1. Introduction
Numerous autogenous cells and membranes have gained interest in the field of reconstitution and regeneration of lost tissues. In this series, the periosteum is one of the extensively studied and established membranes in the medical literature (1). The periosteum is a vascular connective tissue that covers the external surface of the bone, comprising of two layers. The inner layer consists of osteoprogenitor and osteoblasts; however, the outer layer shows the presence of dense collagen fibers and their progenitor cells. The osteoprogenitor cells from the inner layer are responsible for instigating the cell differentiation leading to bone repair and remodeling (2).
The literature shows that the periosteum consists of three allocated zones. The first zone predominantly comprises osteoblasts; they represent 90% of the total cell population in this layer, with collagen fibrils constituting 15% of the volume. The second zone primarily consists of fibroblasts, and endothelial cells constitute the remaining spaces. The third zone is fibrous indicating the presence of the highest amount of collagen fibers and fibroblasts (3). The presence of such an extensive population of osteoprogenitor cells makes it an attractive tool for periodontal regeneration.
The biologic principle involved in guided tissue regeneration is to stimulate the growth of the specific cell population to develop new bone, cementum, and periodontal ligament. The periosteum has caught attention for the same reason because it is capable of producing progenitor cells which is an assured way of stationing osteoblasts cell in an intrabony defect (4). This was supported by studies that have used periosteal pedicle flap as a barrier membrane in the treatment of intrabony defects and showed significant improvement in the results (5, 6). Therefore, the present case report shows the periosteum use as a barrier membrane.
2. Case Presentation
A 25-year-old female patient was referred to an outpatient department of periodontology with a chief complaint of food lodgement and bleeding from gums since 2 months ago. The patient intended to go for orthodontic treatment for protruded maxillary anteriors. Complete periodontal examination was carried out, and detailed medical and dental history was obtained. The patient revealed a history of periodontal therapy; however, the patient was systematically healthy with no contraindications for surgical periodontal treatment.
Clinical examination revealed generalized periodontal pockets that were deeper in posteriors, generalized bleeding on probing, and grade I mobility in lower anteriors suggestive of bone loss. Radiographic examination showed the presence of vertical and horizontal bone loss. Therefore, phase I periodontal therapy was given to the patient to evaluate the response to phase II therapy, wherever required. On revaluation, a pocket of 6 mm was observed in relation to the right first maxillary molar (16) distally. Modified flap operation, along with bone graft and periosteum as barrier membrane, was planned. The patient was informed about the procedure, and informed consent was obtained from the patient.
2.1. Surgical Procedure
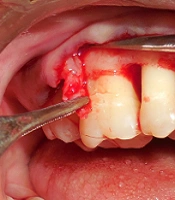
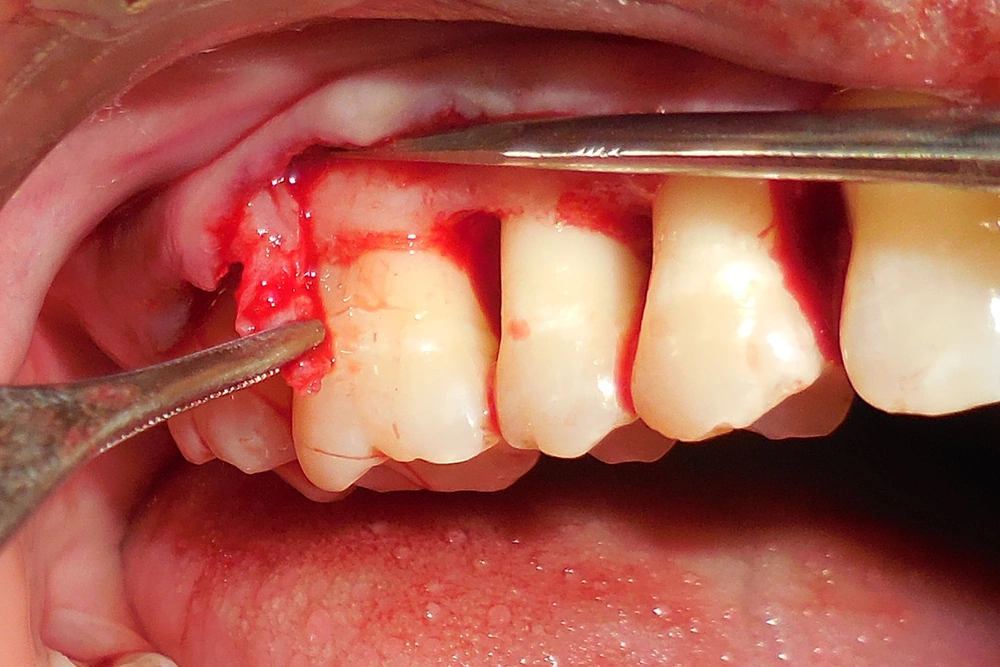
The extraoral surface was made sterile using a 5% povidone-iodine solution. Local anesthetic solution (2% Xylocaine hydrochloride, epinephrine 1:80,000) was injected in the area of surgery after preprocedural rinse with 0.2% chlorhexidine. A crevicular incision was made to raise the mucoperiosteal flap. A full-thickness flap was elevated. Complete debridement and root planing were performed. The area was copiously irrigated using 0.9% normal saline solution. Then, a buccal flap adjacent to the defect area was incised from the inner surface in such a manner that a trapezoidal tissue of the periosteum and underlying connective tissue was detached while avoiding any tear on the external surface of the flap. Once the periosteal pedicle was obtained (Figure 1), a resorbable suture (4-0 Vicryl) was passed through it. A bone graft was placed in the defect, and the periosteal pedicle was sutured. The flaps were then sutured using 3 - 0 silk sutures. A periodontal pack was placed over the surgical area. The patient was given routine postoperative instruction and prescribed antibiotics and analgesics. She was advised to refrain from brushing up to the next appointment.
