1. Background
Cardiovascular diseases are among the leading causes of death worldwide (1). One of the most critical cardiovascular diseases is coronary artery disease (CAD), characterized by stenosis and blockage of the coronary artery due to atherosclerosis (2). Common treatments include nonpharmaceutical and pharmaceutical treatments, coronary artery bypass graft (CABG) surgery, and angioplasty (3); meanwhile, the most effective treatment is CABG (4). In Iran, 16,000 to 18,000 people undergo this surgery annually (5). Respiratory complications after CABG are expected, with an incidence rate of 90%. These complications prolong postoperative hospitalization and impose increased financial and psychological burdens on patients and society (6). Breathing disorders can result in irreversible cardiac complications and increase postoperative mortality (7). Postoperative respiratory complications include atelectasis, arterial hypoxemia (8), pneumonia, respiratory failure, prolonged use of a ventilator, pneumothorax (9), pleural effusion, and pulmonary edema (10).
Breathing exercises are cost-effective techniques with minor complications, aiming to improve the respiratory condition (11). These exercises enhance the respiratory tract function, strengthen respiratory muscles, improve thoracoabdominal movements, increase lung volume, clear the lungs from secretions, and manipulate breathing patterns, e.g., by reducing the breathing rate and abdominal breathing and breathing through the nose instead of the mouth (12, 13). The most common breathing exercises are deep breathing, effective coughing, pursed-lip breathing (PLB), diaphragmatic breathing, respiratory muscle training (RMT), and incentive spirometry (14).
The use of breathing exercises to enhance respiratory function in asthma and chronic obstructive pulmonary disease (COPD) has a long history (15-17). Many studies have investigated the positive effect of breathing exercises on postoperative atelectasis, arterial oxygen saturation (SpO2), and pulmonary function, yet there are controversies regarding the results (11, 18-20). A study showed that five deep breathing exercises and effective coughing four times a day have no significant effect on SpO2, lung collapse, or atelectasis postoperatively (18). Similarly, another study reported that breathing exercises, such as deep breathing, effective coughing, and incentive spirometry, are not significantly associated with the incidence of atelectasis and SpO2 after CABG (20). On the other hand, a study revealed that deep breathing exercises and effective coughing reduce atelectasis incidence (21). Similarly, a study conducted in Iran demonstrated a decline in the incidence of atelectasis and an increase in the respiratory function of patients who performed breathing exercises compared to the control group (11).
Most studies have examined the effect of breathing exercises on the respiratory condition after CABG separately (11, 19, 22, 23). Meanwhile, no study has explored the simultaneous use of breathing exercises, including PLB, diaphragmatic breathing, incentive spirometry, and effective coughing on pulmonary and respiratory condition, i.e., SpO2 level, the incidence of atelectasis, respiratory auscultation, and breathing rate postoperatively. Simultaneously performing two complementary treatment methods, such as breathing exercises, may help prevent pulmonary complications in patients undergoing CABG.
2. Objectives
This study aimed to determine the effect of breathing exercises on the respiratory condition after CABG.
3. Methods
This was a randomized controlled clinical trial with the IRCT code of IR.MAZUMS.REC.1400.11515. The researcher obtained permission from the Research Ethics Committee of Mazandaran University of Medical Sciences and the National Ethics Committee (Iran).
3.1. Statistical Population
The statistical population comprised patients who were CABG candidates and visited the Heart Center of Mazandaran University of Medical Sciences. The participants were recruited based on the inclusion and exclusion criteria.
3.2. Inclusion and Exclusion Criteria
The inclusion criteria consisted of age over 18 years, provision of informed consent, and lack of an ejection fraction (EF) of less than 30% based on a preoperative echocardiogram (24). In addition, the eligible participants did not have chronic respiratory diseases such as COPD (25-27), heart and lung surgery (27), lung injury or damage, broken ribs, chest tube placement (27), cognitive and neurological disorders (25), trauma to the head or nose, frequent sinus infections (25), or chemotherapy. They had not taken immunosuppressive drugs in the last three months (28) and had a body mass index (BMI) of more than 40 (28).
