1. Background
Psoriasis is a chronic, multifactorial immune-mediated hyperproliferative inflammatory skin condition (1). It is relatively common with a prevalence rate of 1% - 3% of the world population accounting for 2% - 6% of yearly dermatology new clinic attendees in Malaysia (2, 3). Psoriasis is beyond skin deep and had been linked to other co-morbidities, including metabolic syndrome (MetS), obesity, and non-alcoholic fatty liver disease (NAFLD).
NAFLD is a spectrum of liver diseases, ranging from hepatosteatosis, which is harmless and characterized by non-inflammatory intracellular fat deposition, to non-alcoholic steatohepatitis (NASH), which occurs when fatty infiltration is accompanied by inflammation, with evidence of cellular injury and fibrosis (4). NAFLD may progress to cirrhosis and is a possible cause of cryptogenic cirrhosis (5). NAFLD is seen worldwide with a prevalence of 6% - 45%, which is closely related to MetS, type 2 diabetes mellitus (T2DM), dyslipidemia, and central obesity (6). In Malaysia, the prevalence of NAFLD in the suburban population was 22.7%, and in patients with T2DM, the prevalence of NAFLD was almost 50% (7, 8).
Although the pathogeneses of both entities are not completely clear, it is thought that the increased levels of pro-inflammatory adipokines (e.g., TNF-α and IL-6), and hepatokines, with the presence of decreased levels of anti-inflammatory adipokines (e.g., adiponectin) play a possible role in the chronic, low grade inflammation in both conditions. These imbalances could result in insulin resistance and facilitate the progression of NAFLD in psoriasis patients (9).
FibroScan is a clinically validated device using transient elastography indicated for the non-invasive measurement of liver stiffness (E) and controlled attenuation parameter (CAP), which allows the assessment and quantification of liver steatosis. It has a good correlation with the severity of steatosis and measures the liver volume up to 100 times larger than a liver biopsy. CAP measurement is not affected by liver fibrosis (10, 11). Although hepatobiliary ultrasound is the more common method for the screening of NAFLD, in the setting of mild steatosis, acuity decreases dramatically. Ultrasound also has substantial inter- and intra-observer variability (12). While transient elastography is operator independent, it provides reliable and reproducible quantitative measurements, and results are immediately available. With these advantages, transient elastography by using FibroScan was chosen as the mode of detecting NAFLD in our study. Detection of NAFLD in psoriasis patients will have clinical implications in preventive and treatment strategies.
2. Objectives
This study was performed to determine the prevalence of NAFLD in patients with psoriasis attending tertiary dermatology outpatient clinics in Kuala Lumpur, Malaysia, using CAP in transient elastography and to identify its predisposing factors.
3. Methods
An observational cross-sectional study was conducted prospectively on consecutive psoriasis patients aged > 18 years in two dermatology tertiary centers in Kuala Lumpur (Hospital Kuala Lumpur and UKM Medical Centre) from November 2015 to May 2016. We excluded patients who had underlying secondary causes of chronic liver disease, patients taking traditional supplements, and patients currently or for the past three months on treatment with hepatotoxic medication. Patients who had excessive alcohol intake (>21 units of alcohol per week for males and >14 units of alcohol per week for female patients) and those with contraindication for Fibroscan examination (e.g., pregnant, ascites) were also excluded.
A comprehensive history and physical examination, including demographic data, smoking and drinking habits, height, weight, blood pressure (BP), and waist circumference (WC), were obtained. In this study, metabolic syndrome is defined using NCEP-ATP III 2005 criteria, revised for Asians, which includes the presence of at least three risk factors (WC of ≥ 90 cm for males, ≥ 80 cm for females, systolic BP ≥ 130 and/or diastolic BP ≥ 85 or on treatment for hypertension, FBG ≥ 5.6 mmol/L or on treatment for elevated glucose). BMI classification used is based on the classification for adult Asians, as stated in the Clinical Practice Guidelines on Management of Obesity 2004, Ministry of Health, Malaysia.
The severity of psoriasis was assessed using the Psoriasis Area and Severity Index (PASI) by a single assessor. Patients with psoriatic arthritis (PsA) were classified according to Moll and Wright criteria.
Blood sample for full blood count consisting of hemoglobin, total white cell count, and platelet level, liver function test (LFT), comprised of total protein, albumin, bilirubin, alanine transaminase (ALT), alkaline phosphatase (ALP) and aspartate transaminase (AST); fasting serum lipids (FSL), comprising of total cholesterol, LDL-cholesterol, HDL-cholesterol and Triglyceride (TG), and fasting blood glucose (FBG) were sent for analysis at enrolment. All the participants were also screened for viral hepatitis by measuring hepatitis B surface antigen (HBsAg) and hepatitis C antibody (anti-HCV) with the enzyme immunoassay.
