1. Background
Swallowing is a sensorimotor process that is performed in both autonomic and voluntary manners in a complex neural network (1). Swallowing disorder or dysphagia is one of the most common motor symptoms in Parkinson's disease (PD) (2). The prevalence of this disorder in people with PD has been estimated between 12% and 90%, depending on the evaluation approach (3). Medical complications from dysphagia include aspiration of food and liquids into the lungs, malnutrition, and pneumonia, which can be life-threatening (2, 4). Swallowing disorders can be modified and adapted (4). Swallowing rehabilitation techniques are being developed to restore and improve the swallowing function in patients with PD (5-7). In examining the treatment of dysphagia in patients with PD, it was found that although traditional treatments (such as medication and deep brain stimulation) have been useful for improving motor aspects in PD, they are largely ineffective in helping speech and swallowing in these patients (8, 9). Also, studies have shown that behavioral treatments or swallowing training can produce positive results in swallowing outcomes (4, 6, 10, 11).
Intensive speech therapy is non-instrumental behavioral therapy to reduce swallowing problems in patients with PD, which has shown positive results (4, 6, 11). Lee Silverman’s voice therapy (LSVT LOUD®) is also an intensive and high-effort voice therapy that increases the ability and capacity of the respiratory and larynx muscles, thereby improving the loudness of the voice in patients with dysarthria (12). Although LSVT does not specifically target swallowing function, its exercises have been shown to engage many muscles, such as the submental muscles (probably due to similarities in peripheral and central neural control elements), and create positive results for swallowing (6, 13). Studies also have demonstrated that the effects of improvement in respiration and vocalization are related to increased voice strength, improved voice quality, and increased maximum phonation time, which improves swallowing performance in these patients (4). These studies have concluded that LSVT treatment improves swallowing function in patients with PD (4). Implementation of LSVT by therapists requires specific training and certification (14); however, the number of LSVT-certified speech and language pathologists is small (15). Limited access to speech and language pathologists trained in LSVT, distance, cost, motor/cognitive problems of PD, and lack of caregiver availability are limitations of this intensive treatment protocol (15).
Music therapy is a new method for the treatment of speech and swallowing disorders in persons with PD (16). The theoretical basis of investigating the effectiveness of music therapy (singing) in the treatment of disorders related to dysarthria has been explained with biomedical theories and principles of neuroplasticity. In this regard, it has been suggested that neural processes may be activated by musical stimuli and used to influence non-musical behavior (17). Studies have shown that music therapy techniques, such as singing and vocal training, are effective in managing functional communication in patients with PD (18). Studies on music therapy as a swallowing rehabilitation method in persons with PD are limited (10). The only study conducted in this field showed that music therapy in combination with voice therapy could play an effective role in improving the voice and swallowing problems in people with PD (10). This study investigated an interdisciplinary approach to voice and swallowing therapy in people with PD that combined both music and voice therapy paradigms. Although this study had a relatively small sample size, its results suggest that singing or music therapy may help improve dysphagia, and it is worth further investigation. Therefore, more studies are needed to evaluate the effectiveness of this method.
Receiving continuous health care for people with PD is very important to reduce the symptoms of this disease and increase the quality of life. Due to factors such as mobility problems, distance barriers, social distancing during outbreaks of pandemics (such as COVID-19), financial issues, and lack of time, a large number of patients with PD have limited access to face-to-face interventions (19, 20). In this situation, telerehabilitation strategies can be adopted as alternative methods of providing health care services. Several studies have been conducted on the effectiveness of telerehabilitation to provide speech and language pathology services in the treatment of a wide range of communication disorders, including PD (21). Therefore, telerehabilitation can be a suitable option for receiving speech therapy services in patients with PD (22).
So far, no behavioral treatment has been conclusively proven to improve swallowing function in PD patients (23). Studies that have examined non-instrumental behavioral therapy for dysphagia in patients with PD are different in terms of treatment methods and outcome measures, and most of them are non-randomized clinical trials with a small sample size (10). Due to the lack of sufficient evidence, more studies are needed to develop and evaluate other evidence-based treatment programs, as well as to demonstrate the effectiveness of any type of speech-behavioral therapy in improving swallowing function in PD (6). No studies have been conducted on the effect of combined speech therapy and music therapy provided through telerehabilitation on swallowing outcomes in patients with PD.
