1. Context
Lower respiratory tract infections (LRTIs) are prevalent diseases and a major cause of referral to primary healthcare centers. Antibiotics are mostly prescribed for the treatment of these disorders as bacterial pathogens are considered to be the most common etiological factors that induce LRTIs (1, 2).
The diagnosis and treatment of LRTIs require a multidisciplinary approach that integrates current clinical, microbiological, and immunological data. Such an approach is essential due to the large number of the etiological factors that cause these infections, the unfavorable symptoms caused by particular pathogens, the increasing number of antibiotic-resistant bacteria, and limited microbiological diagnostic capabilities (3). Physicians may not routinely opt for etiologic diagnoses and prefer a therapeutic approach based on the distribution of pathogens and previous epidemiological findings (4).
The early and accurate identification of etiological agents is the most effective approach to avoiding inappropriate antibiotic use (5). The first-line treatment for LRTIs should be planned in accordance with national recommendations on appropriate antibiotics, as well as the spectrum of the common pathogens and resistance patterns in a specific region (6).
2. Objective
The present study aimed to identify the bacterial etiology of LRTIs, determine the trend of changes within the past three decades, and help ascertain the new empirical LRTI therapy scenarios in Turkey.
3. Methods
This systematic review was conducted in four phases of data sources and keywords, qualitative analysis of studies, determination of eligibility criteria, and data analysis.
3.1. Data Sources and Keywords
A systematic review was performed via searching in databases such as PubMed, Turkish Medline, Scopus, EBSCO, Google Scholar, and other indexed journals using keywords such as lower respiratory tract infections, lower respiratory tract infections in Turkey, respiratory tract infections in Turkey, and alt solunum yolu enfeksiyonları Türkiye.
3.2. Eligibility Criteria
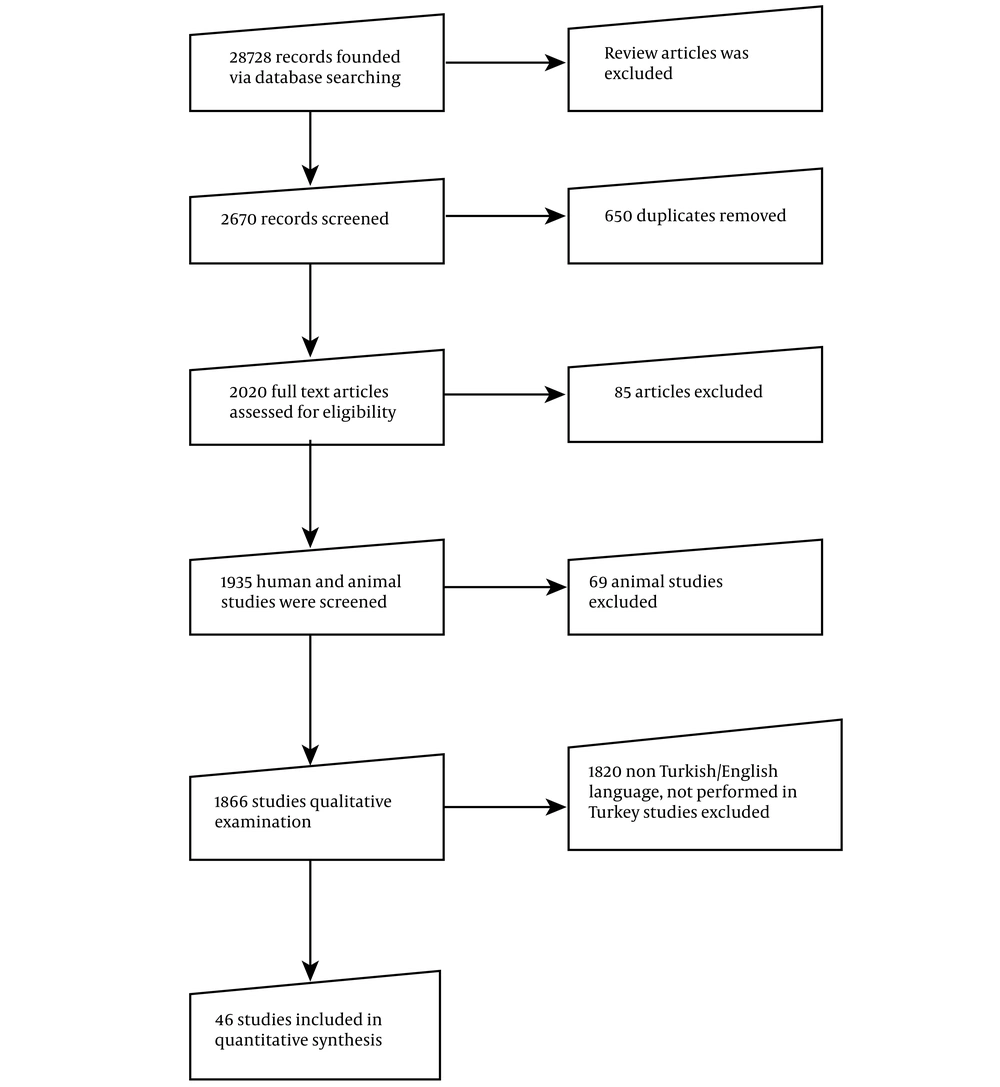
The eligibility criteria of the articles were the scientific studies conducted in Turkish or English language, studies performed in Turkey, and studies presenting consistent data. The exclusion criteria were unavailable full texts, data on lower respiratory tract infections, data on the infections between upper/lower respiratory tracts without distinction, assessment of fewer than 10 specimens, and lack of detailed description. Based on the screening criteria, 46 studies were selected for our systematic review. Figure 1 depicts the flowchart of the systematic review.