Transient elastography procedure was performed in the Gastroenterology Unit at UKM Medical Centre by a single experienced consultant gastroenterologist. For the purpose of this study, NAFLD was diagnosed using the CAP, which is available in the transient elastography using FibroScan 502. CAP is computed from ultrasounds acquired for liver stiffness measurement and is expressed in decibel per meter (dB/m). It is calculated only if liver stiffness measurement is valid. CAP is correlated with the grade of steatosis, with a cutoff value of < 215 dB/m in the healthy population, 215 – 300 dB/m in mild steatosis and > 300 dB/m in moderate to severe steatosis (13).
3.1. Statistical Analysis
Analysis of the research data was performed using SPSS version 23.0. The majority of our continuous variables were not normally distributed; thus, non-parametric tests were used for further data analysis. The tests used were chi-square, Fisher’s exact test, Mann-Whitney U test, and Spearman rank order correlation. Multivariate analysis (logistic regression) was used to analyze the predictors of NAFLD. Data were analyzed in totality and further subdivided according to the presence or absence of NAFLD. A 2-tailed P-value of < 0.05 is considered statistically significant.
4. Results
Of the 152 psoriasis patients screened during this six-month study, 33 patients were excluded initially, mainly due to the current usage of hepatotoxic drugs. Two patients had excessive alcohol intake, two patients had positive hepatitis B surface antigen on the screening test, and another patient was in her second trimester of her pregnancy. Twelve patients withdrew from the study because they were unable to comply with the study requirements. Therefore, 109 patients with psoriasis were finally included in data analysis.
In this study, the male to female ratio was 3:2. Patients were mostly overweight with a median (IQR) BMI of 25.73 kg/m2 (4.86 kg/m2). Most of the patients had mild disease severity with a median (IQR) PASI score of 8.7 (8.9). Only five patients had psoriatic arthropathy.
The prevalence of NAFLD in this study was 85.3%. Out of this, 40 patients had mild NAFLD, while another 53 patients had moderate to severe NALFD.
In Table 1, psoriasis patients with NAFLD had significantly higher weight, BMI, and waist circumference, when compared to those patients without NAFLD. They were comparable in terms of age, ethnicity, gender, height, cigarette smoking and alcohol intake. The two groups were comparable with respect to age at the onset of psoriasis, duration of psoriasis, severity of psoriasis, presence of PsA, hypertension, diabetes and dyslipidemia. There was a significant association between MetS and NAFLD.
| Clinical Characteristics | Psoriasis Patients with NAFLD, (N = 93) | Psoriasis Patients Without NAFLD, (N = 16) | P Value |
|---|---|---|---|
| Weight, kg | 71.0 (19) | 57.6 (12) | < 0.0005* |
| BMI, kg/m2 | 26.23 (4) | 22.76 (4) | < 0.0005* |
| Waist circumference, cm | 93 (11) | 80 (19) | < 0.0005* |
| Age, y | 53 (23) | 52.5 (39) | 0.732 |
| Height, m | 1.65 (0.14) | 1.60 (0.08) | 0.176 |
| Co-morbidities, n | 40 (43) | 10 (62.5) | 0.148 |
| Metabolic syndrome, n | 51 (54.8) | 2 (12.5) | 0.002* |
| Diabetes mellitus, n | 23 (24.7) | 3 (18.8) | 0.757 |
| Dyslipidemia, n | 19 (20.4) | 6 (37.5) | 0.194 |
| Hypertension, n | 24 (25.8) | 5 (31.3) | 0.760 |
| Psoriatic arthropathy, n | 5 (5.4) | 0 (0.0) | 1.000 |
| PASI score | 0.516 | ||
| < 10 | 50 (54) | 10 (63) | |
| ≥ 10 | 43 (46) | 6 (37) |
aValues are expressed as No. (%) or median (IQR).