2. Objectives
This randomized clinical trial study was conducted to evaluate a telerehabilitation intervention for the treatment of dysphagia in these patients. This study investigated and compared possible changes in swallowing function in persons with PD after conventional speech therapy, music therapy, and combination (speech and music) therapy. Also, to investigate the long-term outcomes, the results were evaluated 3 months after the intervention.
3. Methods
3.1. Trial Design
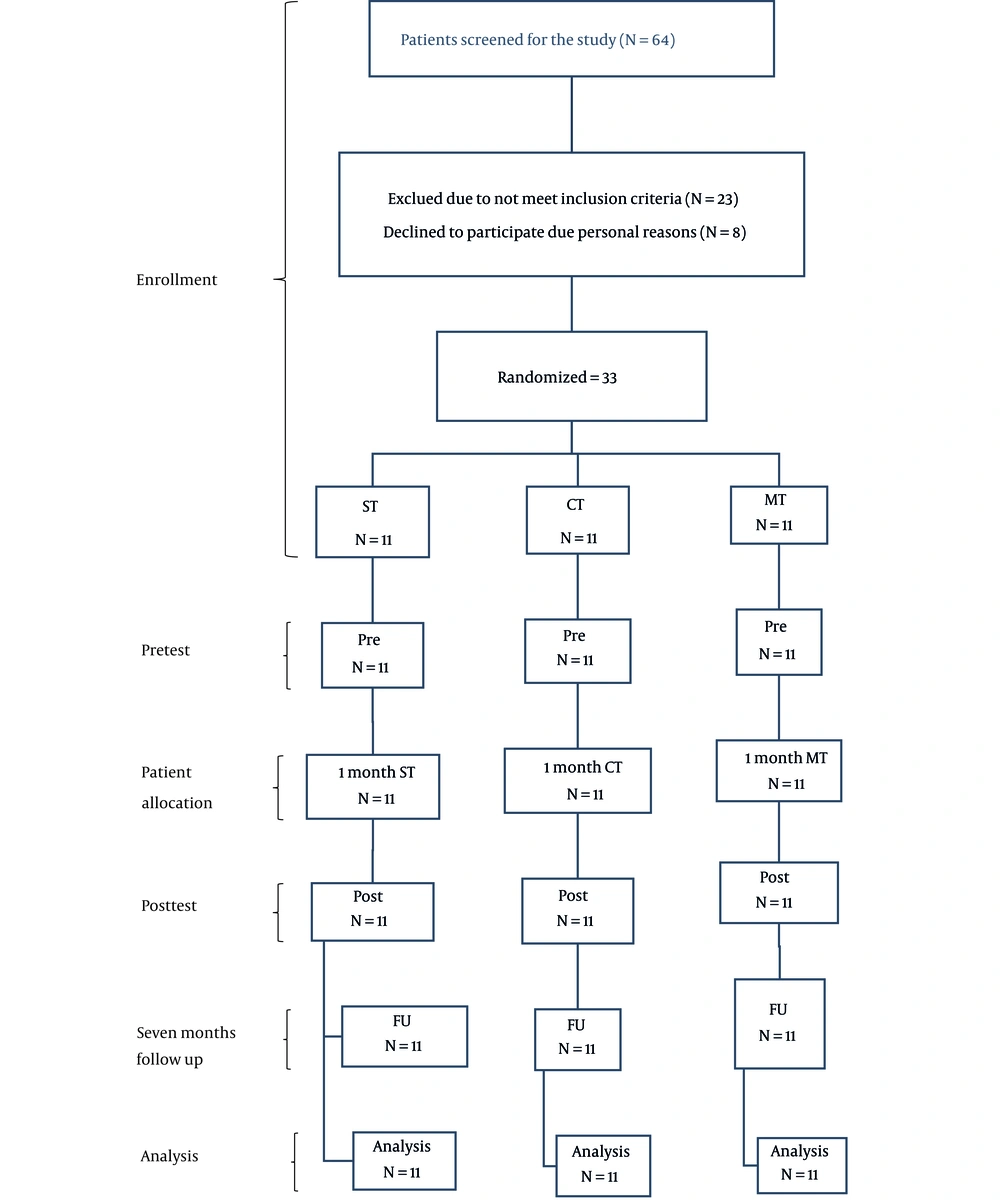
This study was a randomized clinical trial with an available sampling method and 3 parallel treatment arms, which was conducted according to the guidelines of consolidated standards of reporting trials (CONSORT) 2010 (Figure 1). This study aimed to determine the effect of conventional speech therapy combined with music therapy techniques compared to conventional speech therapy and music therapy alone on the dysphagia handicap index (DHI; functional, physical, and emotional sub-tests, as well as total score) and Swallowing Disturbance Questionnaire (SDQ) scores in individuals with PD. Assessments were performed face to face with all patients individually by a blind speech and language pathologist. Evaluations were done 1 week before the first intervention session (pretest), 1 week after the last intervention session (posttest), and 3 months after the last evaluation (follow-up). The collection methods were the same for all 3 groups. This study was approved by the Ethics Committee of Iran University of Medical Sciences (code: IR.IUMS.REC.1400.044). This trial was registered at clinicaltrials.gov before recruiting the participants (code: NCT04966689; clinicaltrials.gov/ct2/show/NCT04966689). All participants completed a research written informed consent form.
