1. Background
Acute pain should be defined as the initiation phase of a wide ranging, persistent nociceptive and behavioral cascade triggered by tissue damage, neuropathic injury, inflammation and provocation of nociceptive transducers at the site of the injury (1, 2). Inflammatory pain is a type of pathological pain caused by direct stimulation of nociceptors, which are altered by release of many inflammatory mediators such as bradykinin, histamine and cytokines (3, 4). Rheumatoid arthritis (RA) is an inflammatory autoimmune disease which is characterized by pain, edema, hyperalgesia, allodynia and cartilage and bone destruction (3). Cytokines (interleukins (ILs), interferon, and tumor necrosis factor (TNF-α)), are released by immune system cells and have a notable role in the inflammatory pain process. Pro-inflammatory cytokines (IL-1β and TNF-α) can activate peripheral nociceptors directly and lead to acute pain. They also modify the response properties of primary afferent fibers to noxious stimuli. This may arise as a result of changes to the sensitivity of the receptor ligand themselves or through voltage gated channels modulation (5, 6). Transcription factors for instances nuclear factor kappa-B (NF-κB) are a major regulator of pro-inflammatory cytokines-inducible gene regulation. NF-κB is activated in the dorsal root ganglion and spinal cord, which are involved in the nociceptive information transduction, conduction and transmission (7).
Probiotics are living and health-beneficial bacteria in the human gut flora and are commensalism and harmless. Probiotic bacteria, including Lactobacillus and Bifidobacterium species, can inhibit the growth of many types of pathological cells and may also have more direct effects on the immune system. Probiotics have immune regulatory properties and can modify the function (proliferation, activation and effective performance) of a wide variety of immune cells including B-lymphocytes, T-lymphocytes, anti and pro-inflammatory cytokines production and modulating the intracellular signaling pathways including NF-κB (4, 8).
2. Objectives
Based on the important role of the cellular and humoral immunity in induction of inflammatory diseases such as RA and potency of probiotic bacteria in immune response modulation, in this study, the effect of oral administration of probiotics on hyperalgesia, edema, serum levels of TNF-α and IL-1β and NF-κB expression during the acute phase of inflammatory pain in male rats was investigated.
3. Methods
3.1. Laboratory Animals
Adult (200 - 220 g) male Wistar rats were kept in groups of 6 animals in cages maintained at 20 ±2°C with a 12-hour light/dark cycle, 60% - 70 % humidity and food and water ad libitum. All experimental procedures were approved by the local ethics committee for the use of animals in the research, and were followed guidelines issued by the international association for the study of pain (IASP). Inflammation was induced by a single intraplantar injection (100 µL) of complete Freund’s adjuvant (CFA) (suspension of heat-attenuated Mycobacterium tuberculosis in sterile mineral oil) (Sigma, St Lois, MO, and USA) into the left rats hind paw.
3.2. Experimental Procedure
Rats were randomly divided into 7 experimental groups and each group was divided into 3 subgroups based on different time points (days 0, 3, and 7) (n = 6 rats / each group). Group 1 served as CFA and group 2 served as CFA control and was given sterile mineral oil. Group 3 served as CFA + vehicle (CFA+ Vehi) and was given distilled water. Groups 4, 5 and 6 served as CFA + Pb1, 2, 3 and were oral (gavage) administered different dosage of probiotics daily (1/250, 1/500 and 1/1000 (109 CFU/g), respectively). Group 7 served as positive control (CFA + Indo) and was given indomethacin orally at a dose level of 5 mg/kg daily (Figure 1) (9-11).
3.3. Behavioral Assessment
Transcription factors such as nuclear factor-kappa B (NF-kB) provide a link between early signaling events and changes in gene expression.
As in the periphery, target genes for NF-kB in the brain encode proteins with immune and inflammatory activities.
Recent reports indicate that the activation of NF-kB occurs in the dorsal root ganglion (DRG) and spinal cord, which are both involved in the transmission and processing of nociceptive information.
Nociceptive thresholds to thermal stimulation were determined using the Hargreaves test. Animals were placed in a Perspex chambers and thermal stimulus was projected to the plantar surface of the hind paw and paw withdrawal latency (PWL) was recorded. Each rat received three trials at an interval of 5 - 10 minutes. The mean value for the affected paw was subtracted from other paw and the obtained result considered as the hyperalgesia sign in the injured paw (11).
