1. Background
Phenolic compounds are priority pollutants due to their adverse effects on human health (1). Bisphenol A (BPA) is an example of these compounds, which is used in large quantities to produce disposable containers, fluid containers, and baby feeding bottles (2). Furthermore, BPA is used to manufacture polycarbonate plastics and epoxy resins of which food and beverage containers and dental materials are composed (3). This compound has a low estrogenic activity, thereby posing the risk of testicular and breast cancer, infertility, developmental abnormalities, endocrine disorders, immune system attenuation, and adverse fetal outcomes (2, 4).
Several studies have been focused on the effects of phenol and its derivatives, such as BPA (5), octylphenol (6), and pure phenol (7), on the male reproductive system in the prenatal and postnatal periods. Previous animal studies in this regard have indicated that the intraperitoneal administration of nonylphenol (42.5 mg/kg) for 35 days could reduce testicular weight (8). In addition, BPA has been reported to increase oxidative stress and cause testicular damage by decreasing the activity of antioxidant enzymes (9). BPA could also impair sexual hormones (especially androgens), thereby affecting sperm production (10).
Bromelain (BROM) is a natural amalgamation of the enzymes that are derived from pineapple, containing various thiol endopeptidases and proteases inhibitors (11-13). The significant pharmacological properties of BROM are associated with proteolytic effects (14). The Food and Drug Administration (FDA) has classified BROM as a permitted dietary supplement and approved its therapeutic effects (12). The injectable lethal dose of BROM is 37 mg/kg in mice and 85 mg/kg in rats, while the oral administration of up to 10 g/kg is associated with no mortality in mice and rabbits (15). BROM could prominently increase antioxidant enzyme activity and inhibit oxidative stress (16). Moreover, the protective effects of pineapple juice on the survival of cryopreserved sperm have been demonstrated (17).
2. Objectives
Phenolic compounds (especially BPA) are found abundantly in the environment, and the debilitative effects of these compounds on the male reproductive system have been previously reported. On the other hand, BROM is a dietary supplement of plant origin with beneficial biological effects (eg, antioxidant and anti-inflammatory properties). To date, no studies have investigated the effects of phenolic compounds (especially BPA) combined with BROM on the male reproductive system of mice. The present study aimed to evaluate the possible therapeutic effects of BROM on the testicular damage caused by BPA toxicity in adult male mice.
3. Methods
3.1. Laboratory Animals
This study was conducted on 40 adult male mice (weight: 25 - 30 g) aged eight weeks, which were obtained from the Center of Research and Reproduction of Laboratory Animals at Ahvaz Jundishapur University of Medical Sciences (AJUMS), Iran. The animals were randomly divided into four groups of 10, including control (daily treatment with olive oil as a BPA solvent with an equal volume to the treatment groups), BPA (Merck, Germany; 600 mg/kg daily) (18), BROM (Acros Organics, Thailand; 70 mg/kg daily) (16), and BPA + BROM (600 mg/kg BPA followed by 70 mg/kg BROM ).
The study was performed in compliance with the ethics of working with laboratory animals (ethics code: EE/97.24.3.49907/scu.ac.ir). The animals were kept at the temperature of 23 ± 2°C within a 12-hour light/dark photocycle and had access to adequate food and water during the study. All the compounds were administered orally for 35 consecutive days at the same conditions and time.
3.2. Testicular Morphometry
Testicular morphometric characteristics were measured, including length (great diameter), width (small diameter), thickness, and weight. Moreover, the gonadosomatic index (GSI) was calculated using the following formula: [Testes weight/body weight] × 100 (19)
3.3. Sperm Collection and Parameter Assessments
Both cauda of epididymis from each animal were crushed and conserved in a warmed (37°C) petri dish containing 10 milliliters of Hank’s balanced salt solution, and the spermatozoa were allowed to disperse into the buffer. After 15 minutes, the suspension was slightly shaken to be normalized and observed using a light microscope. Two indices of sperm parameters were also assessed (sperm motility and sperm count). Three levels of sperm motility were calculated based on the World Health Organization (WHO) guidelines, including progressive (long-distance motility), non-progressive (curved motility with no distance), and immobility (stable sperms). To this end, 50 microliters of seminal fluid and culture medium were aspirated and placed on a culture slide, which was previously cleaned and dried with alcohol, for examination by a microscope. In addition, sperm cell counting was performed using a cell count device, and about 100 sperm cells were counted in each sample (20).
