1. Background
Sinus of Valsalva aneurysm (SOVA) is an uncommon cardiac anomaly. It can be congenital or acquired with a prevalence about 0.09% of population (1). It was first reported in the literature in the beginning of nineteenth century (2). It can rupture into the heart's cavities, most frequently extending into the right ventricular outflow tract's upper portion (3). Congenital SOV aneurysms are usually observed in conjunction with other cardiac anomalies such as ventricular septal defect (VSD) or aortic valve dysfunction (4). The transesophageal echocardiography (TEE) is the diagnostic method of choice, in addition to cardiac magnetic resonance and computed tomography (CT) which can be helpful (5). Indications for surgery in non-ruptured SOV aneurysms are symptomatic patients or rapid enlargement. Additionally, untreated ruptured sinus of Valsalva aneurysm (RSOV) almost always results in worsening of cardiac function. Intervention is therefore required, either by surgery or the use of endovascular closure devices that have successful outcomes (6).
2. Objectives
The purpose of the current study was to review the outcomes of 42 patients who underwent surgery to treat sinus Valsalva aneurysms at the university of cardiovascular and research center of Iran during a 12-year period.
3. Methods
Approval and Institutional Ethics for this review were obtained from Iran University of medical sciences, we retrospectively reviewed database for all patients who underwent surgical repair of sinus of Valsalva aneurysm between November, 2005 and august 2018 and all data pre, intra and postoperative have been extracted. Preoperative diagnostic study included: history and physical examination with chest X-ray, ECG and echocardiography. Prior to surgery, all patients underwent a conventional clinical examination, echocardiography, and multislice CT angio. Cardiac catheterization and angiography were performed in 27 (64%) of patients to study the origin and termination of the fistula and presence of associated coronary artery disease or anomalies, particularly VSD, AR and pulmonary stenosis. Symptomatic patients with non-ruptured SOV aneurysm and patients with RSOV were operated. Surgical approaches varied depending on rupture or non-ruptured aneurysm, number of SOV involved and co-existing lesion (VSD and other congenital disease, aortic insufficiency and aortic root dilation). The right ventricle and right atrium of the heart were used as the surgical approach, along with the aorta or a combination of both. The ascending aorta was always used to access non-ruptured SOV. Operative technique of surgical repair:
(1) At the base of the fistula, the aperture of the ruptured Valsalva sinus aneurysm was closed off with one or two distinct types of patches.
(2) Non-ruptured sinuses of Valsalva were repaired by excision or by exclusion of aneurysm.
(3) In all patients with more than moderate aortic valve regurgitation (AR) the aortic valve was repaired or replaced according the surgical findings and surgeons' preference.
(4) Concomitant VSDs were closed with a-single-patch technique or by direct closure.
(5) All associated pathologies (atrial septal defect, patent foramen oval, mitral regurgitation and others) were repaired concomitantly.
Post operatively, the mean follow up period was 31 ± 33 months. Data are presented as mean value ± standard deviation, median value, number of patients or as a percentage related to entire patients. For statistical analysis we use following software: Microsoft Excel 2010 and SPSS version 22 (IBM Corp. IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp.).
4. Results
Most patients were men (34 vs 8), the mean age was 31.3 ± 16.09 years (Table 1). The most common clinical symptoms were: dyspnea on exertion (80%), chest pain (11%), palpitations (4.7%), fever (2.7%), and growth retardation (2.7%).
| Variables | Values a |
|---|---|
| Sex | |
| Male | 34 (80.95) |
| Female | 9 (19.05) |
| Average age of men | 31.3 ± 13.2 |
| Average age of women | 33.1 ± 34.92 |
| Average age of total patients | 33.3 ± 16.02 |
a Values are expressed as No. (%) or Mean ± SD.
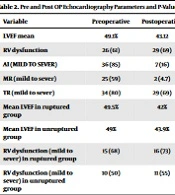
Preoperative echocardiographic data are shown in Table 2. Aortic valve regurgitation as the main concomitant abnormality was severe in 14 (33.3 %), moderate in 9 (21.4%) and mild in 13 (30.9%). Intervention on aortic valve were done in 28 case (66.6%), including 20 (71.42%) AVR and 8 (28.5%) AV repairs P = 0.03. VSD repair was performed in 23 patients out of 25 associated cases (59.5%), which were primarily of the perimembranous variety (54.76%).
| Variables | Preoperative | Postoperative | P-Value |
|---|---|---|---|
| LVEF mean | 49.1% | 43.12 | 0.009 |
| RV dysfunction | 26 (61) | 29 (69) | 0.34 |
| AI (mild to sever) | 36 (85) | 7 (16) | 0.048 |
| MR (mild to sever) | 25 (59) | 2 (4.7) | 0.56 |
| TR (mild to sever) | 34 (80) | 29 (69) | 0.29 |
| Mean LVEF in ruptured group | 49.5% | 42% | 0.26 |
| Mean LVEF in unruptured group | 49% | 43.9% | 0.08 |
| RV dysfunction (mild to sever) in ruptured group | 15 (68) | 16 (73) | 0.8 |
| RV dysfunction (mild to sever) in unruptured group | 10 (50) | 11 (55) | 0.13 |
a Values are expressed as No. (%) unless otherwise indicated.
