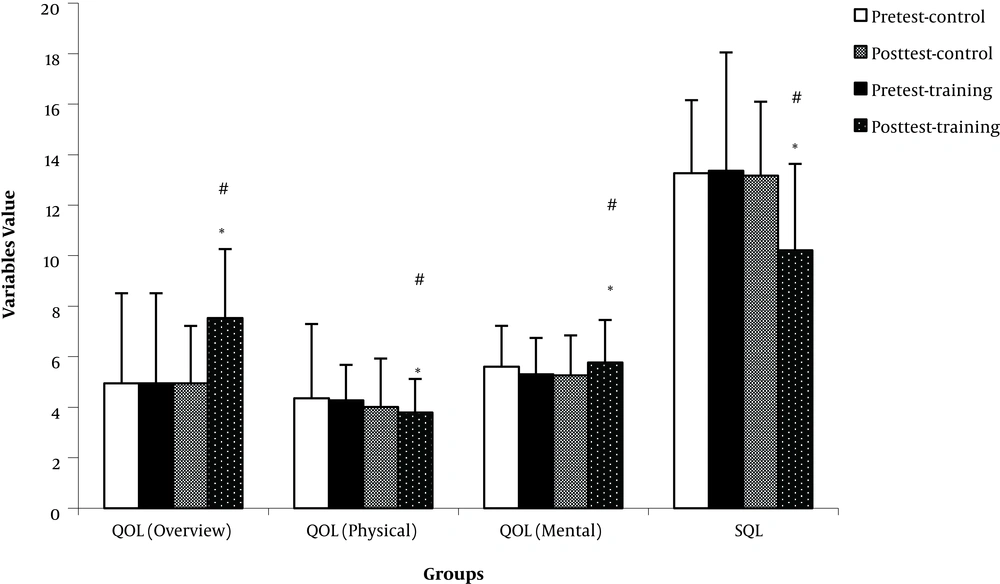
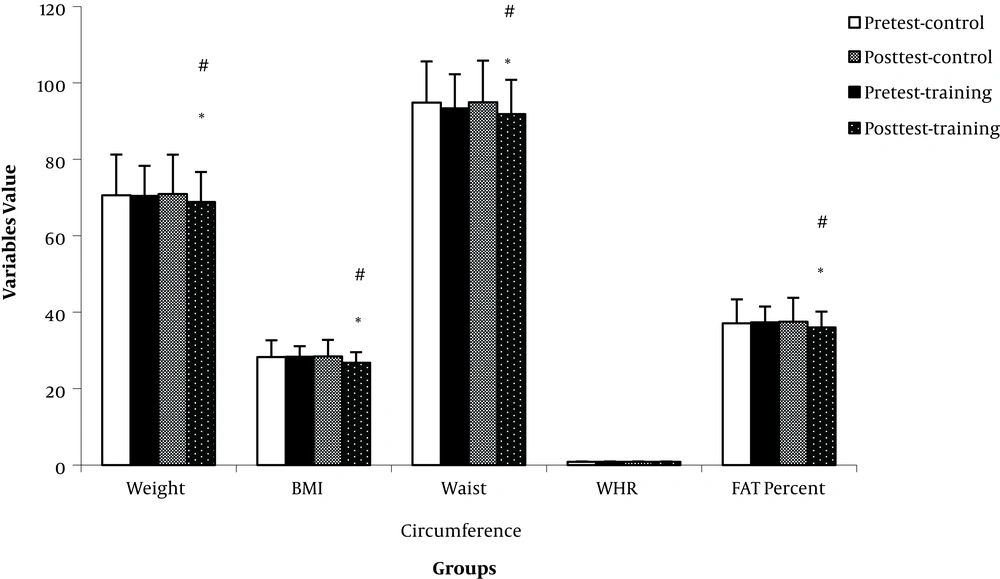
The results showed that after eight weeks of concurrent training in breast cancer patients, the score of sleep quality decreased significantly and the effect of time and group interaction was also significant (P = 0.007) in the training group. Also, the effect of this type of training on the quality of life in the training group was significant based on physical and mental dimensions, respectively (P = 0.012). The overall QOL increases significantly in the training group, and also, the effect of time and group interaction on QOL was significant (Figure 1) (P = 0.012). Besides, the results of weight, BMI, Fat percentage and waist circumference data showed that both variables were lower in the training group and these changes were significant Figure 2 (P < 0.05). The effect of time and group interaction in these variables were significant (P < 0.05). There wasn’t any significant change in WHR and the effect of time and group interaction on this indicator wasn’t significant (P > 0.05). These changes are shown in Table 2.
