1. Background
Obstructive sleep apnea syndrome (OSAS) is shaped by total or partial obstruction of the upper airway during rest like sleeping which results in a decrease in arterial oxygen saturation and transient awakenings (1). OSAS is more common among males who are between 45 - 65 years old and is the most common respiratory disorder related to sleep. Its prevalence in the general adult population ranges from 6% to 17% and can be as high as 49% with advanced age (2). However, this rate is 36% in diabetic sufferers, 63% in sufferers with resistant hypertension, and 71% in sufferers with congestive coronary heart failure and in those with cerebrovascular strokes in acute phase. The sedentary lifestyle has been improving fast in recent years throughout the world along with cigarette and alcohol use, obesity and aging (3-6).
OSAS may present with various symptoms such as chronic fatigue, morning headache, witnessed apnea, excessive daytime sleepiness or difficulty in concentrating. Also, if OSAS is left untreated, it has some risks such as decreased quality of life, serious medical complications, and increased mortality. Patients with suspected sleep apnea should undergo polysomnography testing (7-9).
Apnea is a temporary stop of breathing for at least 10 seconds during sleep. Hypopnea is a partial obstruction of airflow and is not a temporary stop of breathing. Hypopnea is 30% discount in breath amplitude and 4% discount of oxygen saturation or 50% discount in breath amplitude and 3% reduction of oxygen saturation for more than 10 seconds (10). The variety of apnea-hypopnea episodes in line with hour is known as apnea-hypopnea index (AHI). According to the international clinical sleep disorders-3 (American Academy of Sleep Medicine, AASM); based on AHI and oxygen saturation, severity of OSAS is classified as: (i) mild (AHI is 5 - 15 and oxygen saturation is 85% - 95%); (ii) moderate (AHI is 15 - 30 and oxygen saturation is 65% - 84%); and (iii) severe (AHI is > 30 and oxygen saturation is < 64%) (10).
The most essential complications of OSAS are connected to the cardiovascular system and OSAS may cause or exacerbate myocardial infarction, congestive heart failure, and atrial fibrillation (11-14). Nocturnal arrhythmias are quite common in OSAS. The most common tachyarrhythmia is early ventricular extrasystole (VES) (15, 16).
Continuous positive airway pressure (CPAP) tool is usually suggested for the OSAS cure (13, 17). Fifty percent of the patients cannot tolerate the CPAP appliance or cannot use it as effective as they should (18). Oral apparatus (such as mandibular advancing device) is recommended as an alternative for patients with mild to moderate OSAS who cannot tolerate CPAP. Patient-specific oral packages are prepared based on patient’s characteristics to lessen top airway fall apart and amplify the airway at some stage in sleep (19, 20). Weight loss and positional sleep techniques may be regarded as additional medical measures. Unfortunately, most patients do not benefit from medical treatment.
UPPP was conducted in 1981 by Fujita in order to relieve retropalatal obstruction (anatomical narrowing of the soft palate and uvula) on patients with OSAS (21). In this application, the inferior part of the uvula and soft palate are resected after tonsillectomy. Tonsil plica and the soft palate mucosa in the excised region are mutually sutured. In another study, it was reported that there was a significant regression in cardiovascular system-related complications after UPPP (22). Removing the obstruction in the upper airway is the main principle of surgical treatment of OSAS. Uvulopalatopharyngoplasty (UPPP) is the most common surgical procedure performed by otolaryngologists for the treatment of OSAS (23, 24). In the meta-analysis studies, the success rate for UPPP (with polysomnography) was reported as 50%. However, results from local studies have reported success rates up to 83% (9, 25). In spite of partial reductions in AHI and limited success as a stand-alone treatment option for OSA, UPPP is still an important therapeutic option for many patients who refuse or tolerate CPAP (24). Studies on UPPP and OSAS have extremely focused on its effectiveness and on determining the best patient choice, and there are few data evaluating the effects of UPPP on reducing the incidence of cardiovascular complications in OSAS (14).