3.2. Participants
Thirty-three subjects with idiopathic PD (IPD; mean ± SD age: 58.88 ± 8.12 years; 8 women and 25 men) and complaints of swallowing problems were recruited from speech therapy clinics, outpatient clinics, neurology offices, and hospitals in Tehran, Iran. Demographic information and initial screening data were collected by a speech and language pathologist. Inclusion criteria were patients with IPD diagnosed by a neurologist using the UK Parkinson’s Disease Community Brain Bank clinical diagnostic criteria, being at stages 1 to 2 (mild) based on the Hoehn and Yahr (H&Y) scale, having normal or modified hearing, and visual range, having at least a high school education level, complaining of swallowing problems, and being familiar with information technology applications such as WhatsApp. Exclusion criteria included atypical PD or other neurological disorders, history of stroke, head injury/brain surgery, receiving neurosurgical treatment such as subthalamic deep brain stimulation (STN-DBS), and history of laryngeal surgery or injury. The medication used by the patients (levodopa) and its dosage remained unchanged during the study process. The assessment and treatment sessions were performed in the on-state of the medication cycle, which is usually 1 hour after medication administration and has the peak effect of the medication cycle.
3.3. Intervention
Each patient received 12 individual therapy sessions over 4 weeks (3 sessions per week). Also, home exercises were performed by patients on non-treatment days. The duration of each session depended on the patient’s ability to complete the treatment protocols, but an average of 1 hour was considered (more in the combination therapy group). Sessions were performed at patients’ homes online by videoconferencing using WhatsApp. The treatment plans of all 3 groups were carried out by a speech and language pathologist and in combination therapy and with the cooperation of a music and singing specialist in the music therapy groups. In patients unable to complete the program without caregiver support, a caregiver was asked to help them during the sessions. Before starting the treatment sessions, all participants and their caregivers were trained to set up a videoconference system on their smartphones. To standardize the visual and audio quality of the information exchange, the cell phones were placed on a holder 30 cm away from the users (22). The volume of smartphones was set to its maximum value. Videoconferencing was set up by the mobile internet using Verizon’s 4G LTE network (15).
Conventional speech therapy included common and routine speech exercises adapted from LSVT therapy and vocal exercises (12). Treatment exercises were applied depending on individual needs and the patient’s learning approach (24). These treatment exercises were delivered through telerehabilitation and included exercises targeting breathing, phonation, loudness, pitch, and intelligibility. Exercises were repetitive and focused on the maximum vocal effort to increase maximum loudness, pitch range, and intelligibility. Each treatment session in this group consisted of repetitions of vowel (“ah”) in a sustained form, exercises on high/low-pitch range, and repetitions of some frequently used phrases with good quality and the highest level of loudness. Exercises were extended hierarchically and ranged from simple to complex in different linguistic levels (words < phrases < sentences < reading < conversation) (12).
The music exercises used for the music therapy group were designed based on the music therapy protocols of previous studies (25). They included motor, breathing, vocal, and singing exercises. Singing exercises included semi-occluded exercises, pitch glide, laryngeal tilt, Cuperto, vowel alternation, staccato, vocal adduction, Messa di Voce, vocal articulation, vocal agility, chanting exercises, and repertoire. Therapists provided cues related to the music rhythm, such as visual and proprioception cues (i.e., gestures, monitoring, or feeling orofacial muscle tension) to better understanding.
The combination therapy protocol included both conventional speech therapy and music therapy exercises of the other 2 groups. The exercises of this group were the same exercises used in the speech therapy and music therapy groups and simultaneously provided to the patients through telerehabilitation. This intervention was provided by a speech and language pathologist (to perform speech exercises) and a music and singing specialist (to perform singing exercises) through videoconferencing on WhatsApp. In each session, the music therapy protocol was first presented by a singing specialist in collaboration with a trained speech and language pathologist, and then the speech tasks were presented by the speech and language pathologist alone.