3.4. Histological Assessments
The testes were fixed in 10% formalin buffer solution for one month. Following that, a routine tissue processor was applied, and 5-micrometer cross-sections were prepared and stained by Hematoxylin and Eosin. In total, 30 slides were prepared from each sample for quantitative analysis. The measured histological parameters include the number, diameter, and wall thickness of the seminiferous tubules. Moreover, the counts of spermatogenic, Sertoli, and Leydig cells were determined using a digital lens (Dino-Lite, China) and the DinoCapture II software (21).
3.5. Statistical Analysis
Data analysis was performed in SPSS version 19 (Chicago, USA). After assessing the normal distribution of data and normalization, the data were analyzed using one-way ANOVA, followed by Tukey's post-hoc test. In all the statistical analyses, the P-value of less than 0.05 was considered significant.
4. Results
4.1. Testicular and Total Body Weight of Animals and GSI
A significant decrease was observed in the total body weight of the animals in the BPA group compared to the controls (P < 0.05). In addition, the weight of the animals significantly increased in the BPA + BROM group compared to the BPA group (P < 0.05). The weight of the testes significantly decreased in the BPA group compared to the control group (P < 0.05), indicating a significant incremental trend in the BPA + BROM group compared to the BPA group (P < 0.05). Furthermore, the GSI significantly increased in the BROM and BPA + BROM groups compared to the control and BPA groups, respectively (P < 0.05) (Table 1).
Abbreviations: BPA, bisphenol-A; BROM, bromelain; GSI, gonadosomatic index.
a Significant difference compared to control.
b Significant changes compared to BPA group.
4.2. Morphometric Parameters of Testes
The parameters of length, width, and thickness of the testes decreased significantly in the BPA group compared to the controls (P < 0.05). These values also increased significantly in the BPA + BROM group compared to the BPA group (P < 0.05) (Table 2).
Abbreviations: BPA, bisphenol-A; BROM, bromelain.
a Significant difference compared to control.
b Significant changes compared to BPA group.
4.3. Sperm Parameter Evaluation
The value of progressive motility showed significantly decremental levels in the BPA group compared to the control group (P < 0.05). In this group, the non-progressive and immotile factors also increased significantly compared to the control subjects (P < 0.05). After the administration of BROM, the levels of progressive motility were observed to increase, while the levels of non-progressive and non-motility factors decreased significantly in the BPA + BROM group compared to the BPA group (P < 0.05). Moreover, the value of sperm count decreased significantly in the BPA group compared to the controls (P < 0.05). This factor also showed significantly increased levels in the BPA + BROM group compared to the BPA group (P < 0.05) (Table 3).
| Group | Progressive Motility | Non-progressive Motility | Non-motility | Sperm Count (×106) |
|---|---|---|---|---|
| Control | 38.98 ± 5.12 | 1.22 ± 0.36 | 0.80 ± 0.02 | 13.12 ± 3.1 |
| BPA | 12.10 ± 2.51 a | 18.23 ± 2.41 a | 58.6 ± 6.12 a | 4.98 ± 0.17 a |
| BROM | 37.56 ± 7.15 | 2.65 ± 0.82 | 0.38 ± 0.03 | 14.76 ± 2.29 |
| BPA + BROM | 37.02 ± 6.15 b | 2.23 ± 0.91 b | 3.12 ± 1.09 b | 12.71 ± 2.37 b |
Abbreviations: BPA, bisphenol-A; BROM, bromelain.
a Significant difference compared to control.
b Significant changes compared to BPA group.
4.4. Histopathological Assessments
According to the results of the normal histology of the testes, the seminiferous tubules showed compact sections with a high rate of sperm in the lumen. In addition, high levels of interstitial tissue containing Leydig cells were observed in the control and BROM groups. Various ranges of seminiferous tubule degradation were also detected in the BPA group with less sperm production in the lumen. Different gaps with decreased levels of interstitial tissue were also observed in the loose connective tissue surrounding the seminiferous tubules in the BPA groups.