3.3. Data Analysis
Data were divided into five groups based on the publication year of the selected studies, including 1990 - 1995, 1996 - 2001, 2002 - 2007, 2008 - 2013, and 2014 - 2020. Two independent researchers analyzed the data, and disagreements were resolved unanimously in line with the eligibility criteria. Data were collected on the name of the first author, study area, total number of isolates, methodologies, characteristics of the patient population, and prevalence of LRTI agents. In addition, tables were drawn to demonstrate the prevalence data in terms of numbers, so that the selected studies could be assessed by a common unit.
4. Results
The literature search based on the eligibility criteria resulted in 2,670 articles, which had been published during 1990 - 2020 and could be potentially used, and 46 scientific studies meeting the eligibility criteria were selected for the final review (Table 1). Frequencies were determined by the analysis of the data on LRTI agents reported in different studies.
| Author | Year | City | N | Bacterial Species | Comorbidities | Clinics |
|---|---|---|---|---|---|---|
| Kurt et al. (7) | 1990 | Ankara | 16 | H. influenzae, S. pneumoniae | 1 LA, 6 pneumonia, 5 bronchiectasis, 4 bronchiectasis + COPD | - |
| Saka et al. (8) | 1994 | Ankara | 85 | S. pneumoniae | 24 COPD, 3 COPD + 13 DM, COPD + CP, 8 bronchiectasis, 2 APT, 2 IPT, 11 pneumonia, 1 pneumonia + PE, 1 LA, 18 LC, 1 LC + DM, 5 BA, 2 DIF, 1 DL | - |
| Berkiten et al. (9) | 1994-1997 | İstanbul | 291 | H. influenzae | - | - |
| Koseoglu et al. (10) | 1996 | Ankara | 24 | S. pneumoniae, H. influenzae, B. catarrhalis | 27 COPD, 13 BA, 7 bronchiectasis, 7 CB, 2 LC, 1 emphysema | |
| Ceylan et al. (11) | 1996 | İzmir | 60 | P. aeruginosa, K. pneumoniae, Enterobacter spp. | 29 COPD, 25 CAP, 12 HAP | |
| Berkiten et al. (12) | 1997 | İstanbul | 34 | S. pyogenes | - | |
| Yurdakul et al. (13) | 1997 | Ankara | 514 | S. pneumoniae, P. aeruginosa, S. aureus, K. pneumoniae, M. catarrhalis | - | ICU |
| Senol et al. (14) | 1999 | İzmir | 365 | S. pneumoniae, H. influenzae, M. catarrhalis | - | ICU, Outpatient |
| Aydın et al. (15) | 1999 | Sivas | 35 | S. pneumoniae | 35 COPD | - |
| Coplu et al. (16) | 1999 | Ankara | 64 | H. influenzae | 33 CF | Outpatient |
| Aktepe et al. (17) | 2000 | Ankara | 288 | H. influenzae, P. aeruginosa, S. aureus | 288 CF | - |
| Ünel et al. (18) | 2000 | İstanbul | 100 | H. influenzae, M. catarrhalis, S. pneumoniae, Pseudomonas spp. | 100 COPD | - |
| Berkiten et al. (19) | 2001 | İstanbul | 102 | S. pyogenes | - | - |
| Erdogan et al. (20) | 2001-2002 | İstanbul | 466 | H. influenzae, H. parainfluenzae, S. pneumoniae, M. catarrhalis, MSSA | - | - |