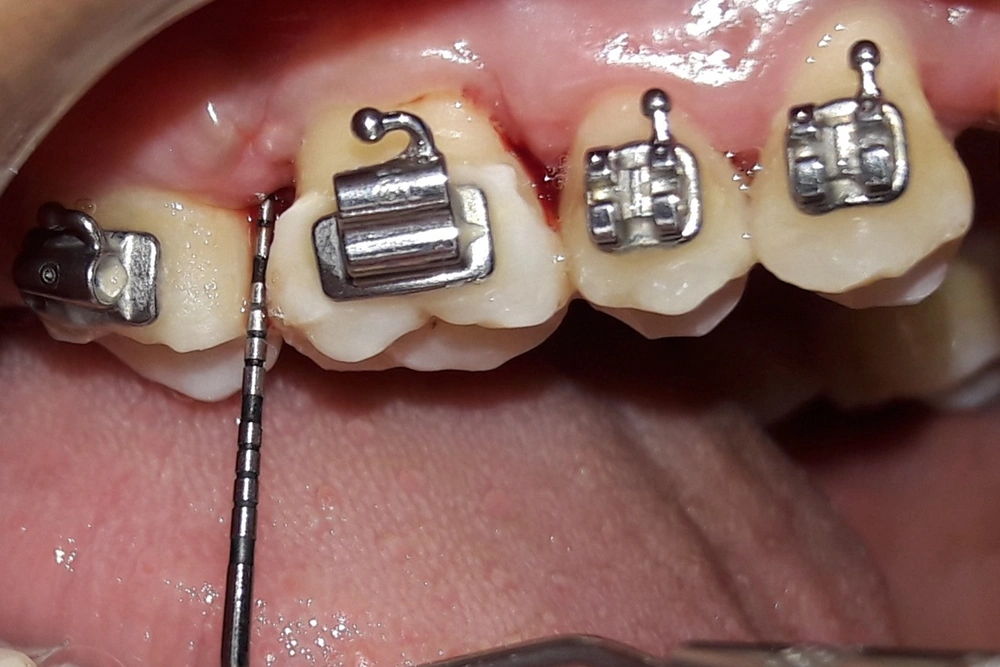
The patient was recalled after 7 days for suture removal. Healing was uneventful; the patient reported no discomfort at the recall appointment. She was advised to continue her routine oral hygiene habits. She was kept in the maintenance phase to reinforce oral hygiene habits and monitor any improvement or deterioration. After 6 months, the surgical site showed good healing with reduced probing depth and bleeding on probing, suggestive of a positive outcome (Figure 2).
3. Discussion
The regenerative materials that can provide osteoinductive effects or progenitor cells have always held their supremacy in periodontal regeneration. The periosteum has the added advantage of being autogenous apart from the ability to supply osteoprogenitor cells. The pedicle obtained will retain its blood supply as it is attached to the flap. Mahajan suggested that periosteal progenitor cells have the ability to differentiate into fibroblasts, osteoblasts, chondrocytes, adipocytes, and skeletal myocytes. They also have the capability to produce cementum with periodontal ligament fibers and bone (2). This might result in the continuous supply of cells in periodontal wounds combating the vascular space created by root and barrier (7). The present case report suggests defect resolution in terms of periodontal healing. The probing pocket depth has been reduced from 6 to 3 mm. The gingival biotype has also changed from thin to thick. The radiographic bone filling also appeared at the end of 6 months.
Gamal et al. has described the method of achieving periosteal pedicle graft where they have raised a supra periosteal partial-thickness flap. The attached periosteum was then released to position it in the defect (8). The present case report used the periosteum from under the surface of the elevated flap. This was performed because the gingival biotype was thin, and the elevation of a partial-thickness flap can endanger the flap if any unfortunate nicks happen, resulting in tearing the flap. Therefore, the full-thickness flap was raised, and the periosteum was obtained. Saimbi et al. concluded that the periosteum is an effective barrier membrane that can significantly improve pocket depth, clinical attachment level, and bone defects in intrabony defects (9). Verma et al. conducted a study using the periosteum in the treatment of buccal grade II furcation defects and obtained improved clinical parameters in horizontal and vertical dimensions (6).
The use of the periosteum from the site involved in the surgical area eliminates the need for a second surgical area. It does not pose a problem in terms of availability and size. The use of osteoprogenitor cells in the field of tissue engineering can be highly promising. The long-term effects of periosteal pedicle grafts and their comparison to other regenerative materials should be evaluated to augment the interest in this autogenous membrane.
3.1. Conclusions
The present case report shows the efficacy of the periosteum in the treatment of intrabony defects. Further clinical trials should be carried out to assess the long-term results and the potential of the periosteum as a barrier membrane.