3.4. Outcomes
The scores of SDQ and DHI were selected as the outcomes of this study to evaluate swallowing status. DHI is a self-report questionnaire designed to measure the negative impact of dysphagia on the quality of life and can be a useful tool for screening and treatment planning patients with dysphagia (26). DHI consists of 25 items and has 9 questions in the functional subscale, 9 questions in the physical subscale, and 7 questions in the emotional subscale. The patient can give 3 answers for each question (never, sometimes, and always), and each answer has a score ranging from 0 (the least disability) to 100 (the most disability (27). This questionnaire has good internal consistency (the Cronbach α coefficient varies from 0.82 to 0.94) and test-retest reliability (r = 0.89) (28).
SDQ is a self-rated dysphagia screening tool with 15 questions, of which 5 questions are related to the oral phase of swallowing, and 10 questions are related to the pharyngeal phase. Fourteen questions were rated by a 4-point (0 - 3) scale (0 for no disability and 3 for severe disability) and 1 “yes/no” question (yes was scored 2.5, and no was scored 0.5). The total SDQ score ranges from 0.5 to 44.5. SDQ score ≥ 11 indicates a swallowing problem (27). The Cronbach α coefficient for the adapted version of this questionnaire was 0.86, and its sensitivity and specificity in predicting aspiration were 96.7 and 91.2% (29).
3.5. Sample Size
The sample size was determined using the formula n = λ/Δ (30). The value of λ was 9.64, according to the confidence and power level of the test (95% and 80%). The value of Δ was calculated with the formula
according to the results of previous studies (6, 27). As a result, there were 9 samples in each group, but considering the dropout rate of 15%, 11 samples were determined in each group, and the total sample size was 33 people.
3.6. Randomization
Participants were randomly divided into 3 groups of combination therapy (intervention group), speech therapy (comparison group), and music therapy (comparison group) in a ratio of 1:1:1 (Figure 1). A simple random assignment approach was used. To randomize participants, a random allocation list was created using SPSS version 23 (SPSS Inc, Chicago, Ill, USA). Each list was placed in a closed envelope. The patients who were selected based on the inclusion and exclusion criteria took one of the envelopes from the top, and in this way, the treatment group of each patient was determined.
3.7. Blinding
In this study, the assessor and data analyzer were blinded to the treatment allocation during the trial period. Participants knew they were receiving one of 3 possible treatments but did not know the other treatment methods. It was impossible for the therapist to be blinded because the therapist conducted the research.
3.8. Statistical Methods
Statistical analysis was performed using SPSS version 23 (SPSS Inc, Chicago, Ill, USA). Demographic characteristics and study outcomes were evaluated using descriptive statistics, including measures of mean and SD criteria for quantitative variables and frequency and percentage for qualitative variables. The Shapiro-Wilk test was used to investigate the standard normality of the data for all quantitative variables. To investigate the existence of differences between the 3 groups at baseline, an analysis of variance (ANOVA) was completed to compare age, gender, time since diagnosis, mini-mental state exam (MMSE), and disease severity scores between groups. A repeated-measures ANOVA with the between-subjects factor group (conventional speech therapy, music therapy, and combination therapy interventions) and the within-subjects factor time (pretest/posttest/follow-up) was conducted to compare progress across intervention sessions, as well as the variables between groups over time. Wherever time × group interaction results were statistically significant, a post hoc Bonferroni analysis was performed to identify significant differences between the 3 groups. In all analyses, a critical P value of less than 0.05 was considered statistically significant for within and between-group comparisons.
4. Results
Thirty-three patients with IPD (28 men and 5 women) participated in this study. No dropout occurred in the treatment process, and all participants completed all treatment and follow-up sessions. The demographic characteristics of the studied groups are presented in Table 1, including age, sex, disease duration, MMSE, and H&Y scale. There was no significant difference between the groups (P > 0.05) for these demographic characteristics. Also, ANOVA showed that all 3 groups did not differ significantly from each other in SDQ and all sub-tests (functional, physical, and emotional), as well as the total score of the DHI at baseline (P > 0.05; Table 2). A repeated-measures ANOVA, with time points as a within-subjects factor and intervention as a between-subjects factor, was used to assess whether study outcomes significantly changed.