| Talay et al. (21) | 2002 | İstanbul | 41 | S. pneumoniae, K. pneumoniae, P. aeruginosa, E. coli | 17 COPD, 25 pneumonia | inpatient |
| Ciragil et al. (22) | 2002 | İstanbul | 59 | P. aeruginosa | 59 CF | ICU, inpatient |
| Azap et al. (23) | 2002 | Ankara | 77 | S. pneumoniae | COPD, CAP | ICU, inpatient, Outpatient |
| Gonlugur et al. (24) | 2004 | Sivas | 355 | Klebsiella spp., E. coli, Proteus spp., Enterobacter spp., A. baumannii, Serratia spp. | NRI | ICU, inpatient, Outpatient |
| Gurol et al. (25) | 2004 | İstanbul | 44 | S. pneumoniae | - | - |
| Arikan Akan et al. (26) | 2005-2007 | Ankara | 25 | A. baumannii, K. pneumoniae | - | ICU, inpatient, |
| Atasever et al. (27) | 2006 | İzmir | 837 | S. aureus, M. catarrhalis, H. influenzae, P. aeruginosa, S. pneumoniae, K. pneumoniae, E. coli, Acinetobacter baumannii, Enterobacter spp. | 837 LC, 535 pneumonia, 515 COPD | ICU, inpatient, Outpatient |
| Komus et al. (28) | 2006 | İzmir | 16 | P. aeruginosa | 16 bronchiectasis | - |
| Uncu et al. (29) | 2007 | Ankara | 82 | S. pneumoniae, H. influenzae | - | - |
| Demir et al. (30) | 2007-2010 | Trabzon | 78 | P. aeruginosa | - | inpatient |
| Bayram et al. (31) | 2007-2010 | Van | 191 | A. baumannii | - | ICU, inpatient |
| Gazi et al. (32) | 2008 | Manisa | 835 | Pseudomonas spp., Acinetobacter spp., Klebsiella spp., E. coli, S. maltophilia, Enterobacter spp., S. marcescens | - | ICU, inpatient |
| Eksi et al. (33) | 2008 | Gaziantep | 116 | S. pneumoniae | - | ICU, inpatient, Outpatient |
| Kume et al. (34) | 2008-2010 | İzmir | 175 | Acinetobacter spp., Pseudomonas spp. | 33 pneumonia, 40 COPD, 12 LC | ICU, |
| Akin et al. (35) | 2009 | Konya | 62 | S. pneumoniae, H. influenzae, M. catarrhalis | 62 COPD | - |
| Ince et al. (36) | 2009 | Duzce | 98 | M. catarrhalis, S. pneumoniae, H. influenzae, P. aeruginosa, E. coli | 98 COPD | ICU, inpatient, Outpatient |
| Bacakoglu et al. (37) | 2009 | İzmir | 37 | A. baumannii | 8 COPD, 10 CAP, 1, pyopneumothorax, 1 LC | ICU, |
| Beşli et al. (38) | 2012-2014 | İstanbul | 586 | S. pneumoniae, H. influenzae | - | - |
| Özer et al. (39) | 2012-2014 | Hatay | 1516 | A. baumannii, P. aeruginosa, K. pneumoniae, E. coli, S. aureus, Enterobacter cloacae, Streptococcus pneumoniae, Stenotrophomonas marcescens | - | ICU, inpatient, |
| Akkoyun Bilgi et al. (40) | 2013 | İstanbul | 163 | H. influenzae, S. pneumoniae, P. aeruginosa | - | ICU, inpatient, Outpatient |
| Bayramoglu et al. (41) | 2014 | Trabzon | 67 | S. pneumoniae, M. catarrhalis, H. influenzae | - | - |
| Direkel et al. (42) | 2015 | Mersin | 277 | A. baumanniii | ||
| Savcı et al. (43) | 2015 | Ankara | 67 | A. baumanniii | - | - |
