1. Background
Diazinon is an insecticide and pesticide, which is commonly used in agriculture for pest control, while it is extremely noxious for humans and animals (1, 2). Diazinon is available in organophosphate agrochemicals (3), and organophosphate compounds could be absorbed through the skin (4). Diazinon causes intoxication through direct inhalation, direct dermal contact, and the phosphorylate catalytic site of the cholinesterase, causing the inactivity of cholinesterase (5). In male rats, organophosphates exert toxic effects on the tissues and reproductive system. This chemical substance causes severe major degeneration and necrosis in the germ cells associated with tubular atrophy in the seminiferous tubules of the testis (6, 7). Several studies have revealed that in animals and human models, organophosphate pesticides and insecticides cause oxidative stress in the reproductive system (8, 9).
Selenium is an important trace element of humans and numerous animals and has antioxidant properties (10, 11). Evidence suggests that this compound acts on the glutathione peroxidase active site (12). GPx is considered the most important antioxidant enzyme system, which prevents cell damage (13).
Selenium also acts as a free radical modifier and plays a key role in the reduction of oxidative damage (14). Previous studies have indicated that selenium prevents the cardiac damage induced by free radicals (15). This compound is also involved in various physiological processes, such as ion passage through the cell membrane, coenzyme Q biosynthesis, and immune system stimulation (16). The protective role of selenium is attributed to proteins such as glutathione peroxidase and thyroxine reductase (14). To date, no proper investigations have been focused on in-vitro fertilization and zygote development after selenium administration in Wistar rats.
2. Objectives
The present study aimed to assess the testosterone levels, DNA maturity, sperm viability, sperm count, sperm maturation, in-vitro fertilization (IVF), hatching, and blastocyst with selenium administration in diazinon-treated Wistar rats.
3. Methods
3.1. Experimental Animals
This experimental study was conducted on 32 adult male Wistar rats. The body weight of each rat was approximately 200 grams. Prior to the experiment, the animals received no treatment for seven days for adaptation. All the standard conditions were controlled meticulously. The animals were fed pellets and tap water ad libitum and kept at room temperature (20-23°C) within a 12-hour light/dark cycle. The animals were divided into four groups of eight, including control, diazinon, selenium, and diazinon with selenium, and received daily oral treatment for 60 days. The control group received corn oil (0.2 mg/kg/B.W), the diazinon group received diazinon (300 mg/kg/B.W), the selenium group received selenium (0.5 mg Na2SeO3/kg of diet), and the diazinon with selenium group received diazinon (300 mg/kg/B.W) with selenium (0.5 mg Na2SeO3/kg of diet).
All the experiments were performed in accordance with the guidelines of the Ethics Committee for Research on Laboratory Animals of Uremia University (No. 50.201.509.240).
3.2. Testosterone Assay
After the treatment period, blood samples were collected from the animals directly from the heart, and serum samples were centrifuged at 3,000 rpm for 15 minutes; the serum samples were collected for hormonal analysis. Serum testosterone levels were determined by the radioimmunoassay method using the special kit for Wistar rats (WHO/Sigma Asso-RFGC-94/125). The limit of detection for testosterone was 0.12 ng/ml, and the inter-assay and intra-assay coefficients variances for testosterone were estimated at 9.9 (10 times) and 4.8 (10 times), respectively (17).
3.3. Sperm Parameters Assay
Testis epididymis was meticulously separated by 10-fold magnification via stereo zoom microscopy and divided into three parts of head, body, and tail. The epididymis tails were suspended in five milliliters of Ham’s F10 medium and incubated for 20 minutes. Following that, the epididymis was separated from the medium, and 10 drops were used for the assessment of sperm viability. Sperms with stained cytoplasm in the head, neck, and tail were considered as dead sperms, and eosin-nigrosin staining was performed for this purpose. In addition, spermatozoa with each cytoplasmic drop were designated as immature spermatozoa.
The caudal epididymal sperm was determined using a standard hemocytometer, and sperm motility was measured using the Olympus microscope at 10 fields and expressed as the mean of motile sperm based on the method proposed by the World Health Organization (WHO) (18).
3.4. DNA Double-strand Breaks Assay
In order to evaluate DNA damage (frank strand breaks), the standard comet assay was performed on the sperm samples (19) using the detection kit supplied by Sigma Chemical Co. in accordance with the instructions of the manufacturer. The objective device (×40) was attached to an epifluorescence microscope, which was used for the visualization of the DNA damage.
3.5. Ovulation and IVF Assay
After two days, superovulation in adult female rats was stimulated via the intraperitoneal injection of 10 insulin units (IUs) of pregnant mare serum gonadotropin and 10 IU of human chorionic gonadotropin (HCG). After 12-24 hours of HCG injection, the female rats were scarified by cervical dislocation, and the oviducts were extracted. The ampulla segment of the oviducts was placed inside a medium dish containing human tubal fluid (HTF) with bovine serum albumin (BSA; 4 mg/kg). At the next stage, the oocytes in the oviducts were removed and placed in the medium (4 mg/kg of BSA and HTF), and microdrops of sperms were collected in the medium. In total, 10-15 oocytes were placed in each microdrop of the fertile sperms (150 μl). The fertilization dish was incubated for 4-6 hours at the temperature of 37oC with 5% CO2.
