1. Background
Acne vulgaris is the most common inflammatory skin disease, which is primarily observed in adolescents. It is characterized by comedones, papules, pustules, nodules, and cysts on the face, back, chest, chin, and body skin (1). Acne vulgaris affects about 80 percent of people worldwide (2). Four major factors, including excessive sebum production, hyper-cornification of the pilosebaceous canal, abnormal colonization of Propionibacterium acnes, and inflammation, play a role in the pathogenesis of the disease (3). As one of the most common treatment agents, isotretinoin can be used for the acne vulgaris treatment and can resolve major etiologic factors in acne pathogenesis. However, it leads to oxidative damage (4, 5).
The hydroxyl radical (OH●) as the oxygen free radical is the most reactive member that reacts with biological molecules such as DNA, which is an important target for free radicals (6). Herein, 8-hydroxy-2’-deoxyguanosine (8-OHdG) is the most common base damage marker for DNA (7). The human 8-oxoguanine DNA glycosylase 1 (hOGG1) enzyme plays a role in a base excision repair mechanism and can repair 8-OHdG (8).
Isotretinoin is known to cause oxidative damage. However, hOGG1, as an enzyme used for the 8-OHdG repair mechanism, has not been evaluated in acne. Moreover, the effects of 8-OHdG and levels of its DNA repair enzyme, hOGG1, are unclear on acne vulgaris before and after the isotretinoin treatment.
2. Objectives
We evaluated the effect of 8-OHdG and its repair enzyme, hOGG1, on acne vulgaris before and after the isotretinoin treatment.
3. Methods
Study population: In the current study, blood samples were taken from individuals referring to the Department of Dermatology at the Health Practice and Research Hospital affiliated to the Mustafa Kemal University. Our study was approved by the Clinical Research Ethics Committee of the Mustafa Kemal University. Patients over 18 years of age, who were diagnosed with acne vulgaris, were included in the study (n = 43). The patients with acne vulgaris were treated with isotretinoin (1 mg/kg/day) for six months. Blood samples from the patients were put into serum separating tubes and centrifuged for 10 minutes at +4ºC. Supernatants were portioned, and serum samples were stored at -80ºC until analysis.
Biochemical measurements: The 8-OHdG and hOGG1 levels were measured from the serum samples with the enzyme-linked immunosorbent assay (ELISA) method using a spectrophotometer (Thermo, Multiskan Go, Type: 1510, Finland). Commercial kits were used for the analysis of 8-OHdG and hOGG1 enzymes (Cell Bio Labs; USA and Cloud-Clone Corp; USA, respectively).
Statistical analysis: The GraphPad Prism 5 statistics programmer was used to evaluate all the data. Statistical analyses were performed using a t-test. Differences were considered statistically significant when P < 0.05.
4. Results
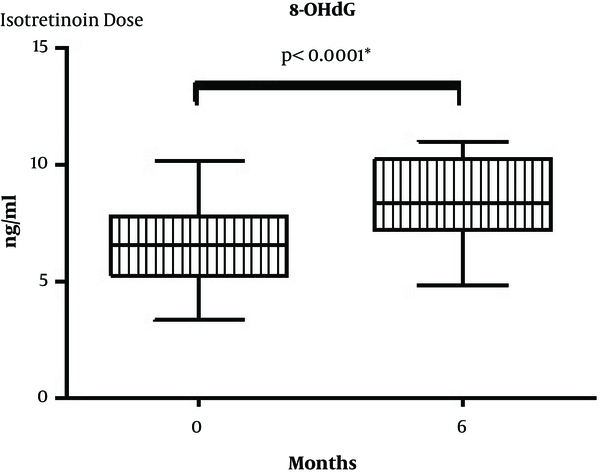
In our study, the serum 8-OHdG (P < 0.05; P < 0.0001*) and hOGG1 (P < 0.05; P = 0.04*) levels were found to be significantly higher in the sixth month after the isotretinoin treatment (Figures 1 and 2). However, there was no statistical difference between the levels of 8-OHdG and hOGG1 in terms of time (P > 0.05).
5. Discussion
Reactive oxygen species (ROS), including the superoxide radical (O2●), the hydrogen peroxide (H2O2), and OH● are produced by active neutrophils (9). The alteration of sebum composition leads to the irritation and destruction of a follicular wall responsible for inflammatory processes of acne vulgaris (9, 10). In addition, isotretinoin causing a form of ROS and DNA damage, is administrated as acne vulgaris therapy (4). Thus, we evaluated the risk of using isotretinoin for DNA damage in acne vulgaris patients in the current study. For this purpose, we assessed the oxidative DNA damage level (the measures of the 8-OHdG and hOGG1 levels) in acne vulgaris before and after the isotretinoin treatment.
There is only one previous study that evaluated the oxidative DNA damage level before and after the isotretinoin treatment in patients with acne disease (11). However, they did not assess the hOGG1 level as well as the relationship between the hOGG1 and 8-OHdG levels (11). In the mentioned study, patients with cystic acne (n = 18) and healthy controls (n = 22) were included. The patients were evaluated before and 45 days after the isotretinoin (0.5mg/kg per day) treatment. The 8-OHdG level was demonstrated to be two-fold higher in the patients after the 45-day isotretinoin treatment (0.21 ± 0.03 ng/mL) and three-fold higher when compared to the controls (0.07 ± 0.01 ng/mL; P < 0.0001) (11).
Song et al. investigated ambient fine and ultrafine particles in urban air to demonstrate the oxidative DNA damage level between children with and without eczema. The results showed a higher 8-OHdG level in children without eczema (urinary 8-OHdG μg/g creatinine 12.0 ± 6.3 and 10.1 ± 5.5 for children with and without eczema, respectively) (12). A recent investigation by Nakai et al. indicated that the 8-OHdG level was high in psoriasis patients and that the elevated urine 8-OHdG level was statistically significant (P = 0.034) (13). Furthermore, Basavaraj et al. demonstrated that the serum 8-OHdG level was high in psoriasis patients (P < 0.05) (14).
As mentioned above, hOGG1 plays a role in the repair of damaged DNA. However, no study has been so far carried out on the association between hOGG1 and acne vulgaris. Murtas et al. investigated the 8-OHdG and hOGG1 expressions in tumor tissues and demonstrated low expression of the nuclear 8-OHdG. However, hOGG1 was reported not to have a relationship with cancer-specific survival (15). A study by Huang et al. found that hOGG1 expression significantly decreased in basal cell carcinoma (BCC) lesions compared to the epidermis of neonatal foreskins (P < 0.0001), normal sun exposure skin (P < 0.001), normal skin adjacent to BCC (P < 0.0001), and epidermis overlying BCC lesions (P < 0.0001) (16). We demonstrated the association of hOGG1 and acne vulgaris with isotretinoin administration.
The use of isotretinoin therapy for acne vulgaris as an inflammatory skin disease causes DNA damage. Moreover, 8-OHdG is a biomarker for DNA damage, and hOGG1 is a biomarker for 8-OHdG repair. In the light of these facts, we, for the first time, evaluated the levels of both 8-OHdG and hOGG1 in acne vulgaris before and after the isotretinoin treatment. We observed that 8-OHdG and hOGG1 had statistically significant high levels after the isotretinoin treatment. According to our results, the 8-OHdG level increased in acne vulgaris patients after isotretinoin administration. Furthermore, the hOGG1 level increased for DNA damage repair. However, the increased level was not sufficient for DNA renovation. The healing likely continues after the dose of isotretinoin is reduced for DNA, which needs to be further clarified in future studies.