| Alışkan et al. (44) | 2016 | Adana | 184 | S. Pneumoniae,H. İnfluenzae | - | - |
| Eroğlu et al. (45) | 2016 | Samsun | 1183 | A. baumanniii | - | ICU, inpatient, Outpatient |
| Aydemir et al. (46) | 2016 | Sakarya | 130 | A. baumannii, K. pneumoniae, E. coli, S. aureus, E. cloacae, P. aeruginosa | - | ICU |
| Maçin et al. (47) | 2017 | Ankara | 130 | P. aeruginosa | - | - |
| Sagmak-Tartar et al. (48) | 2017 | Elazığ | 535 | A. baumanniii, P. aeruginosa, K. pneumoniae | - | ICU |
| Kahraman et al. (49) | 2017 | Sakarya | 71 | K. pneumoniae | - | - |
| Altun et al. (50) | 2018 | Ankara | 52 | S. pneumoniae | - | - |
| Tanriverdi et al. (51) | 2019 | Samsun | 330 | H. influenzae, M. catarrhalis, M. catarrhalis, S. Pneumoniae | - | ICU, inpatient, Outpatient |
| Altay Koçak et al. (52) | 2019 | Adana | 181 | A. baumannii, Klebsiella spp., P. aeruginosa, E. coli, H. influenzae, S. aureus, S. pneumoniae | - | ICU, inpatient |
Data of Reviewed Studies
According to the findings, the most commonly isolated bacteria were Acinetobacter spp. (31.68%), P. aeruginosa (16.59%), H. influenzae (14.30%), and S. pneumoniae (13.80%). Table 2 shows the frequency of the isolated bacterial species by years. Accordingly, S. pneumoniae was the most common bacterial species isolated during 1990 - 1995, M. catarrhalis was mostly frequently isolated during 1996 - 2001, S. aureus was most commonly isolated during 2002 - 2007 and 2008 - 2013, and A. baumannii was most frequently isolated during 2014 - 2020.
| Interval | |||||
|---|---|---|---|---|---|
| 1990 - 1995 | 1996 - 2001 | 2002 - 2007 | 2008 - 2013 | 2014 - 2020 | |
| Acinetobacter spp. | 0 | 0 | 81 | 872 | 2,024 |
| B. catarrhalis | 0 | 3 | 0 | 0 | 0 |
| E. coli | 0 | 5 | 98 | 142 | 78 |
| Enterobacter spp. | 0 | 4 | 94 | 32 | 12 |
| H. influenzae | 4 | 227 | 298 | 380 | 435 |
| K. pneumoniae | 0 | 25 | 152 | 147 | 318 |
| M. catarrhalis | 0 | 345 | 0 | 56 | 0 |
| P. aeruginosa | 0 | 220 | 270 | 760 | 308 |
| Proteus spp. | 0 | 0 | 10 | 0 | 0 |
| S. aureus | 0 | 70 | 315 | 37 | 55 |
| S. maltophilia | 0 | 0 | 0 | 42 | 0 |
| S. pneumoniae | 112 | 290 | 118 | 288 | 489 |
| Serratia spp. | 0 | 0 | 6 | 21 | 0 |
| Total | 116 | 1,189 | 1,442 | 2,777 | 3,719 |
Total Frequencies of Bacterial Isolates by Year (N)
The findings of the current review indicated significant changes in the reported LTRI data over time, which could be associated with the increased prevalence of A. baumannii infections. Furthermore, a three-fold increase was observed in the data of A. baumannii infections, especially within the past seven years. On the other hand, the reports on P. aeruginosa infections have decreased (Table 2).