| Characteristics | Total | CT Group | ST Group | MT Group | P Value b |
|---|---|---|---|---|---|
| Age (y) | 58.88 ± 8.12 | 59.18 ± 8.95 | 59.63 ± 8.05 | 57.81 ± 8.01 | 0.869 |
| Gender (M: F) | 25: 8 (84.85: 15.15) | 9: 2 (81.82: 18.18) | 8: 3 (72.73: 27.27) | 8: 3 (72.73: 27.27) | 0.860 |
| Disease duration (y) | 7.15 ± 1.48 | 6.91 ± 1.64 | 7.36 ± 1.43 | 7.18 ± 1.47 | 0.780 |
| MMSE | 26.79 ± 2.34 | 27 ± 2.41 | 26.72 ± 2.28 | 26.63 ± 2.54 | 0.935 |
| Disease severity (H&Y) | 1.85 ± 0.57 | 1.82 ± 0.603 | 1.82 ± 0.603 | 1.91 ± 0.54 | 0.915 |
Abbreviations: SD, modified standard deviation; MMSE, mini-mental state exam; H&Y, Hoehn & Yahr scale; CT, combined therapy; ST, speech therapy; MT, music therapy.
aValues are expressed as Mean ± SD or No. (%).
b P < 0.05, ANOVA test.
| CT Group | ST Group | MT Group | F | P Value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pretest | Posttest | Follow-up | Pretest | Posttest | Follow-up | Pretest | Posttest | Follow-up | Time | Time & Group | Group | Time | Time & Group | Group | |
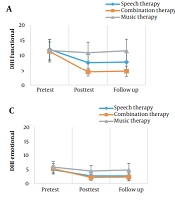
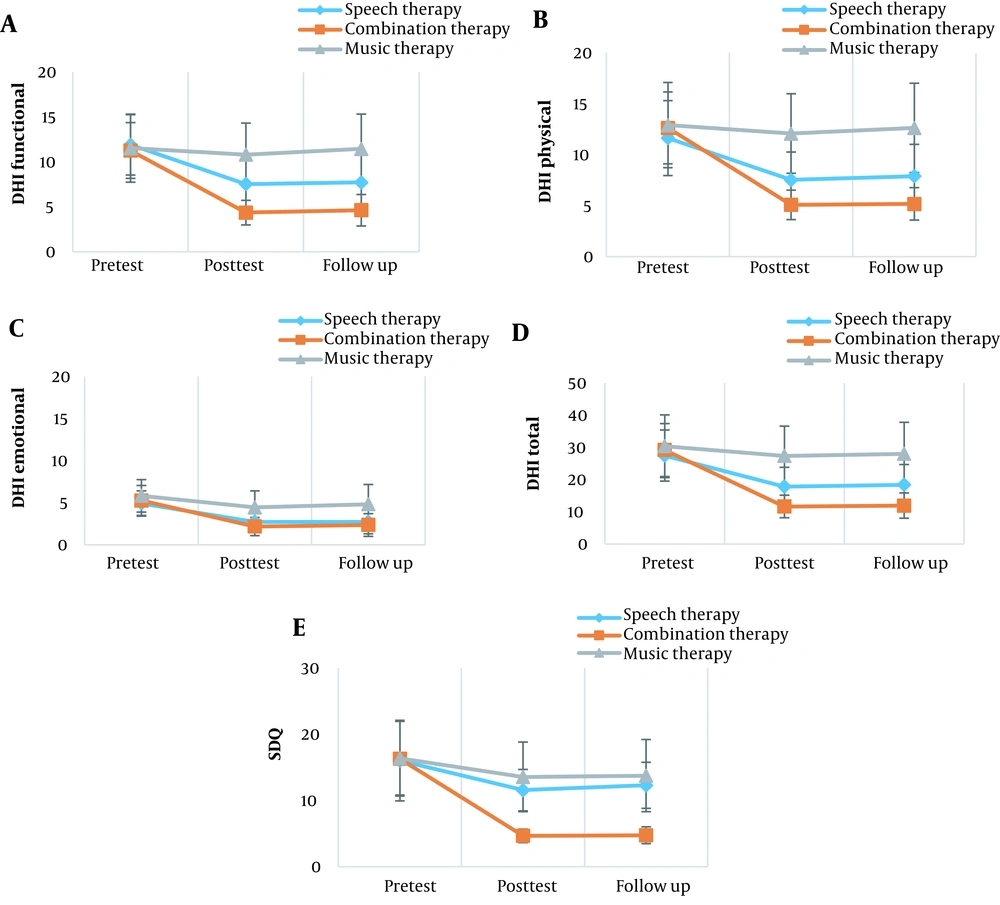
| DHI functional (n) | 11.27 ± 3.10 | 4.36 ± 1.36 | 4.64 ± 1.75 | 11.91 ± 3.36 | 7.54 ± 2.73 | 7.73 ± 3.35 | 11.54 ± 3.78 | 10.82 ± 3.49 | 11.45 ± 3.88 | 203.56 (1.36,40.70) | 56.53 (2.71,40.70) | 6.18 (2,30) | 0.000 | 0.000 | 0.006 |
| DHI physical (n) | 12.64 ± 3.53 | 5.09 ± 1.45 | 5.18 ± 1.60 | 11.64 ± 3.67 | 7.54 ± 2.73 | 7.91 ± 3.11 | 12.91 ± 4.18 | 12.09 ± 3.91 | 12.64 ± 4.39 | 187.52 (1.11,33.45) | 47.52 (2.23,33.45) | 6.73 (2,30) | 0.000 | 0.000 | 0.004 |
| DHI emotional (n) | 5.27 ± 1.79 | 2.18 ± 1.08 | 2.36 ± 1.36 | 4.91 ± 1.51 | 2.73 ± 1.19 | 2.73 ± 1.42 | 5.82 ± 1.94 | 4.45 ± 1.97 | 4.82 ± 2.36 | 187.34 (1.38,41.45) | 11.79 (2.76,41.45) | 3.92 (2,30) | 0.000 | 0.000 | 0.031 |
| DHI total (n) | 29.18 ± 8.22 | 11.64 ± 3.50 | 11.91 ± 3.91 | 27.45 ± 7.95 | 17.82 ± 5.96 | 18.36 ± 6.33 | 30.36 ± 9.73 | 27.36 ± 9.23 | 28.00 ± 9.77 | 334.08 (1.04,31.35) | 62.73 (2.09,31.35) | 6.34 (2,30) | 0.000 | 0.000 | 0.005 |
| SDQ (n) | 16.27 ± 5.64 | 4.64 ± 1.03 | 4.73 ± 1.27 | 16.00 ± 6.08 | 11.54 ± 3.14 | 12.27 ± 3.47 | 16.36 ± 5.55 | 13.54 ± 5.24 | 13.73 ± 5.46 | 104.49 (1.07,32.12) | 21.11 (2.14,32.12) | 6.27 (2,30) | 0.000 | 0.000 | 0.005 |