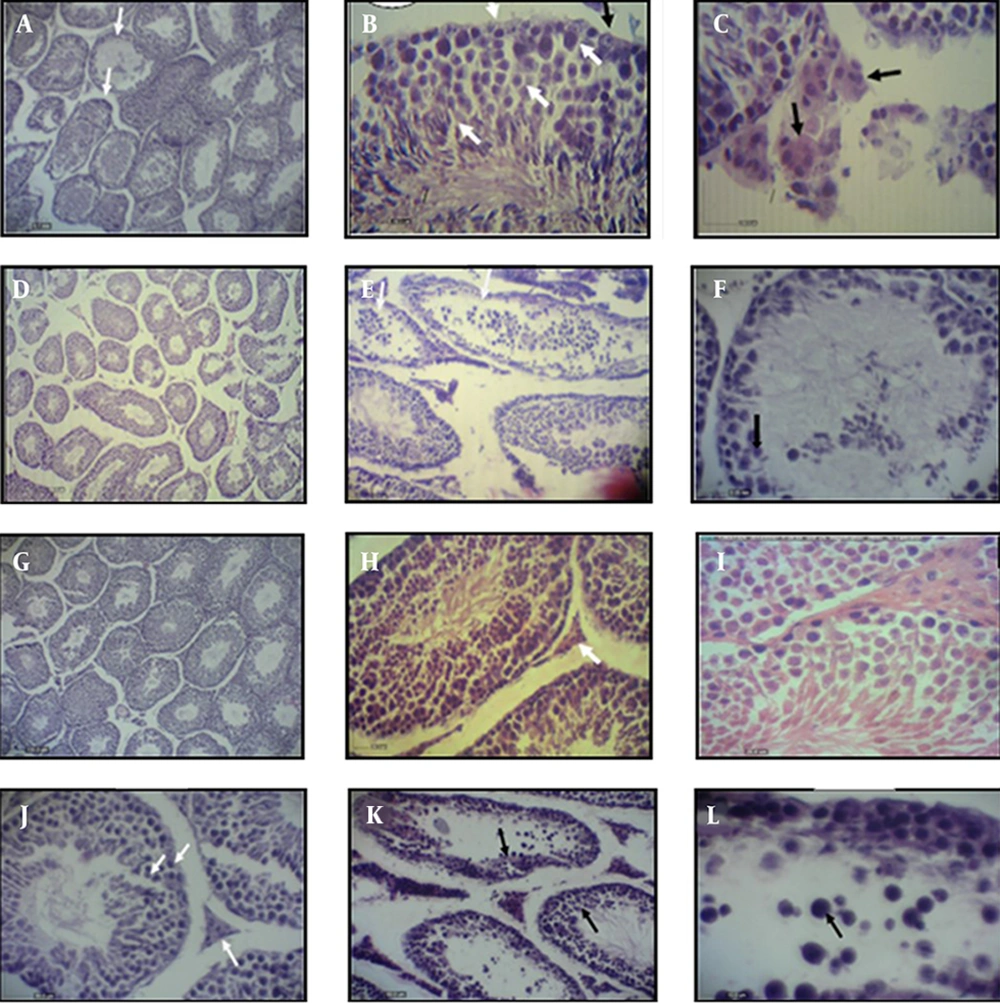
The spermatogenic cell lines had a non-regular arrangement with the disperse distribution of the nuclei. Following the administration of BROM in the BPA + BROM group, the normal structure of the interstitial tissue was observed with a high compact of Leydig cells. In addition, the seminiferous tubules of the animals in this group had a regular arrangement of the spermatogenic lineage in the presence of sperms in the lumen (Figure 1).
Histological images of testis in control (A - C), pathologic BPA group (D - F), BROM group (G - I) and BPA + BROM group (J - L). A, Normal seminiferous tubules; B, Sertoli cells in seminiferous tubules (white arrows); C, Pale Leydig cells in interstitial tissue (black arrows); D, seminiferous tubules in BPA group; E, Sertoli cells (white arrows); F, Leydig cells; G, Seminiferous tubules in BROM group; H, Leydig cells (white arrows); I, Sertoli cells; J, Seminiferous tubules and Leydig cells (white arrows); K, Sertoli cells (black arrows) and L, Leydig cells. BPA: bisphenol-A, BROM: Bromelain. H&E staining, × 100.