Sputum, tracheal aspirate (TA), endotracheal aspiration (ETA), and bronchoalveolar lavage (BAL) have been the most commonly used samples in the diagnosis of LRTI. Table 3 shows the frequency of bacterial isolation and the clinical sample table correlations. In terms of isolation frequency, A. baumannii has been isolated from TA, sputum, BAL, and ETA samples, P. aeruginosa and K. pneumoniae have been isolated from BAL and sputum samples, and S. pneumoniae and H. influenzae have been the most frequently isolated agents from sputum samples.
| Sample Type | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| TA | S | BAL | DTA | ETA | PF | US | BA | TS | PE | |
| A. baumannii | 1,339 | 536 | 330 | 115 | 319 | 206 | 129 | 0 | 3 | 0 |
| B. catarrhalis | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| E. coli | 59 | 96 | 78 | 0 | 36 | 29 | 23 | 0 | 0 | 0 |
| Enterobacter spp. | 11 | 23 | 10 | 0 | 28 | 61 | 9 | 0 | 0 | 0 |
| H. influenzae | 256 | 680 | 186 | 6 | 0 | 182 | 0 | 34 | 0 | 0 |
| H. parainfluenzae | 0 | 17 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| K. pneumoniae | 95 | 218 | 99 | 17 | 0 | 121 | 64 | 0 | 0 | 16 |
| M. catarrhalis | 113 | 112 | 36 | 0 | 0 | 135 | 0 | 3 | 0 | 0 |
| P. aeruginosa | 200 | 787 | 102 | 94 | 87 | 198 | 82 | 0 | 0 | 0 |
| Proteus spp. | 0 | 10 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| S. aureus | 52 | 105 | 133 | 29 | 0 | 158 | 0 | 0 | 0 | 0 |
| S. maltophilia | 0 | 0 | 0 | 0 | 0 | 0 | 42 | 0 | 0 | 0 |
| S. pneumoniae | 173 | 533 | 170 | 61 | 0 | 167 | 0 | 67 | 96 | 0 |
| S. pyogenes | 2 | 34 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Serratia spp. | 0 | 27 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 2,300 | 3,181 | 1,244 | 322 | 470 | 1,257 | 349 | 104 | 99 | 16 |
Total Frequencies of Bacterial Isolates by Sample Type (N)
Table 4 shows the distribution of the isolated infectious agents by geographical regions. Data are scarce on nosocomial LRTI agents in the Black Sea region. Moreover, sufficient data could not be found regarding LRTI agents in the Southeastern Anatolia region. The Aegean region has also been the geographical area with the highest number of notifications on LRTI agents. S. pneumoniae and H. influenzae are community-acquired LRTI agents detected in all regions. Furthermore, nosocomial agents such as A. baumannii, P. aeruginosa, K. pneumoniae, and S. aureus have been frequently reported. The overall analysis of data indicated that LRTI agents were most commonly reported in the Aegean region during 2014 - 2020.
| Marmara | Mediterranean | Central Anatolia | Eastern Anatolia | Southeastern Anatolia | Aegean | Black Sea | |
|---|---|---|---|---|---|---|---|
| A. baumannii | 123 | 717 | 61 | 1,764 | 0 | 312 | 0 |
| B. catarrhalis | 0 | 0 | 3 | 0 | 0 | 0 | 0 |
| E. coli | 21 | 141 | 18 | 0 | 0 | 140 | 3 |
| Enterobacter spp. | 18 | 16 | 10 | 0 | 0 | 98 | 0 |
| H. influenzae | 457 | 161 | 151 | 30 | 0 | 265 | 280 |
| K. pneumoniae | 130 | 172 | 49 | 101 | 0 | 190 | 0 |
| M. catarrhalis | 41 | 0 | 24 | 0 | 0 | 281 | 55 |
| P. aeruginosa | 151 | 296 | 286 | 127 | 0 | 597 | 101 |
| Proteus spp. | 0 | 0 | 10 | 0 | 0 | 0 | 0 |
| S. aureus | 38 | 72 | 52 | 0 | 0 | 315 | 0 |
| S. maltophilia | 0 | 0 | 0 | 0 | 0 | 42 | 0 |
| S. pneumoniae | 274 | 192 | 506 | 55 | 21 | 137 | 130 |
| Serratia spp. | 0 | 9 | 6 | 0 | 0 | 12 | 0 |
| Total | 1,253 | 1,776 | 1,176 | 2,077 | 21 | 2,389 | 569 |
Total Frequencies of Bacterial Isolates by Geographical Region in Turkey (N)
5. Discussion
The etiological diagnosis of LRTIs, which is a major cause of mortality and morbidity, is essential to the treatment of the disease. The distribution of LRTI etiological agents may vary depending on the geographical region, season, age, ethnicity, and underlying diseases (53). LRTIs have changed during the COVID-19 outbreak, which is currently the most pressing public health concern. Consequently, community-acquired and nosocomial LRTI bacterial agents have been extensively investigated due to the risk of developing secondary infections and the essential differences between the treatment of the viral and bacterial agents that cause LRTIs. Therefore, determining possible LRTI pathogens through large-scale studies is of utmost importance in the planning of empirical treatments (54).