The exclusion criteria were severe postoperative hemodynamic instability (systolic blood pressure < 80 mmHg) (25), arterial blood pH < 7.30 (28), PCO2 > 50 mmHg (28), SpO2 < 80% despite receiving supplemental oxygen (28), serum creatinine > 3.5 mg/dL (28), aortic collapse time > 150 minutes (29), cardiopulmonary bypass time > 240 minutes, using an intra-aortic pump balloon during and after the surgery (30), intubation > 24 hours (25-27), and requiring re-operation or re-intubation not included in the research process (25). Considering the atelectasis incidence (the principal respiratory variable), the significance level of α = 0.05, the test power of 90%, and a previous study (31), the researchers determined that 21 patients in each study group would be adequate. However, they recruited 52 patients (26 in the intervention and 26 in the control group) due to a 20% risk of attrition. They used nonrandomized convenience sampling. To this end, they visited the Mazandaran Heart Center the day before the surgery, checked the waiting list of patients undergoing CABG, interviewed them, and explained the research objectives.
3.3. Sampling
They included the patients if they gave informed consent and were eligible based on the inclusion criteria. Via the block randomization method, the participants were randomly divided into a breathing exercises group and a control group. The single-blind technique was utilized to reduce the biases related to the intervention and outcome evaluation, and the participants did not know whether they belonged to the control or the intervention group.
After assigning the patients to their groups, the researchers gave them the necessary instructions and training in a session before the surgery. They taught the exercises to the intervention group face-to-face, used pictures, and provided practice so that the patients could correctly perform the exercises. Before the intervention, the researchers measured variables such as blood oxygen saturation, respiratory rate, lung auscultation, and atelectasis incidence through an examination performed by three radiologists, provided that at least two confirmed the presence of atelectasis. They also evaluated patients' satisfaction with their pulmonary function in both groups. All the patients received the hospital's routine breathing exercises after the surgery. The exercises included incentive spirometry (once per shift) and vibration (once daily). Besides these breathing exercises, under the researcher's supervision, the intervention group performed breathing exercises, including diaphragmatic breathing, PLB, incentive spirometry, and effective coughing after extubation.
According to diaphragmatic breathing instructions, the patients had to lie down and put one hand on the stomach and the other on the chest. When inhaling, the air entered through the nose, and the abdomen moved upwards while the chest was kept still. Then, by exhaling through the mouth, the stomach moved down. Diaphragmatic breathing was performed three times during waking hours (at 12, 16, and 20 o'clock), and each consisted of 10 breaths (27). For PLB completion, the patients were asked to inhale through the nose by relaxing the neck and shoulder muscles in the tidal volume range, count to two, and then close the mouth. When exhaling, they had to partly press their lips and contract the abdominal muscles, count to five, and exhale slowly through the mouth, counting from one to five. The intervention group performed this exercise three times during waking hours (at 12, 16, and 20 o'clock after the diaphragmatic breathing exercise) for 20 minutes (32, 33).
To reduce the pressure of the exercise on the patients and based on a previous study (11), the researchers administered a combination of incentive spirometry and effective coughing. Incentive spirometry was performed using the Voldyne 500 incentive spirometer in a sitting position with 10 deep breaths at 8, 10, 12, 14, 16, 18, and 20 o'clock. After taking a deep breath, the patients held their breath for 2 - 3 seconds, and when exhaling, they coughed effectively in five breaths and exhaled slowly (without effective coughing) in the rest of the breaths. They coughed effectively by placing their hands on their stomach with 2-3 short and quick coughs (11, 34). The mentioned breathing exercises started one hour after extubation (34) under the researcher's or research assistant's supervision and continued until the fourth day after the surgery (26).