4.5. Histometry of Cells in the Testicular Structure
The count, wall thickness, and epithelial thickness of the seminiferous tubules significantly decreased in the BPA group compared to the controls (P < 0.05). These values also indicated a significant incremental trend in the BPA + BROM group compared to the BPA group (P < 0.05) (Table 4). After counting various cell types in the seminiferous tubules, whole-cell types of spermatogonia, primary spermatocytes, spermatids, Sertoli, and Leydig cells were observed to decrease significantly in the BPA group compared to the controls (P < 0.05). In addition, these values increased significantly following BROM administration in the BPA + BROM group compared to the BPA group (P < 0.05) (Table 5).
| Group | Count of Seminiferous Tubules | Wall Thickness of Seminiferous Tubules (µm) | Epithelium Thickness of Seminiferous Tubules (µm) |
|---|---|---|---|
| Control | 27.36 ± 3.19 | 153.23 ± 10.2 | 13.15 ± 2.32 |
| BPA | 6.12 ± 0.4 a | 34.12 ± 6.18 a | 1.45 ± 0.05 a |
| BROM | 25.20 ± 4.9 | 174.67 ± 6.95 | 14.45 ± 3.67 |
| BPA + BROM | 25.15 ± 2.98 b | 165.22 ± 8.14 b | 12.40 ± 3.18 b |
Abbreviations: BPA, bisphenol-A; BROM, bromelain.
a Significant difference compared to control.
b Significant changes compared to BPA group.
| Group | Spermatogonia | Primary Spermatocytes | Early Spermatids | Late Spermatids | Sertoli Cells | Leydig Cells |
|---|---|---|---|---|---|---|
| Control | 12.71 ± 1.67 | 17.65 ± 1.00 | 36.22 ± 2.89 | 19.81 ± 1.14 | 5.88 ± 0.54 | 12.00 ± 2.79 |
| BPA | 9.75 ± 1.10 a | 5.75 ± 1.33 a | 13.25 ± 1.88 a | 4.00 ± 1.06 a | 0.77 ± 0.03 a | 4.12 ± 1.00 a |
| BROM | 13.02 ± 0.44 | 15.90 ± 1.59 | 36.47 ± 3.41 | 21.38 ± 0.93 | 6.55 ± 0.50 | 14.34 ± 0.19 |
| BPA+BROM | 19.75 ± 1.76 b | 17.25 ± 0.62 b | 34.75 ± 2.46 b | 18.12 ± 1.74 b | 5.20 ± 0.31 b | 11.25 ± 0.75 b |
Abbreviations: BPA, bisphenol-A; BROM, bromelain.
a Significant difference compared to control.
b Significant changes compared to BPA group.
5. Discussion
There are several classes of toxins in nature that are harmful to human health. Furthermore, synthetic materials may exert similar toxic effects on the human body. BPA is a toxic substance used in various industries to produce polycarbonate plastics. BPA is the commonly used name for 4’4-isopropylidenediphenol (CAS NO.80-05-7) with the molecular weight of 228.29 g/mol and chemical structure of C15H16O2. BPA could enter the body via digestive, respiratory, and dermal tracts (23), mimicking the effects of natural estrogen derived from the similarity of phenol groups on BPA and estradiol, which enable this synthetic molecule to trigger estrogenic pathways in the human body.
Typically, the phenol-containing molecules that are similar to BPA exert weak estrogenic effects and act as endocrine disruptors, exhibiting estrogen-like and anti-androgen properties and causing damage to various tissues and organs, including the reproductive system (23). Epidemiological evidence confirms that the reproductive system is a vital target organ of BPA, the primary effects of which are interrupting the exertion and activity of sex hormones and influencing the development and function of the reproductive system. According to the literature, BPA could increase estradiol, progesterone, serum luteinizing hormone, and testosterone levels. BPA has also been shown to reduce sperm quality, sexual function, and even the fertility rate (24).
Today, human societies tend to plant elements and extracts for disease treatment and protection against the debilitative effects of chemical drugs. On the other hand, pharmaceutical companies have defined numerous drugs based on herbal elements. Herbal medicines have no similar side-effects to chemical medicines and also contain a high percentage of natural antioxidants (25). BROM is an enzymatic extract derived from the stems of pineapples and has a history of folk medicine use. As an ingredient, it is used in cosmetics, medication, and meat tenderizers. BROM has numerous therapeutic benefits for conditions such as angina pectoris, bronchitis, sinusitis, surgical trauma, thrombophlebitis, wound debridement, and improving the absorption of drugs, particularly antibiotics. It also relieves osteoarthritis, diarrhea, and various cardiovascular disorders. BROM also possesses anti-cancer activities and promotes apoptotic cell death (26). BROM contains various thiol endopeptidases and other components such as phosphatases, glucosidase, peroxidases, cellulases, glycoproteins, carbohydrates, as well as several protease inhibitors.