For the embryo culture, the fertilized oocytes were transferred to fresh drops of the medium (4 mg/kg of BSA with HTF). Using mineral oil, all the medium droplets were covered, and the inverted microscope pronuclear of the fertilized oocytes and polar bodies were evaluated at 200× magnification. The zygote cells were washed three times with potassium simplex optimized medium (KSOM) after fertilization. Afterwards, the zygotes were transferred to fresh KSOM and cultured in the incubator for five days. After 24 hours of culture, the zygotes were evaluated. On day five, the blastocyst stage was estimated via phase contrast microscopy.
3.6. Statistical Analyses
Data analysis was performed in SPSS version 19.0 (SPSS Inc., Chicago, IL, USA), and the data were expressed as mean and standard deviation (SD). One-way analysis of variance (ANOVA) and Tukey's HSD were used as posttests, and the significance level was considered at P value < 0.05.
4. Results
4.1. Testosterone Levels
The serum analysis indicated that diazinon could significantly decrease the testosterone levels (P value < 0.05). However, the testosterone levels were higher in the animals co-administrated with selenium and diazinon compared to those receiving diazinon alone. No significant serological changes were observed in the testosterone levels of the control rats (Table 1).
| Control | Diazinon | Selenium | DZN + SE | |
|---|---|---|---|---|
| Sperm Count (106) | 54.07 ± 1.06 | 26.01 ± 1.11b | 52.08 ± 1.76 | 37.01 ± 1.07 |
| Sperm Motility, % | 90.05 ± 1.09 | 48.12 ± 2.08b | 89.33 ± 1.89 | 50.06 ± 2.09 |
| Sperm Immaturity, % | 13.08 ± 1.70 | 34.01 ± 3.01b | 13.71 ± 1.50 | 25.08 ± 2.60 |
| Sperm DNA Damage, % | 10.02 ± 1.12 | 34.01 ± 2.90b | 10.37 ± 1.22 | 31.01 ± 1.41 |
| Sperm Mortality, % | 17.40 ± 1.32 | 50.52 ± 1.71b | 17.34 ± 1.32 | 47.22 ± 1.90 |
| Testosterone | 4.61 ± 0.21 | 3.11 ± 0.10b | 4.22 ± 0.12 | 3.60 ± 0.12 |
Abbreviations: DZN: diazinon, SE, selenium.
aValues expressed as Mean ± SD; significance level set at P value < 0.05.
b Compared to controls.
4.2. Sperm Parameters
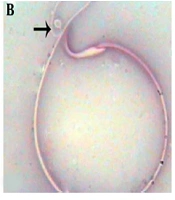
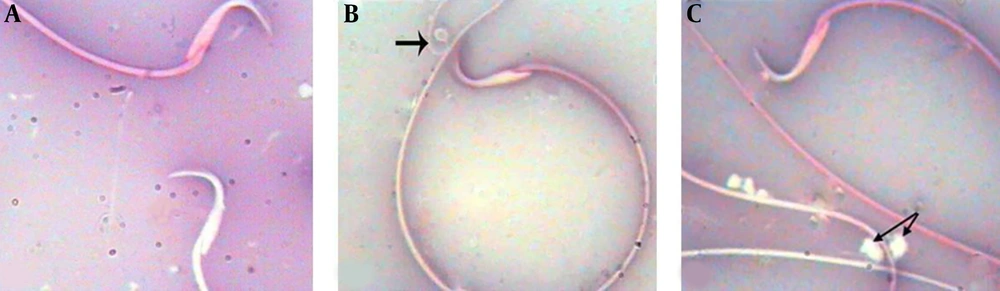
The results of light microscopic and eosin-nigrosin staining indicated that unusual and deformed sperms with high mortality rate significantly increased in the diazinon group (P value < 0.05). On the other hand, the animals receiving selenium with diazinon had a lower count of unusual and immature sperms with high mortality (P value < 0.05) (Table 1; Figure 1).
According to the results, the animals receiving diazinon showed significantly lower sperm count compared to the control animals (P value < 0.05). Furthermore, the comparison of the sperm count in the diazinon and diazinon with selenium groups indicated that the animals treated with selenium had significantly higher sperm counts (P value < 0.05). The findings showed no significant histopathological changes in the sperm evaluation of the control group. Table 1 shows the data on the sperm parameters.