3.4. Intervention
Using a questionnaire, the researchers collected and recorded demographic and medical information, including age, sex, marital status, level of education, smoking history, height, weight, BMI, and underlying diseases (diabetes, hypertension, cardiomyopathy, etc.). They used the content validity method to determine the validity of the medical information questionnaire. The researchers also used a checklist to record the information about the surgical condition (the location and number of bypassed arteries, length of the surgery, time of using the heart-lung pump, time of using the ventilator in the intensive care unit (ICU), the duration of intubation during the patient care process) and respiratory condition (atelectasis incidence, SpO2, and pulmonary auscultation). They used the visual analog scale (VAS) to check the patients' satisfaction with respiratory function. Previous studies have confirmed the validity and reliability of this scale (35, 36). In order not to impose an additional burden on the patients in both groups, the researchers checked the incidence of atelectasis using a chest X-ray (CXR) on the day before the surgery and at the end of the fourth day after surgery at 21:00. To diagnose the incidence of atelectasis, the opinion of three radiologists and the confirmation of at least two of them regarding the presence of atelectasis were necessary considering the changes, including the displacement of the interlobar fissure, increase in density, congestion of the bronchovascular view, ipsilateral elevation of the diaphragm, tracheal displacement, umbilical cord elevation in lower lobe atelectasis, compensatory hyperventilation in other lobes, ipsilateral reduction of the hemithorax volume, and decrease of intercostals space (29).
The researchers measured SpO2 and the number of breaths per minute before inducing anesthesia and after extubation at 12:00 p.m. On the 2nd, third, and fourth days after surgery using, pulse oximetry monitoring (Saadat Co. ALBORZ b9) (25, 27). An anesthesiologist performed the lung auscultation with a stethoscope (3M™ Littmann Classic III) one day before the surgery, every two hours for six hours after the surgery, every six hours for 24 hours after the surgery, every 12 hours on the second day, and once at 18:00 on the third and fourth days. Then, a physician diagnosed the abnormal sounds, i.e., crackles, wheezing, extra voice, reduction of normal voice, and harshness (11, 37). To evaluate the patients' satisfaction with the respiratory function before the surgery and at the end of the study (the fourth day after the surgery), the researchers showed the patients the 10-point VAS and asked them to rate their respiratory function from 0 to 10.
Finally, the data were imported to SPSS v. 18 (IBM, Chicago, IL, USA). For descriptive statistics, frequency, percentage, mean, and standard deviation (SD) were reported. The qualitative data were analyzed using the chi-square test. Mann-Whitney U and Wilcoxon tests were used to analyze the data that did not have a normal distribution (VAS for satisfaction with breathing performance). Repeated measures analysis of variance (ANOVA) and t-test were used to study the variables with a normal distribution (other quantitative variables). The significance level for all the tests was P ≤ 0.05.
4. Results
Most participants were female (72%) in the control group and male (73.1%) in the intervention group. Overall, men constituted 51% of the participants. The chi-square test showed a significant difference between the two groups regarding sex (P = 0.001).
None of the participants had a history of smoking, cardiomyopathy, respiratory disease, heart and lung surgery, lung injury, rib fracture, chest tube placement, cognitive and neurological disorders, head or nose trauma, frequent sinus infections, or chemotherapy. Moreover, 96% of the participants in the control group and 96.2% in the intervention group (overall 96.1%) used nasal cannula (NC). Regarding mouth mask use, 52% in the control group and 69.2% in the intervention group had no history of using a mouth mask; that is, 58.8% of the participants did not use a mouth mask. The chi-square test showed no significant difference between the two groups in the use of mouth masks (P > 0.05). The mean and SD of the number of bypassed arteries in the control and intervention groups were 3.3 ± 0.7 and 3.3 ± 0.8 (P = 0.916); for surgery duration, they were 4.0 ± 1.0 and 5.0 ± 0.8 hours (P = 0.072); and for postoperative intubation time, they were 14.1 ± 6.1 and 14.6 ± 4.6 hours (P = 0.774), respectively.