3.4. Paw Volume Assessment
The paw volume was measured using a plethysmometer (Ugo Basile; model7141; Comerio VA, Italy) with a resolution of 0.01 mL during different time points of the study. Rats’ hind paw was submerged to the tibiotarsal joint into an electrolyte-filled Perspex chamber of the plethysmometer. The volume of liquid displacement, which is equal to the paw volume, was shown on a digital monitor. Volume measurement was done twice for each paw, and the average calculated (12).
3.5. Blood Sampling and Measuring Serum Levels of TNF-α and IL-1β
Rats were anesthetized with methoxy isoflurane and their blood samples were taken from the vessel retro-orbital corners of their eyes. Blood samples were centrifuged (13000 rpm, 4°C) and the serum was stored at -80°C. The serum TNF-α and IL-1β levels of rats were measured in duplicate by a rat standard ELISA kits (High Sensitivity Quantikine kit, R&D Systems, Minneapolis) on different time points of the study and the average of the two measures was used in the analysis (13).
3.6. Spinal NF-κB Expression Detection by Western Blotting
After behavioral and inflammatory tests in order to identify the spinal NF-κB expression during the acute phase of the inflammatory pain, the lumbar segments of rat’s spinal cord (L1 - L5) were removed (150 mg form each). For this purpose, in order to spinal cord tissue extraction, rats were anesthetized with methoxy isoflurane and their heads removed. Their lumbar segment of the spinal cord was separated rapidly on ice, and then they were frozen in liquid nitrogen and maintained at a temperature of -80°C. Then, samples were lysed with a lysis buffer. The proteins were transferred to Immobilon-PVDF membrane (Millipore, Bedford, MA, USA) using mini-protein II (Bio-Rad) at 100 V and 0.35A for 60 minutes. Blocked for 75 minutes in 5% BSA at 25°C. The membrane was incubated at 4°C overnight with specific primary antibody in a blocking buffer (anti-NF-κB) (1: 1000; Abcam). The membrane was washed trice with TBS-T and then incubated (75 minutes, 25°C) with secondary antibody in the blocking buffer (1: 10000; Abcam). The immunoreactivity of the proteins on the membrane was visualized using a chemiluminescence detection system (ECL, Amersham). Band intensities were measured densitometrically by means of the J-Image software and to measure the variations of spinal NF-κB expression, the ratio of NF-κB to β-actin was calculated (14).
3.7. Statistical Analysis
Results were expressed as Mean ± SEM. To compare within the group edema, hyperalgesia and serum TNF-α and IL-1β levels and spinal NF-κB activity, the repeated measurement ANOVA test (One-way ANOVA) and post-hoc Tukey’s test were used. Moreover, to compare more accurately the changes of edema, hyperalgesia and serum TNF-α and IL-1β levels and spinal NF-κB activity on the same days between the two groups, the unpaired student t-test was used. SPSS software version 22 was used for data analysis and the charts were analyzed via Excel. Statistical significance was accepted at P ≤ 0.05.