2. Objectives
The objective of this study was to investigate possible changes in arrhythmias after UPPP in patients with moderate OSAS who continued having cardiac arrhythmias despite the use of antiarrhythmic drugs and those who could not tolerate the CPAP device. In addition, we evaluated the correlation between arrhythmias before and after UPPP.
3. Methods
3.1. Patients and Design
This retrospective study included patients from cardiologists and otolaryngologists working as co-authors in this protocol at the Bahat Hospital. The patients who have OSAS and cardiac arrhythmias despite antiarrhythmic treatment (calcium channel blockers and beta blockers) at optimal doses between 2008 - 2017 by the cardiology clinic of the hospital have been included in the study and consulted by the ear, nose, and throat (ENT) clinic.
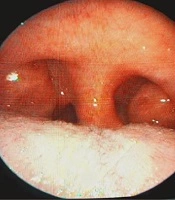
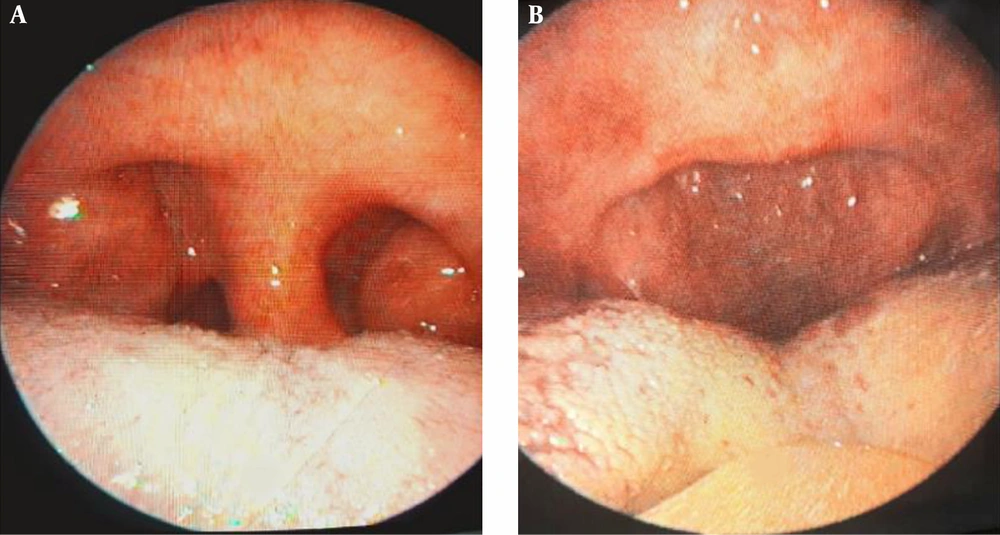
Patients with OSAS were evaluated with detailed ENT examination, fiberoptic laryngoscopy, Muller maneuver, lateral cephalogram, computed tomography, and magnetic resonance imaging. Finally, 38 patients who did not accept oral apparatus or CPAP treatment were selected and UPPP operation were performed. In addition to arrhythmias, 11 patients had mild hypertension in mild heart diseases and other cardiac diseases were excluded from the study. All patients have undergone UPPP. The operations were performed under general anesthesia (Pentothal 5 mg/kg, Atracurium 0.1 mg/kg, 50% N2O + 50% O2 and Isoflurane %1) after endotracheal intubation. Preoperative and postoperative appearance of oropharynx are shown in Figure 1.
3.2. Arrhythmia Data
The rhythm Holter follow-ups of the patients in the 1st month before UPPP (pre-UPPP) and after UPPP (post-UPPP) in the 6th month were performed by a cardiology center and cardiologists. All patients were followed up with Holter electrocardiography (ELA Spiderview Digital Recorder) during sleep. The recordings were transferred to the computer and evaluated with the Holter program, then visually examined and the parasitic regions were excluded. Sinus bradycardia (SB), atrioventricular block (AVB), atrial premature beat (APB), supraventricular tachycardia (SVT), and ventricular extrasystole (VES) parameters were recorded for arrhythmia.