Abbreviations: CT, combined therapy; ST, speech therapy; MT, music therapy; dB, decibels; DHI, dysphagia handicap index; SDQ, Swallowing Disturbance Questionnaire.
a P values less than 0.05 were considered statistically significant; repeated-measures ANOVA test. Statistically significant group difference.
b Pretest: Immediately before the intervention; posttest: Immediately after the intervention; follow-up: 3 months after the intervention.
As shown in Tables 2 and 3, a repeated-measures ANOVA revealed a significant main effect of time for the SDQ and DHI scores in all 3 groups after treatment. The combination and speech therapy groups showed significant differences between the pretest and posttest, as well as between the pretest and follow-up in all study variables (P > 0.05). In the combination therapy group, there was no significant difference between the posttest and follow-up for these variables in this group (P > 0.05). Also, in the speech therapy group, there was no significant difference between the posttest and follow-up in the functional and emotional sub-test of DHI (P < 0.05). The participants in the music therapy group also showed a significant difference between the pretest and posttest and between the pretest and follow-up for SDQ, emotional subset, and the total score of DHI (P < 0.05). In this group, there was no significant difference between the posttest and follow-up for the SDQ score.
| Pre vs Post | Pre vs Follow-up | Post vs Follow-up | |||||||
|---|---|---|---|---|---|---|---|---|---|
| CT | ST | MT | CT | ST | MT | CT | ST | MT | |
| DHI functional (n) | 0.000 | 0.000 | 0.244 | 0.000 | 0.000 | 1.000 | 0.507 | 1.000 | 0.008 |
| DHI physical (n) | 0.000 | 0.000 | 0.341 | 0.000 | 0.000 | 1.000 | 1.000 | 0.038 | 0.001 |
| DHI emotional (n) | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.003 | 0.582 | 1.000 | 0.038 |
| DHI total (n) | 0.000 | 0.000 | 0.013 | 0.000 | 0.000 | 0.033 | 0.461 | 0.019 | 0.006 |
| SDQ | 0.000 | 0.001 | 0.034 | 0.000 | 0.003 | 0.042 | 1.000 | 0.009 | 1.000 |
Abbreviations: CT, combined therapy; ST, speech therapy; MT, music therapy; DHI, dysphagia handicap index; SDQ, Swallowing Disturbance Questionnaire.
a P values less than 0.05 were considered statistically significant; repeated-measures ANOVA test.