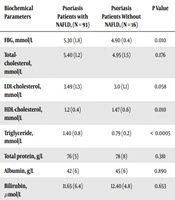
Psoriasis patients with NAFLD had significantly higher values of ALT than their counterparts without NAFLD, although they were still within the normal reference range. The levels of FBG and triglycerides were significantly higher, while the HDL-cholesterol level was significantly lower in the group of patients with NAFLD. The other biochemical parameters did not differ between the groups, as seen in Table 2.
| Biochemical Parameters | Psoriasis Patients with NAFLD, (N = 93) | Psoriasis Patients Without NAFLD, (N = 16) | P Value |
|---|---|---|---|
| FBG, mmol/L | 5.30 (1.8) | 4.90 (0.4) | 0.010 |
| Total-cholesterol, mmol/L | 5.40 (1.2) | 4.95 (1.5) | 0.176 |
| LDL-cholesterol, mmol/L | 3.49 (1.3) | 3.0 (1.1) | 0.058 |
| HDL-cholesterol, mmol/L | 1.2 (0.4) | 1.47 (0.6) | 0.010 |
| Triglyceride, mmol/L | 1.40 (0.8) | 0.79 (0.2) | < 0.0005 |
| Total protein, g/L | 76 (5) | 78 (8) | 0.381 |
| Albumin, g/L | 42 (6) | 45 (6) | 0.890 |
| Bilirubin, µmol/L | 11.65 (6.4) | 12.40 (4.8) | 0.653 |
| ALT, U/L | 28.5 (26) | 15.0 (6) | 0.001 |
| ALP, U/L | 79 (21) | 75 (20) | 0.722 |
| AST, U/L | 24 (13) | 20 (3) | 0.62 |
| Hemoglobin, g/dL | 14.25 (2.6) | 13.2 (2.0) | 0.172 |
| Platelet, ×109/L | 284.5 (76) | 312 (143) | 0.762 |
Abbreviations: ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate aminotransferase; FBG, fasting blood glucose; FSL, fasting serum lipid.
aValues are expressed as median (IQR).
There was a significant positive correlation between the severity of NAFLD and weight, BMI and waist circumference as depicted in Table 3. A weak positive correlation was also observed between the severity of NAFLD and the severity of psoriasis (measured by PASI) as well as the level of systolic blood pressure.
| Clinical Characteristics | rsb | P Value |
|---|---|---|
| Age | -0.038 | 0.694 |
| Weight | 0.574 | < 0.0005 |
| BMI | 0.642 | < 0.0005 |
| Waist circumference | 0.602 | < 0.0005 |
| PASI score | 0.202 | 0.035 |
| Age at diagnosis | -0.021 | 0.829 |
| Duration of psoriasis | 0.075 | 0.438 |
| Systolic blood pressure | 0.191 | 0.047 |
| Diastolic blood pressure | 0.146 | 0.130 |
aSpearman’s correlation.
brs = Correlation coefficient.
Factors that have significant correlation in the bivariate analysis were further analyzed using multivariate logistic regression analysis. The factors that were related to psoriasis include BMI, MetS, FBG, HDL-cholesterol, TG, ALT and SBP. Weight and waist circumference were not included as these variables are correlated with BMI.
Table 4 shows the variables that significantly predict the probability of having NAFLD. Nagelkerke R square is 0.601 indicates that 60.1% of the variance of having NAFLD is being explained by the significant factors. Among these three factors, BMI is the strongest predictor followed by TG and PASI (based on Wald values). Those who have one unit higher BMI, TG and PASI have 1.63, 130 and 1.13 times higher odds to have NAFLD, respectively.
| Predictors | β | Wald | P Value | OR (95% CI) |
|---|---|---|---|---|
| BMI | 0.489 | 6.736 | 0.009 | 1.630 (1.127 - 2.357) |
| Triglyceride | 4.873 | 6.336 | 0.012 | 130.737 (2.94 -5812.75) |
| PASI | 0.129 | 4.087 | 0.043 | 1.138 (1.004 - 1.290) |
| Metabolic syndrome | -0.782 | 0.425 | 0.515 | 0.457 (0.044 - 4.805) |
| HDL-cholesterol | 2.370 | 2.405 | 0.121 | 10.693 (0.535 - 213.738) |
| FBG | 1.084 | 1.421 | 0.233 | 2.958 (0.497 - 17.595) |
| ALT | 0.18 | 0.245 | 0.620 | 1.018 (0.947 - 1.095) |
| Systolic Blood Pressure | -0.013 | 0.251 | 0.616 | 0.987 (0.937 - 1.039) |
Abbreviations: ALT, alanine transaminase; BMI, body mass index; FBG, fasting blood glucose; PASI, Psoriasis area severity index.
aBolded P values are significant.
The chi-square test was performed to examine the relationship between the severity of steatosis and the presence of metabolic syndrome. The relationship between these variables was strongly significant, χ2 (2, n = 109) = 34.81, P < 0.001. The chi-square test was also used to examine the association between the severity of psoriasis and the presence of metabolic syndrome. The relationship between these variables was also significant, χ2 (2, n = 109) = 6.65, P = 0.036.