3.3. Statistical Analyses
Study analysis was carried out with details in SPSS V. 20.00 for Windows package program. The data were recorded mostly as mean ± standard deviation (SD). In comparison to independent group differences, the student’s t-test was used when parametric test assumptions were provided, and the Mann Whitney U test was used when not provided.
3.4. Ethical Permission
This study was approved by the Istanbul Yeni Yüzyil University, Non-Interventional Clinical Research Ethics Board (2019/9-02/09/2019). The study was explained in detail to the patients who were included and written informed consent was signed by patients and were obtained from all cases, and the study results were used for scientific aims. The principles of the Declaration of Helsinki and Guidelines for Good Clinical Practices were followed during the study.
4. Results
4.1. Demographic Data
Among patients, 33 (87%) were male and 5 (13%) were female, between 48 and 59 years (the mean age was 54.8 year). All of the patients had moderate OSAS. Endoscopic and imaging examinations have proved that the grade of obstruction was retropalatal in all cases based on the Fujita classification (26).
4.2. Comparison of Arrhythmias
Selected arrhythmia frequency measured before and after UPPP are shown in Table 1. According to these results, statistically significant differences were observed in the changes in SB, AVB, APB, SVT and VE arrhythmia frequency in pre-UPPP and post-UPPP periods (P < 0.001).
| Pre-UPPP | Post-UPPP | Z | P Value | |
|---|---|---|---|---|
| Pulse | 74.42 ± 3.70 | 74.27 ± 3.88 | -0.090 | 0.929 |
| Sinus bradycardia | 4.93 ± 1.43 | 2.81 ± 1.04 | -4.990 | < 0.001 |
| Atrioventricular block | 1.72 ± 1.03 | 0.93 ± 0.74 | -4.435 | < 0.001 |
| Atrial premature beat | 5.51 ± 1.46 | 2.72 ± 1.03 | -5.073 | < 0.001 |
| Supraventricular tachycardia | 3.63 ± 0.99 | 2.33 ± 0.69 | -5.035 | < 0.001 |
| Ventricular extrasystole | 3.57 ± 0.90 | 1.54 ± 0.71 | -5.088 | < 0.001 |
Change in Arrhythmia Frequency Before and After Uvulopalatopharyngoplasty (N = 33).
4.3. Correlations Between Arrhythmias
The correlation results of arrhythmia frequency before and after UPPP are shown in Table 2. According to this table, the frequency of an arrhythmia in the pre-UPPP period had a positive correlation with the values of the same arrhythmia in the post-UPPP period.
| Pre-UPPP | Post-UPPP | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pulse | SB | AVB | APB | SVT | VES | Pulse | SB | AVB | APB | SVT | VES | |||
| Pre-UPPP | Pulse | r | 1 | |||||||||||
| p | ||||||||||||||
| SB | r | 0.316 | 1 | |||||||||||
| p | 0.073 | |||||||||||||
| AVB | r | -0.010 | 0.093 | 1 | ||||||||||
| p | 0.958 | 0.605 | ||||||||||||
| APB | r | 0.005 | -0.029 | -0.419a | 1 | |||||||||
| p | 0.980 | 0.871 | 0.015 | |||||||||||
| SVT | r | 0.009 | -0.082 | -0.462b | 0.822b | 1 | ||||||||
| p | 0.959 | 0.651 | 0.007 | 0.000 | ||||||||||
| VES | r | 0.074 | 0.655b | -0.194 | 0.218 | 0.206 | 1 | |||||||
| p | 0.682 | 0.000 | 0.280 | 0.222 | 0.251 | |||||||||
| Post-UPPP | Pulse | r | 0.005 | -0.165 | 0.019 | -0.020 | 0.091 | -0.019 | 1 | |||||
| p | 0.979 | 0.358 | 0.916 | 0.912 | 0.614 | 0.914 | ||||||||
| SB | r | 0.303 | 0.660b | -0.220 | 0.207 | 0.145 | 0.446b | -0.080 | 1 | |||||
| p | 0.087 | 0.000 | 0.219 | 0.248 | 0.421 | 0.009 | 0.659 | |||||||