The medicinal properties of BROM are attributed to the combination of sulfur-containing protein-digesting enzymes known as proteolytic enzymes or proteases and several other substances in smaller quantities. Other substances typically include peroxidase, acid phosphatase, protease inhibitors, and calcium. The potency of BROM is rooted in its enzymes and proteins, which stimulate chemical activities in the body (27). In the present study, the therapeutic effects of BROM were observed on the testicular damages induced by BPA. Furthermore, the antioxidant properties of BROM could ameliorate the debilitative damages to the male reproductive system.
Numerous articles have cited the three main biological effects of BROM, which are anti-inflammatory, proliferative (28), and antioxidant effects (22). In addition, some studies have demonstrated the anti-proliferative effects of this extract on cancer cells (29). Substances with antioxidant properties cause the generation of free radicals. Following contact with toxins and pathological substances such as BPA, the normal and physiological activities of the entire cell are disrupted, and lipid peroxidation occurs. In these cases, cell membranes such as plasma membranes, nuclear membranes, and mitochondria are prone to damage (30). With plasma membrane disintegration, cell cohesion is completely lost, thereby leading to premature cell death. If there is an abnormality in the membrane of organs such as mitochondria, the leakage of enzymes into the matrix of this organ occurs, thereby leading to the onset of apoptosis. These pathological processes could eventually cause cell death and decrease the number of tissue-active cells, thereby reducing the size and weight of the tissue over time (31). In the current research, the density of Leydig cells and the spermatogenic lineage and the production of sperm cells in the lumen of the seminiferous tubules showed a descending trend.
Plant-based medicinal extracts contain various effective antioxidants. These enzymatic substances reduce the antioxidant level of cells via two main pathways; antioxidants either act inherently by removing the active oxygen species and oxidative mediators or activate the intracellular antioxidant system to eliminate harmful agents (32). With its inherent antioxidant properties, BROM increases cell viability. This plant substance also helps eliminate antioxidants by activating intracellular enzymes through antioxidant properties. By reducing the oxidative level of cellular ester, the rate of apoptosis reduces, and the necessary opportunity for cell repair is provided. BROM also stimulates tissue repair by providing stable conditions and cell life (22).
Inflammation is a physiological process that increases blood flow to the tissues and lymphocyte infiltration for organ repair. Although inflammation is a physiological process, it uses mechanisms causing tissue damage in some cases (33). During the inflammatory process, the amount of the cytokines secreted by macrophages increases, thereby inducing apoptosis in parenchymal tissue cells. Extensive apoptosis following tissue inflammation could also increase the rate of cell death and decrease the weight and size of the organs. Using BPA with the onset of inflammation in the tissue leads to the mentioned pathological processes.
According to the current research, the exposure of healthy cells in the body to BPA reduced the testicular size and sperm production of the animals. The anti-inflammatory properties of BROM have been confirmed in several studies (28). BROM inhibits inflammation by inhibiting lymphocyte infiltration and inhibiting the presence of macrophages and their cytokine secretions, in which case the rate of cell apoptosis reduces, and the spermatogenic lineage is maintained to produce healthy and abundant sperm cells.
After the tissue damage caused by exposure to toxic substances such as BPA, cell density in tissue decreases. Therefore, the optimal alternative to compensating for tissue cell deficiency is the use of natural materials with proliferative properties. Several studies have highlighted the proliferative properties of BROM (34). BROM increases cell division by employing intracellular mechanisms, thereby increasing the density of the loose connective tissue cells containing Leydig cells, as well as the density of the spermatogenic lineage cells. With the proliferation of Leydig cells after using BROM, blood testosterone levels rise to normal levels. As a result, Sertoli cells are affected, and the spermatogenic lineage begins to proliferate. Following the consumption of BROM in the present study, the rate of cell density in the interstitial tissue and Leydig and Sertoli cells of the animals increased. Moreover, the spermatogenic lineage proliferated and increased sperm production. The high rate of cell proliferation could be attributed to the mitogenic properties of the administered BROM extract.
5.1. Conclusions
According to the results, BPA had detrimental effects on the spermatic cell lineage, which decreased sperm production and subfertility levels. On the other hand, the anti-oxidative properties of BROM resulted in the repair of the induced reproductive damage in the animals. The therapeutic properties of this herbal agent also induced the proliferation of various cell types, including Leydig cells, the spermatogenic lineage, Sertoli cells, and sperms. Therefore, BROM could be used as a natural antioxidant to ameliorate testicular damage and enhance fertility in BPA-induced male toxicity.