4. Results
4.1. Paw Edema Variations During the Acute Phase of Inflammation
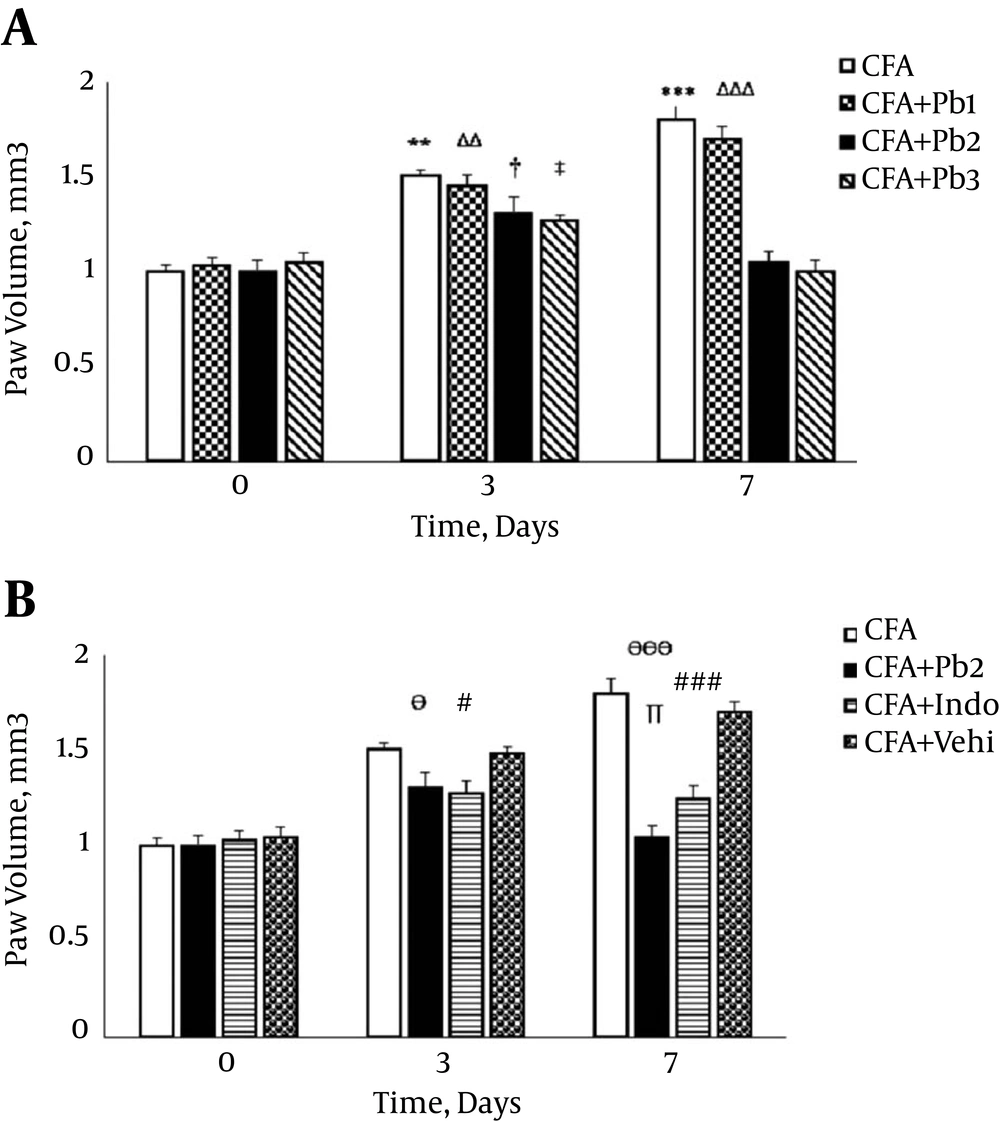
Paw volume significantly increased after the CFA injection compared to day 0 (P = 0.0081 for day 3 and P < 0.001 for day 7). There was no significant difference in paw volume variations compared to the baseline in the CFA control group during the different time points of this study (data not shown). The paw volume significantly increased compared to the baseline in the CFA + Pb1 group (P = 0.0077 for day 3 and P < 0.001 for day 7). The results of this study showed that oral administration of probiotics (1/250) did not have a significant effect on changes of the paw volume in the CFA + Pb1 group compared to the CFA group in different time points of the study. There was no significant difference in changes of the paw volume following the administration of doses of 1/500 and 1/1000 of probiotics (Figure 2A). Furthermore, oral administration of probiotics (1/500) as an effective dose caused a significant reduction in the paw volume in the CFA + Pb2 group, compared to the CFA group (P = 0.032 for day 3 and P < 0.001 for day 7). Also, administration of indomethacin with a dose of 5 mg/kg significantly reduced the paw volume in the CFA+ Indo group compared to the CFA group (P = 0.035 for day 3 and P < 0.001 for day 7). Comparison of the two groups, the CFA + Pb2 and CFA + Indo, showed that the effective dose of probiotics (1/500) significantly reduced the paw volume on day 7 compared to the same day in the CFA + Indo group (P = 0.024). It should be noted that changes in the paw volume in the CFA + Vehi group showed no significant difference compared to the CFA group (Figure 2B).