| Measurement Steps | ||||
|---|---|---|---|---|
| Control | Training | |||
| Pretest | Posttest | Pretest | Posttest | |
| SQL, score | 13.26 ± 0.70 | 12.38 ± 0.76 | 12.76 ± 1.00 | 9.47 ± 0.67b, c, d |
| QOL (physical), score | 4.35 ± 0.71 | 3.48 ± 0.58 | 4.2 ± 0.34 | 3.76 ± 0.33b, c, d |
| QOL (mental), score | 4.35 ± 0.39 | 4.01 ± 0.38 | 4.27 ± 0.35 | 3.78 ± 0.39b, c, d |
| QOL (overall), score | 4.94 ± 0.53 | 5.23 ± 0.55 | 4.52 ± 0.89 | 7.52 ± 0.67 b, d, e |
| Weight, kg | 70.59 ± 2.21 | 70.86 ± 2.14 | 70.13 ± 1.71 | 68.72 ± 1.70b, c, d |
| BMI, kg/m2 | 28.26 ± 0.89 | 43.28 ± 0.88 | 28.42 ± 0.55 | 26.78 ± 0.55b, c, d |
| WHR, m | 0.87 ± 0.01 | 0.88 ± 0.01 | 0.86 ± 0.01 | 0.87 ± 0.01e |
| Fat, % | 37.07 ± 1.31 | 37.53 ± 1.30 | 37.23 ± 0.89 | 35.99 ± 0.90b, c, d |
| Waist, m | 94.82 ± 2.25 | 94.21 ± 2.28 | 93.39 ± 1.89 | 92.04 ± 1.90b, c, d |
aValues are expressed as mean ± SD.
bSignificant difference with pretest.
cSignificant difference with the control group by interaction
dDecrease.
eIncrease.
5. Discussion
The present study aimed to determine the effects of eight weeks of concurrent training on body composition, sleep quality, and quality of life in postmenopausal women with breast cancer. We tried to find out; does concurrent training change body composition, sleep quality, and quality of life in postmenopausal women with breast cancer?
This study showed that after eight weeks of concurrent training, the sleep quality improved in the training group and its score decreased significantly (P < 0.05). Pooranfar et al. (22) examined the effect of 10-weeks concurrent training on the quality and quantity of sleep on renal transplant patients. Their finding showed that after 10-weeks of concurrent training, sleep quality increased by 27% and the duration increased by 30 minutes. This improves can lead to a state that the melatonin hormone secreted by the allergic pineal glands, which has a hypnotizing effect with central body temperature. Also, anabolic activity is stronger in sleep, whereas the catabolic activity is more intense during vigilance. Therefore, for a possible balance of energy, the body consumes more energy to relax and the body tends to more sleep. exercise training causes favourable changes in daily rhythms and elevated adenosine levels that improve sleep (22). Our findings agree with these results, but not agree with the findings of Sprod et al. (15). They found that four weeks of moderate aerobic and resistance training on patients with breast cancer under radiation therapy improved sleep quality in the training group, but this improvement hasn’t been significant (P = 0.05). Sleep quality and sleep mediators, as a result of concurrent training, can be adjusted by the H-P-A axis. Radiation therapy also can activate the signalling pathways that are associated with a short sleep period and sleep disorder (15).
We found that eight weeks of concurrent training can improve the overall score of QOL significantly in patients with breast cancer. It seems that concurrent training is more effective than aerobic and strength training alone on improving body composition, cardiovascular strength and fitness, and decreasing weight and eventually increasing the quality of life (17-19). Our finding is consistent with the results of Raghavendra et al. (17), and De Luca et al. (18). There is also evidence of positive effects of concurrent training on maximum output power, happiness, self-esteem, fatigue, and several subsets of QOL (17-19). Herrero et al. (19) studied the effect of 8-week concurrent training on body composition and quality of life in women survived from breast cancer. They concluded that combined cardiorespiratory and resistance training, even of very brief duration, improves the QOL and the overall physical fitness of women survived from breast cancer (19).
We found that eight weeks of concurrent training in patients with breast cancer decreased the weight, BMI, fat percent and waist circumference in the training group significantly (P < 0.05). Also, there was no significant change in WHR in the training group (P > 0.05). This could be due to our limitation in this study that the control of the subject’s nutrition was not possible during the training period and maybe more research on gender, numbers of subjects and duration of training protocols are necessary. Studies show that exercise training improves body composition and fitness while the findings of Matthews et al. (23) are different. They did not observe any significant change in weight, BMI, and body composition due to exercise training in women with breast cancer. This discrepancy seems to be due to the type, intensity, duration, and weeks of training, the type and age of the subjects, and the methods for measuring these indicators. Kim et al. (24) examined the effects of 12 weeks of walking on body composition and immune system in patients with breast cancer. This study was consistent with our findings regarding weight, fat percentage, and BMI (24).
In general, this study shows that concurrent training can improve body composition, QOL, and sleep quality in patients with breast cancer. Concurrent training with the effect on the H-P-A axis may be helpful to the treatment process, and leads to improvements in sleep quality and quality of life; and also causes improvements in body composition which lead to decrease in cancer progression (17-19). This finding suggested that concurrent training can be used in patients with breast cancer.