There was no significant mitral, tricuspid or pulmonary valvular involvement.
Most patients were in normal sinus rhythm preoperatively (95.2 %). One patient had LBB and one other had CHB. However 14.3% of patients had LBBB postoperatively. CHB occurred in two patients (7.3%) who needed permanent pace maker post operatively and AF in one 2.79%. These alterations were significantly different in the ruptured group (P = 0.001).
Functional status of all patients, were assessed by New York Heart Association (NYHA) functional classification as you see in (Table 3), and were improved postoperatively and 82% were in FC I postoperatively.
| NYHA Class | Pre and Postoperative Patient NYHA Functional Class | |
|---|---|---|
| Preoperative | Post Operative | |
| NYHA I | 12 (28.5) | 34 (82) |
| NYHA II | 23 (54.7) | 7 (18) |
| NYHA III | 6 (14.2) | 0 (0) |
| NYHA IV | 1 (2.3) | 0 (0) |
a Values are expressed as No. (%)
The involved aneurysmal sinus (Table 4) was mostly observed in right coronary sinus in (76.1%), followed by noncoronary sinus in (16.6%) and left coronary sinus in (7.1%). Ruptured aneurysm happened in 23 patients (54.7%). The ruptures were in:
(1) Right ventricle in 18 cases (78.2%) and all of these fistulas originate from right coronary SOV;
(2) Left ventricle in 5 patients (21.7 %) where the fistula originated from right, left and non-coronary SOV in 3, 1 and 1 case respectively;
(3) No rupture to other cavities or pericardium in our group (Table 5).
| Involved Sinus | No. (%) |
|---|---|
| Right coronary sinus | 32 (76.1) |
| Non coronary sinus | 7 (16.7) |
| Left coronary sinus | 3 (7.1) |
| Variables | No. (%) |
|---|---|
| Ruptured aneurysm | 23 (54.7) |
| Rupture into the right ventricle | 18 (78.2) |
| Rupture into the left ventricle | 5 (21.8) |
| Rupture into the other cavities or pericardium | 0 (0) |
| No Rupture | 19 (55.3) |
Through the use of multivariate analysis, we discovered that female gender and aortic valve regurgitation were predictors of rupture SOV aneurysm; however, we did not discover a relationship between rupture and HTN, DM, PAP, TR, CAD, aortic valve annulus, or aortic cross clamp time.
Intra operative TEE was performed in 37 (88%) of cases and revealed that mean intra-operative ejection fraction was 49 ± 9.6. There were no residual VSD or AI.
Post operatively 2 patients had low cardiac output syndrome LCOS leading to death. In the survived patients 3 tamponade (with need to re-exploration), 1 gastrointestinal bleeding, 2 cases of pulmonary emboli, 2 patient with stroke and 2 cases with acute renal dysfunction (CL cr ≥ 1.5 without need to dialysis) were found. All surviving patients were followed for a mean of 31 months. When comparing the left ventricle EF in the ruptured and non-ruptured groups during the follow-up period, the non-ruptured group showed a significant post-operative improvement (P = 0.009).
Right ventricle systolic function had been improved significantly post operatively which may be related to closing of the fistulas.
4.1. Surgical Notes
In all patients the senior consultant (one team) make the final decision about how and what to do. In general: Ascending aorta cannulation was done in all cases, tow stage venous canula was used in NRSOVA and Bi caval cannulation (SVC AND IVC) in patients with RSOVA or in concomitant Tricuspid or mitral valve procedure, cardioplegia was given in aortic root in patients without AI and directly in coronary ostium in AI cases and RSOVA cases. In patients with more than moderate aortic valve regurgitation (AR) the aortic valve was repaired (the technique differ up to the mechanism sub-commissural triangioloplasty, leaflet repair, valve sparing repair... etc.) or replaced according the surgical findings. Surgical technique for repairing sinus of Valsalva was done by patch closure. In 26 (61.9%) of patients, the most used patch was gortex patch in 14 cases (53.8%) but in RSVO the base of fistulas was closed by gortex patch in (21.7%) and pericardial patch in (21.7%) of patients. VSD repair was done in 25 patents by using gortex patch in 10 (40%), direct closure in 8 (32%) and using pericardial patch in 7 (28%). Cold crystalloid cardioplegic solution was used antegrade when the aortic valve was competent, but inside coronary artery ostium or by retrograde methods in AR and ruptured SOVA cases.