A, Oral administration of probiotics reduced edema caused by the plantar injection of CFA in acute inflammation dose dependently; Results were expressed as mean ± SEM and n = 6 / group; ** P ≤ 0.01 and *** P ≤ 0.001: comparison of edema in different days in the CFA group with day zero; ΔΔ P ≤ 0.01 and ΔΔΔ P ≤ 0.001; comparison of edema in different days in the CFA + Pb1 group with day zero; † P ≤ 0.05: comparison of edema in different days in the CFA + Pb2 group with day zero; ‡ P ≤ 0.05: comparison of edema in different days in the CFA + Pb3 group with day zero. B, Effective doses of probiotics (1/500) reduced paw edema significantly during the acute inflammation in the CFA + Pb2 group compared to the CFA+ Indo group; results were expressed as mean ± SEM and n = 6 / group; Ө P ≤ 0.05 and ӨӨӨ P ≤ 0.001: comparing the changes of paw edema between the CFA and CFA + Pb2 groups during different days of the study; # P ≤ 0.05 and ### P ≤ 0.001: comparing the changes of paw edema between the CFA and CFA + indo groups during different days of the study; ∏ P < 0.05: comparing the changes of paw edema between the CFA + Indo and CFA + Pb2 (1/500) groups during different days of the study.
4.2. Paw Hyperalgesia Variations During the Acute Phase of Inflammation
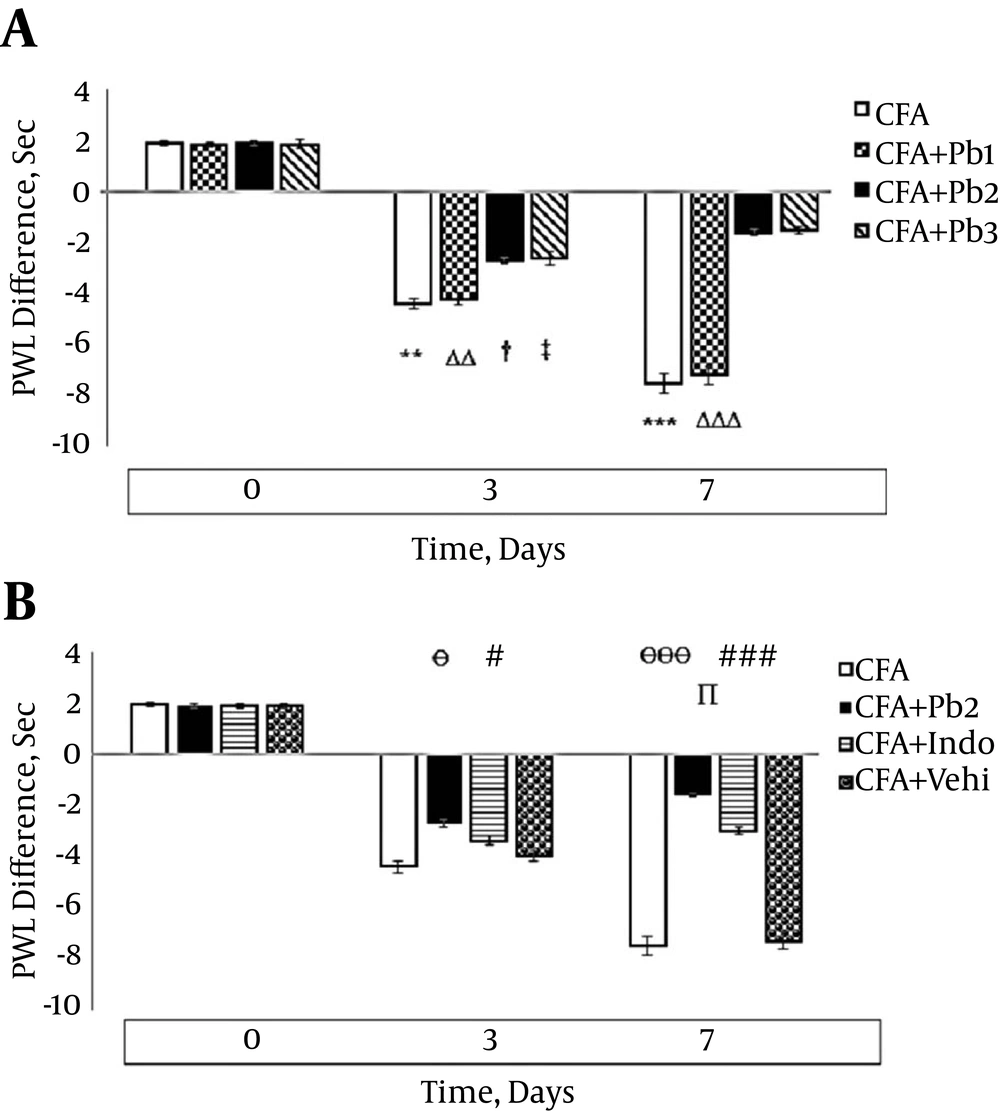
Thermal hyperalgesia significantly increased on the 3rd and 7th days after the CFA injection compared to that on day 0 (P = 0.0084 for day 3 and P < 0.001 for day 7). There was no significant difference in hyperalgesia variations compared to the baseline in the CFA control group during the different stages of this study (data not shown). Hyperalgesia significantly increased on the 3rd and 7th days of the study compared to the baseline in the CFA+Pb1 group (P = 0.0078 for day 3 and P < 0.001 for day 7). Oral administration of probiotics (1/250) did not have a significant effect on changes of hyperalgesia in the