| AVB | r | 0.066 | 0.084 | 0.783b | -0.343 | -0.409a | -0.132 | -0.048 | -0.095 | 1 | ||||
| p | 0.715 | 0.643 | 0.000 | 0.051 | 0.018 | 0.464 | 0.791 | 0.600 | ||||||
| APB | r | -0.018 | 0.072 | -0.418a | 0.672b | 0.627b | 0.206 | 0.073 | 0.356a | -0.384a | 1 | |||
| p | 0.922 | 0.689 | 0.015 | 0.000 | 0.000 | 0.250 | 0.685 | 0.042 | 0.027 | |||||
| SVT | r | 0.028 | -0.168 | -0.174 | 0.629b | 0.817b | 0.083 | 0.116 | 0.043 | -0.201 | 0.521b | 1 | ||
| p | 0.875 | 0.351 | 0.333 | 0.000 | 0.000 | 0.645 | 0.519 | 0.811 | 0.261 | 0.002 | ||||
| VES | r | -0.055 | 0.340 | -0.300 | 0.202 | 0.289 | 0.567b | -0.056 | 0.264 | -0.406a | 0.208 | 0.127 | 1 | |
| p | 0.761 | 0.053 | 0.090 | 0.259 | 0.102 | 0.001 | 0.759 | 0.138 | 0.019 | 0.246 | 0.481 | |||
Correlation of Arrhythmia Frequency Before and After Uvulopalatopharyngoplasty
In addition, it was determined that pulse values did not correlate with any other measurements in both pre-UPPP and post-UPP periods. SB value in the pre-UPPP period: There was a positive and high correlation with pre-UPP VE and post-UPP SB values (r values were 0.655 and 0.660, respectively). AVB value in the pre-UPPP period: There were a negative and moderate correlation with APB and SVT values in the pre-UPPP period (r values were -0.419 and -0.462, respectively), a positive and strong correlation with post-UPP AVB values (r = 0.783), and a negative and moderate relationship with post-UPPP APB values (r = 0.418). APB in the pre-UPPP period: There were a positive and very high correlation with pre-UPPP ST values (r = 0.822), and positive and high correlations with the APB and ST values in the post-UPPP period (r values were 0.672 and 0.629, respectively). SVT in the pre-UPPP period: There were a negative and moderate correlation with post-UPP AVB (r = -0.409), a positive and high-grade correlation with post-UPPP APB (r = 0.627), and a positive and very-high grade relationship with post-UPPP ST (r = 0.817). VES in the pre-UPPP period: There was a positive and moderate correlation with SB and VES n the post-UPPP period (r = 0.446 and r = 0.567, respectively). SB in the post-UPPP period: there was a positive and weak correlation with post-UPPP APB values (r = 0.356). AVB in the post-UPPP period: there were a negative and weak relationship with post-UPP APB (r = -0.384) and a negative and moderate relationship with post-UPP VES values (r = -0.406). APB in the post-UPPP period: there was a positive and moderate correlation with post-UPPP SVT frequency (r = 0.521).
5. Discussion
In this study, we investigated whether arrhythmia changes after UPPP in patients (n = 38) with moderate OSAS who have persisting cardiac arrhythmias despite the use of antiarrhythmic drugs and who cannot tolerate CPAP. For this purpose, we evaluated some of the arrhythmia types (SB, AVB, APB, SVT, and VE) in the 1st month before UPPP and in the 6th month after UPPP and recorded their correlations. In this existing study, the implied age of the patients turned into 54.8 years, of which 87% were male. According to our results, statistically significant decreases were observed in all arrhythmia values after UPPP period (P < 0.001). In addition, positive and very high correlations were found between APB and SVT values in the pre-UPPP period (P < 0.001). There were negative or positive correlations between most of the other parameters except arterial pulse.