The results of between-subjects repeated measurement showed a significant main effect of interaction between the group and time for all outcomes (P < 0.05; Table 2). These results showed a significant group effect for SDQ and functional, physical, and emotional sub-tests, as well as the total score of DHI. A post hoc analysis was performed to investigate these differences and showed that the combination therapy group statistically outperformed the other 2 groups in the SDQ score. Also, the post hoc analysis showed that functional, physical, and emotional sub-tests and the total score of DHI were significantly lower in the combination therapy group than the music therapy group (P < 0.05), but these differences were not statistically significant in the speech therapy group (P > 0.05). Compared to the music therapy group, the speech therapy group significantly outperformed the physical subset of DHI, while it performed similarly to this group in other outcomes. The results are presented in Table 4 and Figure 2.
| CT vs ST | CT vs MT | ST vs MT | ||||
|---|---|---|---|---|---|---|
| Mean Difference | P Value | Mean Difference | P Value | Mean Difference | P Value | |
| DHI functional (n) | -1.97 | 0.410 | -4.51 | 0.004 | -2.54 | 0.172 |
| DHI physical (n) | -1.39 | 0.961 | -4.91 | 0.004 | -3.51 | 0.048 |
| DHI emotional (n) | -0.18 | 1.000 | -1.76 | 0.049 | -1.58 | 0.089 |
| DHI total (n) | -3.64 | 0.771 | -11.00 | 0.004 | -7.36 | 0.078 |
| SDQ | -4.72 | 0.038 | -6.00 | 0.006 | -1.27 | 1.000 |
Abbreviations: CT, combined therapy; ST, speech therapy; MT, music therapy; DHI, dysphagia handicap index; SDQ, Swallowing Disturbance Questionnaire.
a P values less than 0.05 were considered statistically significant; post hoc Bonferroni analysis.
Mean raw data for A, the functional sub-test of the dysphagia handicap index; B, the physical sub-test of the dysphagia handicap index; C, the emotional sub-test of the dysphagia handicap index; D, the total score of the dysphagia handicap index; and E, Swallowing Disturbance Questionnaire. DHI, dysphagia handicap index; SDQ, Swallowing Disturbance Questionnaire. Pretest: 1 week before the first intervention session. Posttest: 1 week after the last intervention session. Follow-up: 3 months after the last evaluation.
5. Discussion
The management of dysphagia in people with PD is limited due to the unclear mechanism of dysphagia (27). As noted in the introduction section, due to the low effect of pharmacological and surgical treatments on swallowing disorder in PD patients, there is a need to develop other treatment options that minimize patient risk and discomfort (8). Few studies without clinical trials have reported that non-instrumental behavioral therapy approaches, including intensive speech therapy (such as LSVT) and music therapy, have beneficial effects on dysphagia in PD patients (4, 6, 11). Therefore, it is reasonable to investigate and evaluate the effectiveness of such behavioral therapies in the treatment of dysphagia in PD patients. Evaluating different treatment strategies for dysphagia provides more information about available treatment options and their effectiveness on swallowing function (10). This study presents the results of a 3-arm randomized clinical trial, which for the first time, evaluated the effectiveness of a combination speech and music therapy intervention compared to conventional speech therapy and music therapy alone through telerehabilitation on swallowing function in PD patients.