CFA + Pb1 group compared to the CFA group during the acute phase of inflammation (Figure 3A). Furthermore, oral administration of probiotics with a dosage of 1/500 as an effective dose caused a significant reduction in hyperalgesia in the CFA + Pb2 group, compared to the CFA group (P = 0.038 for day 3 and P < 0.001 for day 7). Administration of indomethacin reduced hyperalgesia in the CFA + Indo group compared to the CFA group (P = 0.043 for day 3 and P < 0.001 for day 7). Comparison of the two groups, the CFA + Pb2 and CFA + Indo, showed that probiotic administration (1/500) significantly reduced hyperalgesia on day 7 compared to the CFA + Indo group (P = 0.032) (Figure 3B).
A, Oral administration of probiotics reduced paw hyperalgesia caused by plantar injection of CFA in acute inflammation dose dependently; results were expressed as mean ± SEM and n = 6 / group; ** P ≤ 0.01 and *** P ≤ 0.001: comparison of hyperalgesia in different days in the CFA group with day zero; ΔΔ P ≤ 0.01 and ΔΔΔ P ≤ 0.001: comparison of hyperalgesia in different days in the CFA + Pb1 group with day zero; † P ≤ 0.05: comparison of hyperalgesia in different days in the CFA + Pb2 group with day zero; ‡ P ≤ 0.05: comparison of hyperalgesia in different days in the CFA + Pb3 group with day zero. B, Effective doses of probiotics (1/500) reduced paw hyperalgesia significantly during acute inflammation in the CFA + Pb2 group compared to the CFA + Indo group; results were expressed as mean ± SEM and n = 6 / group; Ө P ≤ 0.05 and ӨӨӨ P ≤ 0.001: comparing the changes of hyperalgesia between the CFA and CFA + Pb2 groups during different days of the study; # P ≤ 0.05 and ### P ≤ 0.001: comparing the changes of hyperalgesia between the CFA and CFA + Indo groups during different days of the study; ∏ P < 0.05: comparing the changes of hyperalgesia between the CFA + Indo and CFA + Pb2 (1/500) groups during different days of the study.