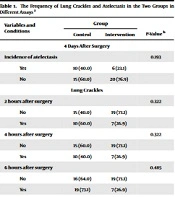
Table 1 shows the frequency of lung crackles and atelectasis in the two groups. Preoperational CXR examination revealed that none of the patients in the two groups had atelectasis. Moreover, there was no significant difference between the two groups concerning atelectasis four days after the surgery. Similarly, there was no significant difference between the two groups regarding crackles at different hours after the surgery. No crackle sound was detected in the lungs of any patient in the two groups during the lung auscultation at 18:00 on the third day.
| Variables and Conditions | Group | P-Value b | |
|---|---|---|---|
| Control | Intervention | ||
| 4 Days After Surgery | |||
| Incidence of atelectasis | 0.193 | ||
| Yes | 10 (40.0) | 6 (23.1) | |
| No | 15 (60.0) | 20 (76.9) | |
| Lung Crackles | |||
| 2 hours after surgery | 0.322 | ||
| No | 15 (40.0) | 19 (73.1) | |
| Yes | 10 (60.0) | 7 (26.9) | |
| 4 hours after surgery | 0.322 | ||
| No | 15 (60.0) | 19 (73.1) | |
| Yes | 10 (40.0) | 7 (26.9) | |
| 6 hours after surgery | 0.485 | ||
| No | 16 (64.0) | 19 (73.1) | |
| Yes | 19 (73.1) | 7 (26.9) | |
| 12 hours after surgery | 0.296 | ||
| No | 17 (68.0) | 21 (80.8) | |
| Yes | 8 (32.0) | 5 (19.2) | |
| 18 hours after surgery | 0.945 | ||
| No | 20 (80.0) | 21 (80.8) | |
| Yes | 5 (20.0) | 5 (19.2) | |
| 24 hours after surgery | 0.945 | ||
| No | 20 (0.80) | 21 (80.8) | |
| Yes | 5 (20) | 5 (19.2) | |
| 8 a.m. the day after surgery | 0.704 | ||
| No | 22 (88.0) | 21 (80.8) | |
| Yes | 3 (12.0) | 5 (19.2) | |
| 8 p.m. the day after surgery | 0.668 | ||
| No | 22 (88.0) | 24 (92.3) | |
| Yes | 3 (12.0) | 2 (7.7) | |
a Values are expressed as No. (%).
b Chi-square test
Table 2 shows the mean and SD of the VAS score for satisfaction with the breathing condition, SpO2 rate, and breathing rate in the two groups in different measurements. There were no significant differences in VAS scores between the two groups before and 4 days after the surgery or in the arterial blood oxygen rate before anesthesia induction and after extubation on the 2nd, third, and fourth days after the surgery. However, a significant difference was observed between the two groups regarding breathing rate before anesthesia induction, after extubation, and on the second day after the surgery. On the third and fourth days, there was no significant difference between the 2 groups.
| Variables and Time | Group | P-Value b | |
|---|---|---|---|
| Control | Intervention | ||
| VAS for satisfaction with the breathing condition | |||
| Preoperation | 8.7 ± 0.6 | 8.9 ± 0.1 | 0.074 b |
| 4th day after surgery | 8.9 ± 0.8 | 9.3 ± 0.8 | 0.097 b |
| P-value c | 0.180 | 0.061 | |
| Arterial oxygen saturation rate | |||
| Before surgery | 97.1 ± 0.7 | 97.3 ± 0.7 | 0.290 d |
| After extubation | 96.7 ± 1.6 | 97.0 ± 1.6 | 0.498 d |
| 2nd day after surgery | 96.8 ± 1.4 | 97.0 ± 1.3 | 0.551 d |
| 3rd day after surgery | 97.1 ± 1.4 | 97.5 ± 1.4 | 0.353 d |
| 4th day after surgery | 97.4 ± 1.4 | 98.0 ± 1.4 | 0.177 d |
| P-value e | 0.303 | ||
| Breathing rate | |||
| Before surgery | 20.8 ± 1.6 | 19.7 ± 1.5 | 0.016 d |
| After extubation | 21.9 ± 1.7 | 20.7 ± 1.9 | 0.020 d |
| 2nd day after surgery | 20.4 ± 1.5 | 19.5 ± 1.3 | 0.038 d |
| 3rd day after surgery | 19.5 ± 1.7 | 19.1 ± 1.4 | 0.317 d |
| 4th day after surgery | 19.3 ± 1.4 | 19.3 ± 1.1 | 0.970 d |
| P-value e | 0.002 | ||
a Values are expressed as mean ± SD.