According to the studies evaluated in our systematic review, the most common community-acquired LRTI agents were S. pneumoniae and H. influenzae, while the most common nosocomial LRTI agents were A. baumannii, P. aeruginosa, K. pneumoniae, and S. aureus. Changes in the frequency of LRTI agents may be affected by factors such as the identification method, type of isolation samples, waiting times/conditions of sampling, healthcare professionals collecting the samples from the patients, interpretation of the results, and the principles of the automated devices.
Our observations indicated that researchers' trends are one of the critical factors affecting the distribution and frequency of LRTI pathogens (Table 4). The regional distribution of the studies showed that they were mostly performed in the Aegean region, and limited data were provided on the Southeastern Anatolia, as well as on the nosocomial LRTI pathogens in the Black Sea region. On the other hand, the distribution of these factors may be attributed to the used antibiotics, pathogen resistance, accompanying diseases, diagnostic techniques used in hospitals, and hospitals' development, climate, and sanitation state.
Once the COVID-19 pandemic is over, new discussions may arise regarding LRTI pathogens. It is assumed that the samples used for the detection of pathogens will change drastically. In the reviewed studies, most of the samples to isolate LRTI agents were collected from sputum, TA, pleural effusion, and BAL. However, the lack of rapid, inexpensive, and easily applicable diagnostic methods and/or tools for these samples remains a critical challenge in this regard. Currently, gram-staining is considered to be the only effective option although its value remains controversial (55). Unfortunately, suggestions on rapid conventional and/or automated diagnostic tools were not proposed in the reviewed studies.
A notable finding on this systematic review was that studies regarding LRTI were more abundant during 2010-2020 within the past three decades, which could be attributed to several factors, such as the progress achieved in identification and antimicrobial susceptibility technologies of pathogens.
The main conclusions of the reviewed studies have been summarized below:
- LRTIs are two major causes of global mortality. In addition, hospital-acquired infections are the leading cause of mortality and morbidity in intensive care units (ICUs). The use of broad-spectrum antibiotics leads to the development of more resistant strains. With increased bacterial resistance, appropriate and effective antibiotic treatment may be delayed, which in turn leads to higher sepsis frequency and mortality rates (52).
- Evidently, the resistance rate of gram-negative microorganisms to various antibiotics has increased over the years due to the continuous or long-term use of antibiotics in hospitals. P. aeruginosa is a typical example in this regard, which remains an important pathogen in terms of nosocomial infections, especially in ICUs (52).
- No significant change has been reported in the short-term antimicrobial susceptibility of LRTI agents, and the antibiotics to be used in empirical treatments should be selected meticulously. Additionally, it is essential to evaluate the long-term effectiveness of empirical treatment approaches to plan for the necessary changes (38).
- It is essential to periodically determine the pathogens that cause LRTIs, as well as sensitivity to the antibiotics used in empirical treatment protocols (44).
5.1. Strengths and Limitations
To the best of our knowledge, this is the first study to compile the reports on bacterial LRTI agents. One of the limitations of this systematic review was that we could not review the studies providing data on fewer than 10 patients and did not discriminate between upper respiratory tract infections and LRTIs. In addition, errors are possible due to the differences in the identification methods and the experts who evaluate culture results. Therefore, the generalization of the results should be further discussed.
5.2. Conclusions
Since the disease course of bacterial LRTIs is generally mild, uncomplicated, and similar to non-bacterial LRTIs, physicians should not prescribe antibiotics to the patients without the detection of LRTI agents even if they are bacterial. This systematic review was focused on the reported data on the 30-year frequency of LRTI agents. As the frequency of LRTI bacterial factors is specific to each country, healthcare professionals may be able to report cultures and prescribe/select antibiotics. These findings could contribute to the prevention and reduction of antibiotic resistance, which is a major health issue today. It is recommended that further investigations be conducted on homogeneous samples to discuss the frequency of LRTI agents more generally. Furthermore, periodic evaluations could largely contribute to the current literature and treatment guidelines in this regard.