4.3. Serum IL-1β Levels During the Acute Phase of Inflammation
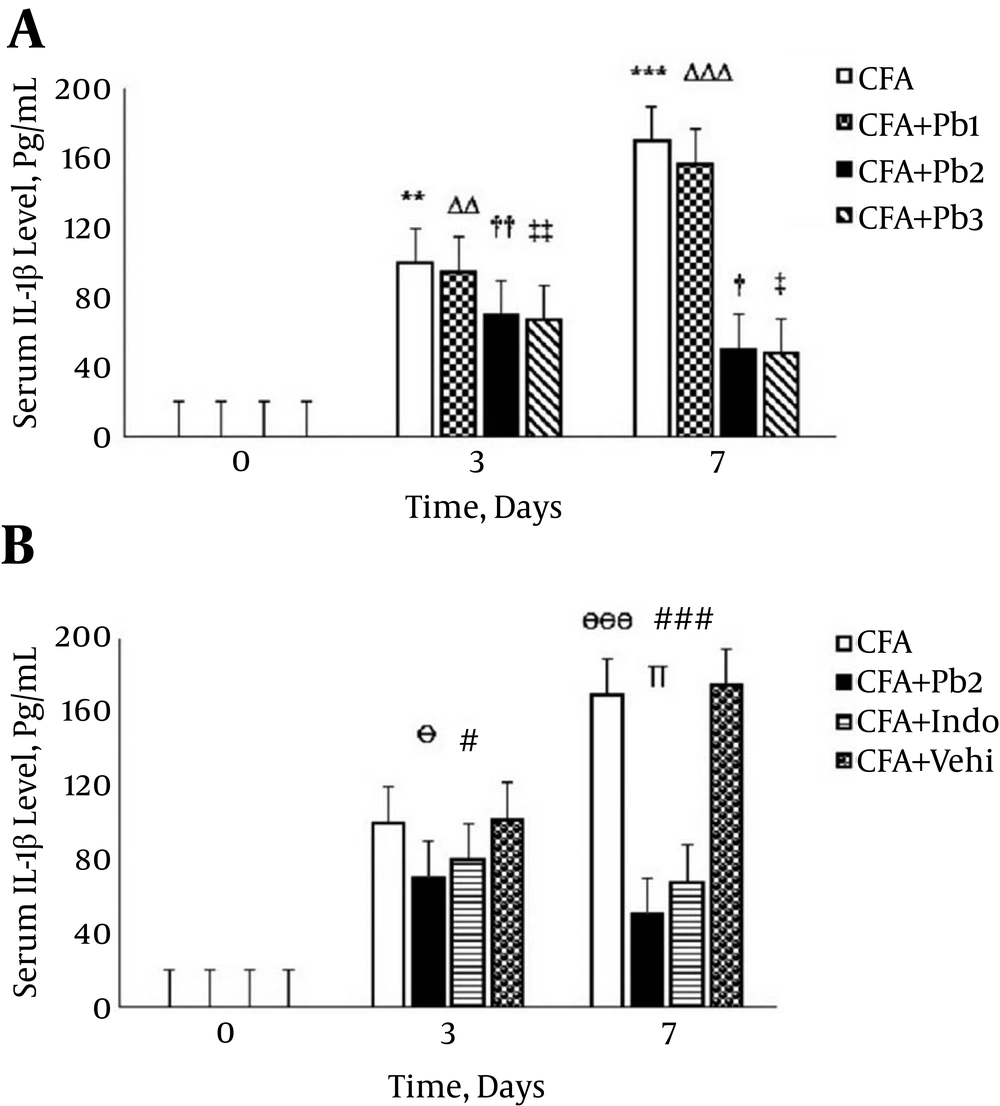
Serum IL-1β levels significantly increased on the 3rd and 7th days after the CFA injection compared to that on day 0 (P = 0.0085 for day 3 and P < 0.001 for day 7). There was no significant difference in serum levels of IL-1β compared to the baseline in the CFA control group during the different stages of the study (data not shown). Serum levels of IL-1β significantly increased on the 3rd and 7th days of the study compared to the baseline in the CFA + Pb1 group (P = 0.0073 for day 3 and P < 0.001 for day 7). Oral administration of probiotics (1/250) did not have a significant effect on changes of serum levels of IL-1β in the CFA + Pb1 group compared to the CFA group during the acute phase of inflammation. There was no significant difference in changes of serum levels of IL-1β following administration of doses of 1/500 and 1/1000 of probiotics (Figure 4A). Furthermore, oral administration of probiotics (1/500) caused a significant reduction in serum levels of IL-1β in the CFA + Pb2 group, compared to the CFA group (P = 0.038 for day 3 and P < 0.001 for day 7). On the other hand, administration of indomethacin significantly reduced serum levels of IL-1β in the CFA + Indo group in days 3 and 7 of the study in comparison with the CFA group (P = 0.041 for day 3 and P < 0.001 for day 7). Comparison of the two groups, the CFA + Pb2 and CFA + Indo, showed that probiotics (1/500) significantly reduced serum levels of IL-1β on day 7 compared to the same day in the CFA + Indo group (P = 0.033) (Figure 4B).