b Mann-Whitney U test
c Wilcoxon test
dt-test
e Repeated measures analysis of variance
5. Discussion
Since the emergence of breathing exercises in the early 20th century, many studies have investigated the effect of these exercises on improving those wounded in action (38, 39), patients with respiratory diseases (15, 17), and traumatic injuries (39), and those undergoing chest and abdomen surgeries (40-42). The postoperative respiratory condition is essential because it increases hospitalization and mortality (6, 43). Still, few studies have examined the effect of the simultaneous use of different breathing exercises, and most have focused on the impact of these exercises separately (11, 19, 20, 22, 23). Therefore, this study was conducted to determine the effect of several breathing exercises (i.e., PLB, diaphragmatic breathing, incentive spirometry, and effective coughing) on the respiratory condition (incidence of atelectasis, SpO2, breathing rate per minute, lung auscultation, and patients' satisfaction with respiratory function) after CABG.
The findings indicated no significant difference between the two groups in SpO2 and atelectasis postoperatively. Moradian et al. reported a 20% incidence of atelectasis after performing breathing exercises, including deep breathing, coughing, and incentive spirometry. They did not demonstrate a significant difference in PaO2 and SpO2 before and after the surgery, which agrees with the findings of the present study (20). This study demonstrated no difference in the outcome of SpO2 using combined breathing exercises compared to routine care. There was a significant difference in breathing rate after extubation and on the second day after surgery, although there was no significant difference between the two on the third and fourth days. A comparison of the difference in the mean breathing rate between the two groups also indicated a considerable difference. In total, the fluctuation of breathing rate in the intervention group was less than in the control group. The breathing rate, its increase or decrease, indicates respiratory function. Breathing exercises that affect the breathing rate also impact respiratory function. In our study, combined breathing exercises resulted in less fluctuation in the breathing rate postoperatively. Amin et al. investigated the effect of pulmonary ventilation exercises in patients undergoing CABG. They found improvement in respiratory function influenced by incentive spirometry, which is consistent with the results of the present study (19). However, the present study investigated the simultaneous effect of several breathing exercises: PLB, diaphragmatic breathing, incentive spirometry, and effective coughing on improving respiratory function, and the results indicated an improvement in respiratory function.
The results revealed that crackles in the pulmonary auscultation of patients in the intervention group were less than that in the control group. The incidence of atelectasis in patients who received combined breathing exercises decreased by 40% compared to the control group. According to the VAS, compared to the control group, patients who received combined breathing exercises were satisfied with pulmonary function, although this difference was insignificant. Note that improving pulmonary function is a significant goal of postoperative care, particularly in heart surgery. Therefore, the lack of a significant difference in pulmonary function between the two groups is expected due to the provision of routine care in patients. Despite this issue, a relative improvement in pulmonary function was observed in the group receiving combined breathing exercises. Oshvandi et al. reported that the atelectasis incidence decreased due to breathing exercises, and pulmonary crackles in auscultation were also significantly different in the group receiving breathing exercises compared to the control group, indicating an improvement in respiratory function due to breathing exercises (11). Vitomskyi et al. also found an improvement in respiratory function based on the FEV1/FVC ratio (forced expiratory volume in one second / forced vital capacity) (23). Mahmoudi and Hojjati Zidashti also documented pulmonary function enhancement due to breathing exercises following heart surgery (22). The present study reported similar results; however, what differentiates the current research from the cited studies is that it investigated the effect of several breathing exercises on respiratory function. Moradian et al. reported that the incidence of atelectasis is 20%, lower than the incidence in our study. Moreover, they found no significant difference in SpO2 between the two groups, similar to the current study. However, they did not investigate the impact of PLB on SpO2 and the incidence of atelectasis (20). Note that in this study, the patients performed breathing exercises under the direct supervision of a researcher or nurse to ensure the correctness of the exercises.
5.1. Conclusions
This study demonstrated the relative superiority of combining different breathing exercises over a single one in improving respiratory function after surgery. Therefore, we recommend the combination of these exercises in post-CABG care.
5.2. Limitations
Access to patients was difficult due to the COVID-19 pandemic and the possibility of virus transmission. To resolve this limitation, we tried to minimize direct contact with the patients and observe all health protocols throughout the study.