Mean cardiopulmonary bypass time was 140 ± 67 minute. Mean aortic cross clamp time was 68.9 ± 53.55. Weaning from CPB was done with the need to low dose inotrope in 9 patients (21.4). All patients were weaned from ventilator in the ICU units after a mean time of 18 ± 10 hours.
5. Discussion
SOV aneurysms are rare abnormalities with male preponderance (4:1) and highest incidence in Asian populations (7). Given the central position of aorta in the base of the heart, make it possible that SOVA could rupture any heart chamber, but mostly to the right ventricle or right atrium (2, 8). Aortic Valve incompetence is common in patients with SVA, which occurs in 30 - 50% of patients and it can influence on disease progression (9). For achieving good long-term results in patients with SVA it is important to correct concomitant AI which usually arises from the effect of concomitant VSD or from other aortic valve abnormality, including a bicuspid valve (10). In our series, there was no bicuspid valve patients and we found that aortic insufficiency was more severe when the Valsalva aneurysm was ruptured into heart cavities (P = 0.05). In this condition asymmetrical deformity of aortic root and rupture of SVA and substantial runoff blood flow in the supra-annular region both can cause severe aortic regurgitation in the absence of a VSD or a notable structural defect in the aortic valve (11). However the cause of AI, when coexist with VSD, is the prolapse of aortic cusp into VSD (12). Some authors believe that in the presence of VSD and AI, because of prolonged hemodynamic trauma, aortic leaflet become fibrotic and deformed and this problem mandates aortic valve replacement instead of repair (13), although we couldn’t find this relationship in our study (P = 0.89). As these surgeries were done by one surgical team, the surgical decision was made in the time of surgery by evaluation of the leaflet mobility and calcification, essentially by the same attendant. Thus, in most patients with unruptured sinus of Valsalva, AVR were done less. There was significant correlation between aortic valve surgery and RSOV (P = 0.03) with more a significant value P = 0.001 with AVR which alludes to the fact that turbulent flow can damage the aortic valve as well, making it unrepairable.
Active surgical repair of an un-ruptured SVA can be achieved with satisfactory results in patients combined with other cardiovascular lesions (2).
Patch closure was more used than direct suturing of VSD, although there is no correlation between the use of patch and mortality or morbidity of patients (P-value > 0.05). Various surgical techniques can be utilized to manage SOVA and RSOV aneurysms, with or without coexisting abnormality or concomitant AI and VSD, anyway the optimal intervention for repair is still not determined and need more discussion (14). In this study patch repair of fistula was performed. Many surgeons believe that direct suture closure of the fistula's ostium is ineffective because it increases the risk of recurring fistulas if the sutures anchor at a thin, deteriorating wall. Most surgeons agree that employing a patch is required, particularly in RSVA because healthy tissue must extend 4-5 mm beyond the apparent rupture hole in order to be properly sutured (15). In 2017, Gupta demonstrated that applying a patch to close a fistula was associated with minimal operating risk, a lower incidence of aortic valve deformation, and high long-term survival (16); however some researchers could not find any relationship between the use of patch in SOV and patients’ outcomes (17). However selecting the repair technique is mostly subjective and there is no definite guideline, but in each type of repair function and geometry of aorta must be considered and surgeon must avoid aortic valve distortion (18). In follow up period there was no reoperation for failed aortic valve or residual VSD. Most patients were in New York Heart Association (NYHA) CLASS I and II. Most postoperative rhythms were sinus rhythm, but there were some abnormal rhythms (CHB, LBBB, AF) that were not present on admission of patient before surgery. Various studies have found that heart block can occurs in 2 to 3% of patient post operatively, occasionally late post operatively may be because of proximity of his bundle and its branch to the area of repair (19). Despite that, some authors claim that aneurysmal pressure and inflammation lead to block (20, 21), but in our series there was no correlation between RSOV and probability of heart block (P = 0.22). In reality, most patients who underwent surgery at University Hospital had right ventricle outlet tract (RVOT) rupture of the Valsalva sinus, and the most frequently involved leaflet was the right coronary cusp. Therefore, given that most fistula routes appear to be between the right and left coronary sinus, which are located far from the septum membranous, injury to the conductive tissue was unlikely. Additionally, patch closure approach can reduce pressure on conductive tissue and consequently postoperative block when used to close an aortic side rupture in the majority of patients and all patients, respectively. This issue has to be further assessed. The operation mortality in our study was 2.3 percent, and when we take into account the post-operative data provided above, we draw the conclusion that treating SOV aneurysms was successfully completed.
5.1. Conclusions
Surgical repair of an SVA is associated with low operative risk, death and reoperation. Results from one surgical team, especially when the same specialist is making decisions, are more trustworthy. Excellent long-term outcomes are obtained, and rupture of the SOV is linked to an increase in AI and, ultimately, to aortic valve replacement surgery, primarily AV. Patch method for fistula closure does not worsen post-operative arrhythmia.