By affecting the somatic and autonomic nervous system, OSAS causes adverse effects in the functions of respiratory cardiovascular, gastrointestinal, endocrine, renal and sexual systems. Its most vital aspect outcomes occur inside the cardiovascular system. In OSAS, the most vital reasons of mortality and morbidity are headaches of cardiovascular system (11). Lee et al. evaluated the effects of UPPP on heart disease in a study of 192,316 patients with OSAS (South Korea). They reported that congestive heart failure, atrial fibrillation, and myocardial infarction (in females) were significantly increased in OSAS patients. In addition, UPPP significantly reduced the incidence of congestive heart failure, atrial fibrillation, and myocardial infarction (in some models) in patients with OSAS (14).
In the literature, OSAS has been frequently linked with atrial fibrillation. Beyond atrial fibrillation, OSAS has also been associated with other cardiac rhythm disorders, including sudden cardiac death. Simantirakis and colleagues declared a 22% prevalence of prolonged pauses and bradycardia in moderate‐to‐severe OSAS patients (27). Patients are assumed to have the “tachy‐brady syndrome” when such bradyarrhythmias change with atrial fibrillation or other forms of SVT. The ventricular arrhythmias were also reported to be higher among patients with OSAS. Mehra and colleagues showed a significantly higher prevalence of non-sustained ventricular tachycardia (5.3% versus 1.2%, P = 0.004) among OSAS patients compared with controls (28). Similarly, severe OSAS was associated with a significantly higher overall risk of complex ventricular ectopy, defined as non-sustained ventricular tachycardia, bigeminy, trigeminy, or quadrigeminy (25% versus 14.5%, P = 0.002) (29).
The attacks of hypoxia, increased negative-intrathoracic pressure and high sympathetic activity are the most common causes of high cardiovascular diseases in patients with OSAS. The intermittent hypoxia due to apnea attacks leads to a decrease in the velocity of cardiac conduction, atrial fibrosis, oxidative stress, and systemic inflammation. In addition, hypoxia induces carotid chemoreceptors and leads to catecholamine discharge and vasoconstriction. Pulmonary vasoconstriction and pulmonary hypertension may also occur due to alveolar hypoxia. Recurrent hypoxia attacks during sleep cause acute elevations in pulmonary artery pressure (30, 31).
Increased negative intrathoracic pressure secondary to increased inspiratory effort due to airway obstruction increases the afterload of heart and leads to left ventricle and left atrial hypertrophy, hypertension, and cardiac arrhythmias (8, 32). Negative intrathoracic pressure fluctuations throughout obstructive attacks increase catecholamine release and so on increase atrial and intrathoracic aortic wall stress, as a result it also increases the likelihood of nocturnal atrial arrhythmias and thoracic aortic dissection (33).
High sympathetic activity due to awakening and fear of death at the end of apnea reduces heart rate variability and increases blood pressure. The three main points mentioned above, and other sequelae probably contribute to an increased risk of cardiac arrhythmias, congestive heart failure and myocardial infarction in OSAS patients (12, 14, 34). Another factor causing catecholamine discharges is the “stimulation” response that pauses the apnea. Another study reported that catecholamine levels in the blood and urine of patients with OSAS decreased slowly after CPAP treatment (35).
In our study, the types of correlations (positive and negative) between arrhythmias in the 1st month before and the 6th month after UPPP are shown in Table 3. According to this table, the frequency of an arrhythmia in the pre-UPPP period has a positive correlation with the values of the same arrhythmia in the post-UPPP period. The values of pre-UPPP APB were positively correlated with three parameters (pre-UPPP SVT, post-UPPP APB, and post-UPPP SVT). It is known that supraventricular arrhythmias have correlations among themselves and ventricular arrhythmias have correlations among themselves too (9, 14, 29). Positive correlations between APB and SVT in pre- and post- UPPP periods can also be explained in this relationship. In addition, there were negative correlations between pre-UPPP AVB and three parameters (pre-UPPP APB, pre-UPPP SVT, and post-UPPP APB).