A, Oral administration of probiotics reduced serum IL-1β levels caused by the plantar injection of CFA in acute inflammation dose dependently; results were expressed as mean ± SEM and n = 6 / group; ** P ≤ 0.01 and *** P ≤ 0.001: comparison of serum IL-1β levels in different days in the CFA group with day zero; ΔΔ P ≤ 0.01 and ΔΔΔ P ≤ 0.001: comparison of serum IL-1β levels in different days in the CFA + Pb1 group with day zero; † P ≤ 0.05 and †† P ≤ 0.01: comparison of serum IL-1β levels in different days in the CFA + Pb2 group with day zero; ‡ P ≤ 0.05 and ‡‡ P ≤ 0.01: comparison of serum IL-1β levels in different days in the CFA + Pb3 group with day zero. B, Effective doses of probiotics (1/500) reduced serum IL-1β levels significantly during acute inflammation in the CFA + Pb2 group compared to the CFA+ Indo group. Results were expressed as mean ± SEM and n = 6 / group; Ө P ≤ 0.05 and ӨӨӨ P ≤ 0.001: comparing the changes of serum IL-1β levels between the CFA and CFA + Pb2 groups during different days of the study; # P ≤ 0.05 and ### P ≤ 0.001: comparing the changes of serum IL-1β levels between the CFA and CFA + Indo groups during different days of the study; ∏ P < 0.05: comparing the changes of serum IL-1β levels between the CFA + Indo and CFA + Pb2 (1/500) groups during different days of the study.
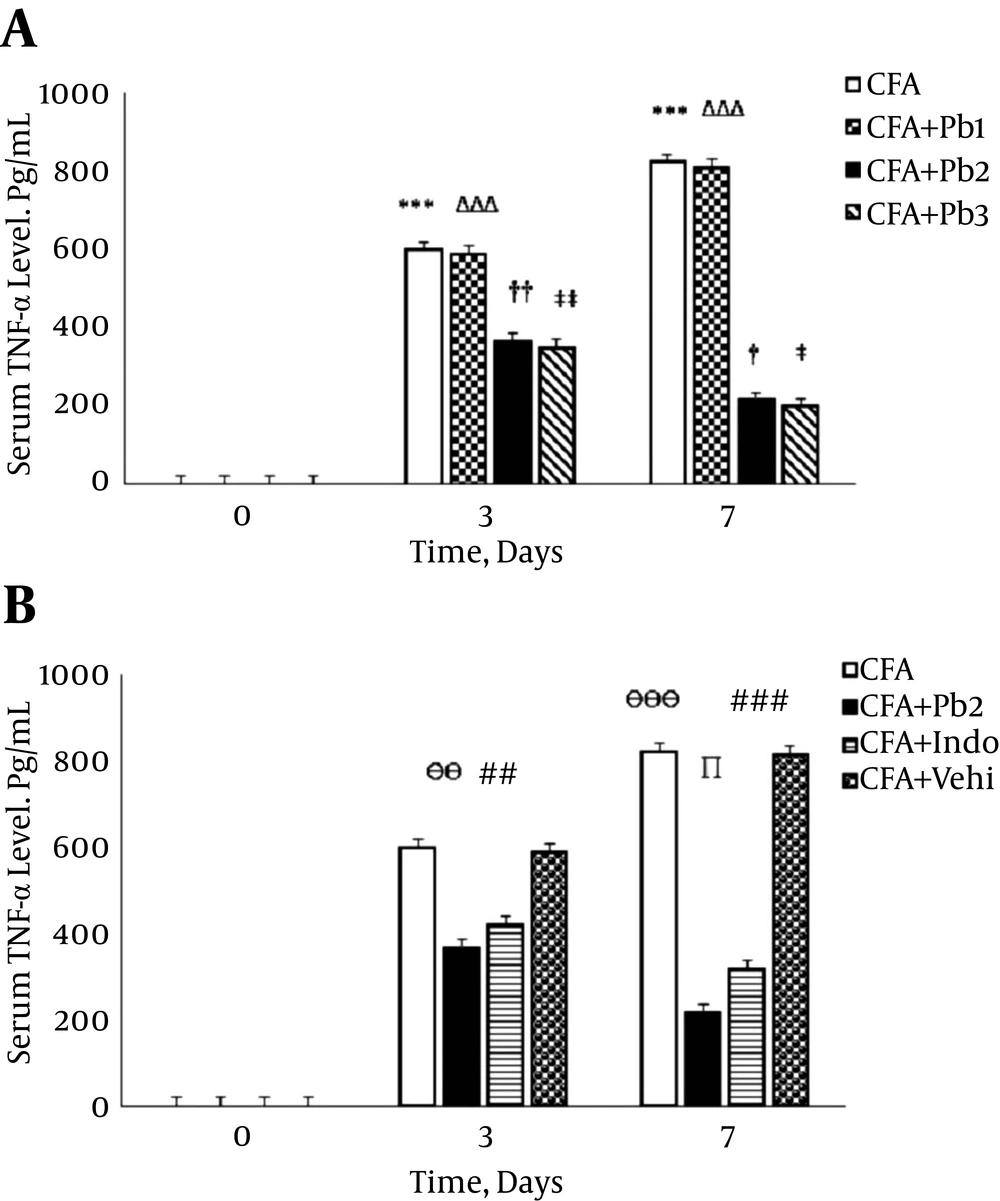
4.4. Serum TNF-α Levels During the Acute Phase of Inflammation
Serum TNF-α levels significantly increased on the 3rd and 7th days after the CFA injection compared to that on day 0 (P < 0.001). There was no significant difference in serum levels of TNF-α compared to the baseline in the CFA control group during the different stages of this study (data not shown). Serum levels of TNF-α significantly increased on the 3rd and 7th days of the study compared to the baseline in the CFA + Pb1 group (P < 0.001). Oral administration of probiotics (1/250) did not have a significant effect on changes of the serum levels of TNF-α in the CFA + Pb1 group compared to the CFA group during the acute phase of inflammation. There was no