5. Discussion
To our knowledge, this is the first study to detect NAFLD using CAP on transient elastography in a cohort of psoriasis patients. We found a high prevalence of NAFLD in our Malaysian psoriasis patients (85.3%). This figure is much higher compared to previous studies conducted among psoriasis patients in the western population, which ranges from 46.2% - 59.2% (14-17). This prevalence rate is reported to be 4%, 17%, and 65% in Taiwan (18), 17% South India (19), and Iran (20), respectively.
The prevalence of NAFLD in the normal population in Malaysia was only 22% (8), thus, demonstrating that the prevalence of NAFLD among our psoriasis population was almost four folds higher than the normal Malaysian population.
One of the reasons for this higher prevalence of NAFLD seen in our study was maybe due to the higher sensitivity and specificity of transient elastography in detecting NAFLD compared to the conventional method with abdominal ultrasound scan. By comparison, most of the previous studies performed in North America and the European countries used abdominal ultrasound to detect NAFLD (14-17).
A much lower prevalence of fatty liver was defined by ICD-9-CM coding (code 571.8) (4% of 51800 patients with psoriasis) in a study of psoriasis population performed by the National Health Insurance Database in Taiwan (18). However, a study in a predominantly rural population in South India detected NAFLD on the basis of abnormal liver enzymes in the presence of hepatic steatosis on ultrasonogram examination, amounting to a prevalence of only 17.4% out of 333 adult psoriasis patients (19).
The high prevalence of NAFLD in our cohort of patients could also be attributed to the mostly overweight and obese patients, with more than half meeting the criteria for MetS. Furthermore, this was in line with the Malaysian Psoriasis Registry in Malaysia, which reported > 50% of adult psoriasis patients were overweight and obese (21).
In our study, we found that NAFLD in psoriasis patients was associated with higher weight, BMI, waist circumference, and MetS. These findings were expected and comparable with previous studies, although different classifications were used for the definition of BMI and MetS in different cohorts of patients from various regions of the world (14, 19, 22).
We did not find any significant difference in the severity of psoriasis between patients with NAFLD and those without NAFLD. This contradicts the finding by Gisondi et al. (15) where the frequency of NAFLD was remarkably greater in patients with severe psoriasis (PASI ≥ 10). This difference could be explained by the fact that in our study, the majority of patients had mild to moderate psoriasis, while in that Italian study > 50% were in the severe spectrum. In our study, patients with more severe disease on hepatotoxic drugs such as methotrexate and acitretin were excluded. Nevertheless, we did observe a weak positive correlation between the severity of psoriasis and severity of NAFLD.
Notably, there was no significant difference in the presence of psoriatic arthropathy (PsA), which may be due to our small number of patients with PsA, as most of them were being treated with systemic agents and were excluded. A systematic review and meta-analysis on the risk of NAFLD in patients with psoriasis showed that the risk of NAFLD was significantly greater in patients with psoriatic arthritis (three studies; n = 505 patients; OR = 2.25, 95% CI = 1.37 - 3.71) and in patients with moderate to severe psoriasis compared to those with mild psoriasis (two studies; 51 930 patients, OR = 2.07, 95% CI = 1.59 - 2.71) (22).
In terms of biochemical markers, those related to MetS were of significant difference in these two groups. In the group of psoriasis patients with NAFLD, the serum level of FBG and triglycerides were significantly higher, while the HDL-cholesterol level was significantly lower. This could be explained by the pathogenesis behind the metabolic syndrome. Insulin resistance and hyperinsulinemia increase the excretion of triglycerides by the liver, resulting in the elevated serum levels of triglycerides (23, 24). These findings correspond to the significantly higher percentage of MetS in the psoriasis group with NAFLD in comparison with the group without NAFLD (14, 15, 19).
The serum ALT level was significantly higher in the group with NAFLD, although most patients (88%) had levels within the normal reference range. The degree of aminotransferase elevation does not predict the degree of hepatic inflammation or fibrosis, and a normal alanine aminotransferase does not exclude clinically important hepatocyte injury (25, 26). Therefore, the presence of NAFLD with normal liver enzymes should be taken into consideration when choosing the appropriate treatment, which is potentially hepatotoxic.
5.1. Conclusions
A high prevalence of NAFLD (85.3%) was detected in psoriasis patients on transient elastography, despite normal biochemical profile. It is important to screen for NAFLD in all psoriasis patients, especially those with high BMI, MetS and severe psoriasis. Early detection of asymptomatic NAFLD is essential to initiate appropriate management and prevent future complications of NAFLD.