| Correlations | ||
|---|---|---|
| Positive Correlations | ||
| Pre-UPPP SB | And | pre-UPPP VES ve post-UPPP SB |
| Pre-UPPP AVB | post-UPPP AVB | |
| Pre-UPPP APB | pre-UPPP SVT, post-UPPP APB ve post-UPPP SVT | |
| Pre-UPPP SVT | post-UPPP APB ve post-UPPP SVT | |
| Pre-UPPP VES | post-UPPP SB ve post-UPPP VES | |
| Post-UPPP SB | post-UPPP APB | |
| Negative Correlations | ||
| Pre-UPPP AVB | And | pre-UPPP APB, pre-UPPP SVT ve post-UPPP APB |
| Pre-UPPP SVT | post-UPPP AVB | |
| Pre-UPPP AVB | post-UPPP APB ve post-UPPP VES | |
The Correlation Types of Arrhythmias Before and After Uvulopalatopharyngoplasty
SB value in the pre-UPPP period: there was a positive and high correlation with pre-UPPP VE and post-UPP SB values (r values were 0.655 and 0.660, respectively). AVB value in the pre-UPPP period: there were a negative and moderate correlation with APB and SVT values in the pre-UPPP period (r values were -0.419 and -0.462, respectively), a positive and strong correlation with post-UPP AVB values (r = 0.783), and a negative and moderate relationship with post-UPP APB values (r = 0.418). APB in the pre-UPPP period: there were positive and very high correlation with pre-UPPP ST values (r = 0.822), and positive and high correlations with the APB and ST values in the post-UPP period (r values were 0.672 and 0.629, respectively). SVT in the pre-UPPP period: there were a negative and moderate correlation with post-UPP AVB (r = -0.409), a positive and high-grade correlation with post-UPP APB (r = 0.627), and a positive and very-high grade relationship with post-UPPP ST (r = 0.817). VES in the pre-UPPP period: there were a positive and moderate correlation with SB and VES n the post-UPP period (r = 0.446 and r = 0.567, respectively). SB in the post-UPPP period: there was a positive and weak correlation with post-UPPP APB values (r = 0.356). AVB in the post-UPPP period: there were a negative and weak relationship with post-UPP APB (r = -0.384) and a negative and moderate relationship with post-UPP VES values (r = -0.406). APB in the post-UPPP period: there was a positive and moderate correlation with post-UPP SVT values (r = 0.521).
5.1. Limitations of the Study
However, there are several obstacles to this work that should be acknowledged. First of all, for easier design, faster completion and more understandable interpretation of our study, we did not evaluate OSAS patients' concomitant diseases such as hypertension, DM, BMI, and dyslipidemia and we did not record their changes after UPPP. However, the relationships between these diseases and both OSAS and arrhythmias should be considered, and these parameters should be considered in regression and correlation analyses.
Secondly, we assessed the effect of UPPP on arrhythmias in just as short as six months, because this time was enough for us to confirm our hypothesis. Also, researches on the long-term effects of UPPP have been harsh in terms of compliance and cost of patients. For this reason and for the reasons stated in the first limitation, we did not evaluate long-term arrhythmia outcomes such as 1, 2 and 3 years after UPPP. However, it is known that the recovery periods of some chronic diseases may take a long time. Therefore, particularly in regression and correlation analyses, time-dependent changes of some positive and negative relationships should be considered for the researchers.
In addition, the lack of assessment of the AHI after UPPP and the evaluation of arrhythmias only in moderate OSAS patients are other limitations of our study.
5.2. Conclusions
In this study, new information about the effects of UPPP which is one of OSAS treatment methods on cardiac arrhythmias is presented. According to our results, UPPP reduces the rate of SB, AVB, APB, SVT and VES arrhythmias in OSAS patients.