The results of the present study showed significant improvements in SDQ, sub-tests (functional, physical, and emotional), and the total score of DHI in the combination therapy and speech therapy groups after treatment. These results were preserved for a 3-month follow-up period in the combination therapy group. This study also showed that the combination therapy group had a greater significant effect on the SDQ score compared to the 2 other treatments. The findings showed that according to the self-evaluation of individuals with PD, their dysphagia improved after treatment. This may indicate the benefits of combination therapy in reducing the swallowing problems of these patients. These results are largely consistent with previous studies, showing improvements in SDQ following therapy (2, 27). Also, sub-tests (functional, physical, and emotional) and the total score of the DHI changed significantly after treatment. This change was similar in the combination and speech therapy groups but greater in the combination therapy group compared to the music therapy group. This indicates that exercises in both the combination and speech therapy groups similarly affected dysphagia. Accordingly, it can be concluded that speech therapy exercises alone and without combining with other methods can improve the symptoms of dysphagia. It was expected that the speech therapy group would perform better than the music therapy group in these variables, but the speech therapy group was significantly different from the music therapy group only in the physical sub-test and the total score of DHI. This finding can be related to the small sample size and, thus, the insufficient power of the study. It seems logical to improve the emotional sub-test in the music therapy group through enjoyable singing exercises. Some studies have shown that the swallowing-specific quality of life was highly correlated with the general health-related quality of life and depression (10).
Previous studies have shown that exercises that recruit the muscles involved in swallowing can improve the symptoms of dysphagia in persons with PD and improve the quality of life related to swallowing in these patients (10, 13). Combination therapy also works on overall pharyngolaryngeal efforts, including respiratory support, vocal cord adduction, and lingual function, all of which are required in speech acts and swallowing (6, 10, 31). In general, the results of these studies are based on the hypothesis that intensive speech therapy leads to stronger expiratory and laryngeal muscle movements and, as a result, improves the pharyngoesophageal swallowing function, reduces the symptoms of swallowing disorders, and improves the quality of life related to swallowing in persons with PD (4, 6). Hypertonia, rigidity, involuntary movements, and bradykinesia in PD can cause disturbances in swallowing motor control and increase the risk of penetration and laryngeal aspiration (4, 6, 27). These symptoms are most likely to be ameliorated by speech therapy, and therefore some of the observed improvements in swallowing function in the present study seem logical (10). Before each swallow, the larynx should be closed to prevent airway violation. The suprahyoid musculature, which elevates and protects the larynx, is likely to be activated by high repetition, high intensity, and increasing speech complexity, pitch, and loudness exercises (6). It is possible that with improvements in airway protection, confidence in swallowing is also improved, and effortful swallowing is facilitated. Because poor pharyngeal constriction and the presence of pharyngeal residue are predictive of aspiration, improvements in these measures reduce the risk of aspiration (6, 10). Interestingly, patients were encouraged to increase their water intake during the treatment program, likely resulting in an increase in total daily swallowing. Also, the patient’s perceptual responses may be influenced by the patient’s frequent interaction with the therapist during treatment (6). Direct feedback combined with frequent encouragement can enhance motor learning and increase motivation. This supportive effect may contribute to the observed positive effects of the therapy, leading to improvement in swallowing symptoms (4, 6). In summary, the findings of this study showed that the positive outcomes of swallowing might result from the activation of the neuromuscular control of the entire aerodigestive tract (10). It can be concluded that combination therapy strengthens the neural pathways and increase the tone, strength, and recruitment of the laryngeal muscles. Now that we have clear evidence of treatment efficacy, further studies with a range of instrumental assessment methods are recommended to quantify changes in swallowing after treatment. It is also suggested to investigate the effect of treatment on other swallowing outcomes in people with PD. Future considerations, including longer duration of follow-up and repeat studies with combination therapy, could show repeated effect or cumulative improvement. Also, future investigations could evaluate whether similar results can be obtained in individuals with more severe swallowing problems.
5.1. Limitations
This study has several limitations, such as the small sample size. The lack of patient blinding for treatment groups creates a further risk of bias. In this study, we use telerehabilitation to deliver the treatment. Since this method is relatively new, it may have limitations. In telerehabilitation, patients need the presence of a caregiver to give feedback and guide the treatment session due to the type of telerehabilitation of this intervention. Internet problems (such as low speed, internet disconnection, etc) are also another limitation of telerehabilitation.
5.2. Conclusions
The findings of this study showed that combination therapy delivered through telerehabilitation might have additional effects on swallowing function in people with PD compared to speech therapy and music therapy alone. This study provided more evidence for the claim of previous studies to improve the swallowing function of patients with PD following speech therapy. We believe that the results of this study can provide a clinical basis for low-cost and enjoyable behavioral treatments for treating dysphagia in persons with PD. This study also demonstrated the potential of delivering therapy via videoconference using smartphones to improve swallowing functions. More information (such as local SLPs’ perspectives) will be valuable to support the utilization of the smartphone videoconference method as an alternative mode of service provision for individuals with PD.