4.3. Fertilized Oocytes
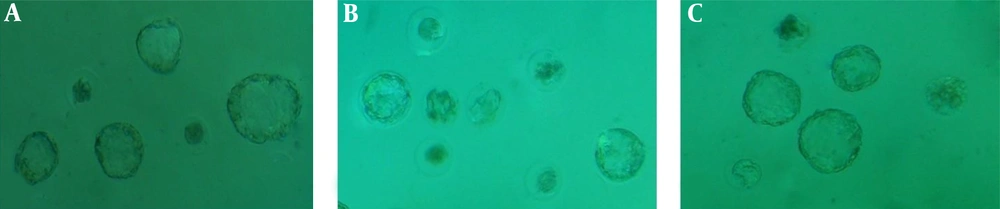
According to the obtained results, the IVF of the oocytes by the collected sperms from the diazinon group to the other embryonic development stages (percentage of the two-cell embryos, hatching, and blastocysts) was significantly lower than the controls (P value < 0.05), while the percentage of the fertilized oocytes, two-cell embryos, hatching, and blastocysts by the collected sperms from the diazinon group co-administrated with selenium had the significant highest value compared to the diazinon group. In addition, several disorders of the cleavage were observed in the zygote cell. In the diazinon group, several cell divisions were arrested, thereby stopping the cleavage (Figure 2). Table 2 shows the data on the fertilization rates.
Abbreviations: DZN: diazinon, SE, selenium.
aValues expressed as Mean ± SD; significance level set at P value < 0.05.
b Compared to controls.
cCompared to diazinon group.
5. Discussion
Diazinon is the most common toxin found in human and animal foods and water. Diazinon is an organophosphorus compound, which is widely used to control essential rice creams by farmers (20) despite its destructive effects on animal and human tissues. The previous findings in this regard have demonstrated that organophosphorus compounds such as diazinon produce free radicals that alter the cellular antioxidant systems and membrane lipid peroxidation (21). Under normal circumstances, there is a balance between the production and elimination of free radicals. Imbalance in these processes causes oxidative stress, which may induce substantial cell damage if it is severe or prolonged (22). On the other hand, diazinon is an insecticide that acts through the inhibition of acetylcholinesterase (AChE) (23). AChE is most commonly known for its role in the termination of cholinergic signaling through the hydrolysis of acetylcholine to choline and acetate.
Selenium is an essential trace element, which plays a key role in antioxidant selenoproteins for protection against the oxidative stress caused by reactive oxygen species and reactive nitrogen species. Therefore, selenium protects the cell membrane from the free radical damage caused by chemical toxins (14). Due to the vast effects of diazinon on animal and human health, its detrimental impact on various human organs requires further investigations. The present study aimed to determine the protective effects of selenium on the DNA damage of sperms, IVF, and early embryonic development in diazinon-treated male Wistar rats.
The hormonal analyses in the present study indicated the reduction of the testosterone levels in the diazinon-treated animals. Therefore, it could be assumed that exposure to diazinon leads to the destruction of leydig cells, which in turn decreases the serum testosterone levels. In contrast, the animals that were co-administered with diazinon and selenium had higher serum testosterone levels, which resulted in the increased number of the leydig cells in one square millimeters of the interstitial tissue. Destruction by the apoptosis of primary spermatogenic cells is another property of the degenerative effects induced by diazinon, which is in line with the previous studies in this regard, suggesting a mechanism for the regulation of spermatogenesis in the mammalian testis (24). However, the findings of Hamza and Diab (2020) demonstrated that selenium nanoparticles could inhibit testicular injury and improve the antioxidant state in males (25).
According to the results of the present study, the sperm count and sperm viability in the diazinon group decreased more significantly compared to the control group, which could be attributed to the disorder in the cell differentiation in the testis, as well as the detrimental effects of diazinon on the spermatogonial stem cells. However, diazinon co-administration with selenium significantly increased both these parameters as opposed to the diazinon groups since selenium has antioxidant activities, which has also been reported in the previous studies in this regard (13).
The current research showed that the percentage of the immature sperms and sperm DNA damage increased in the diazinon group compared to controls. The oxidative properties of diazinon could have given rise to the sperm DNA damage in the present study (26). However, selenium co-administered with diazinon was observed to reduce this disorder compared to the diazinon group. According to the results obtained by Ujang (2008), selenium has antioxidant effects on human health (14). In this respect, the results of the present study indicated that the IVF capacity, hatching, and early in-vitro embryonic development significantly decreased in the diazinon group compared to the control group, while selenium co-administered with diazinon increased the mentioned parameters more significantly compared to the controls.
In the animals receiving diazinon in the current research, various phenomena were observed, including inflammation, testicular damage, DNA destruction, mitochondrial defects, sperm cytoplasmic membrane breakdown, apoptosis, and cell death (26). Similar studies have also reported that sperms with DNA impairment are unable to fertilize an oocyte (27), while selenium could reduce the debilitative effects of diazinon through antioxidant activities, recover the capacity of the sperm to IVF, and continue the other embryonic development stages. Consistent with our findings, Cabaj et al. (2014) reported that selenium co-administrated with diazinon could protect the seminiferous epithelium and interstitial tissue against the toxic effects of diazinon (16).
In conclusion, According to the results, using the sperms collected from the diazinon treatment group could arrest the cleavage stage in the fertilized oocytes, hatching, and blastocysts formation. In addition, the co-administration of diazinon with selenium caused a cell division in the zygotes, prompting the embryonic development to continue.