significant difference in changes of the serum levels of TNF-α following administration of doses of 1/500 and 1/1000 of probiotics (Figure 5A). Furthermore, oral administration of probiotics with a dosage of 1/500 as an effective dose caused a significant reduction in serum levels of TNF-α in the CFA + Pb2 group, compared to the CFA group (P = 0.0074 for day 3 and P < 0.001 for day 7). Also, administration of indomethacin significantly reduced the serum levels of TNF-α in the CFA + Indo group in comparison with the CFA group (P = 0.0083 for day 3 and P < 0.001 for day 7). Comparison of the two groups, the CFA + Pb2 and CFA + Indo, showed that probiotics (1/500) significantly reduced serum levels of TNF-α on day 7 compared to the same day in the CFA + Indo group (P=0.023) (Figure 5B).
A, Oral administration of probiotics reduced serum TNF-α levels caused by the plantar injection of CFA in acute inflammation dose dependently. Results were expressed as mean ± SEM and n = 6 / group; *** P ≤ 0.001: comparison of serum TNF-α levels in different days in the CFA group with day zero; ΔΔΔ P ≤ 0.001: comparison of serum TNF-α levels in different days in the CFA + Pb1 group with day zero; † P ≤ 0.05 and †† P ≤ 0.01: comparison of serum TNF-α levels in different days in the CFA + Pb2 group with day zero; ‡ P ≤ 0.05 and ‡‡ P ≤ 0.01: comparison of serum TNF-α levels in different days in the CFA + Pb3 group with day zero. B, Effective doses of probiotics (1/500) reduced serum TNF-α levels significantly during acute inflammation in the CFA + Pb2 group compared to the CFA + Indo group; results were expressed as mean ± SEM and n = 6 / group; ӨӨ P ≤ 0.01 and ӨӨӨ P ≤ 0.001: Comparing the changes of serum TNF-α levels between the CFA and CFA + Pb2 groups during different days of the study; ## P ≤ 0.01 and ### P ≤ 0.001: Comparing the changes of serum TNF-α levels between the CFA and CFA + Indo groups during different days of the study; ∏ P < 0.05: comparing the changes of serum TNF-α levels between the CFA + Indo and CFA + Pb2 (1/500) groups during different days of the study.
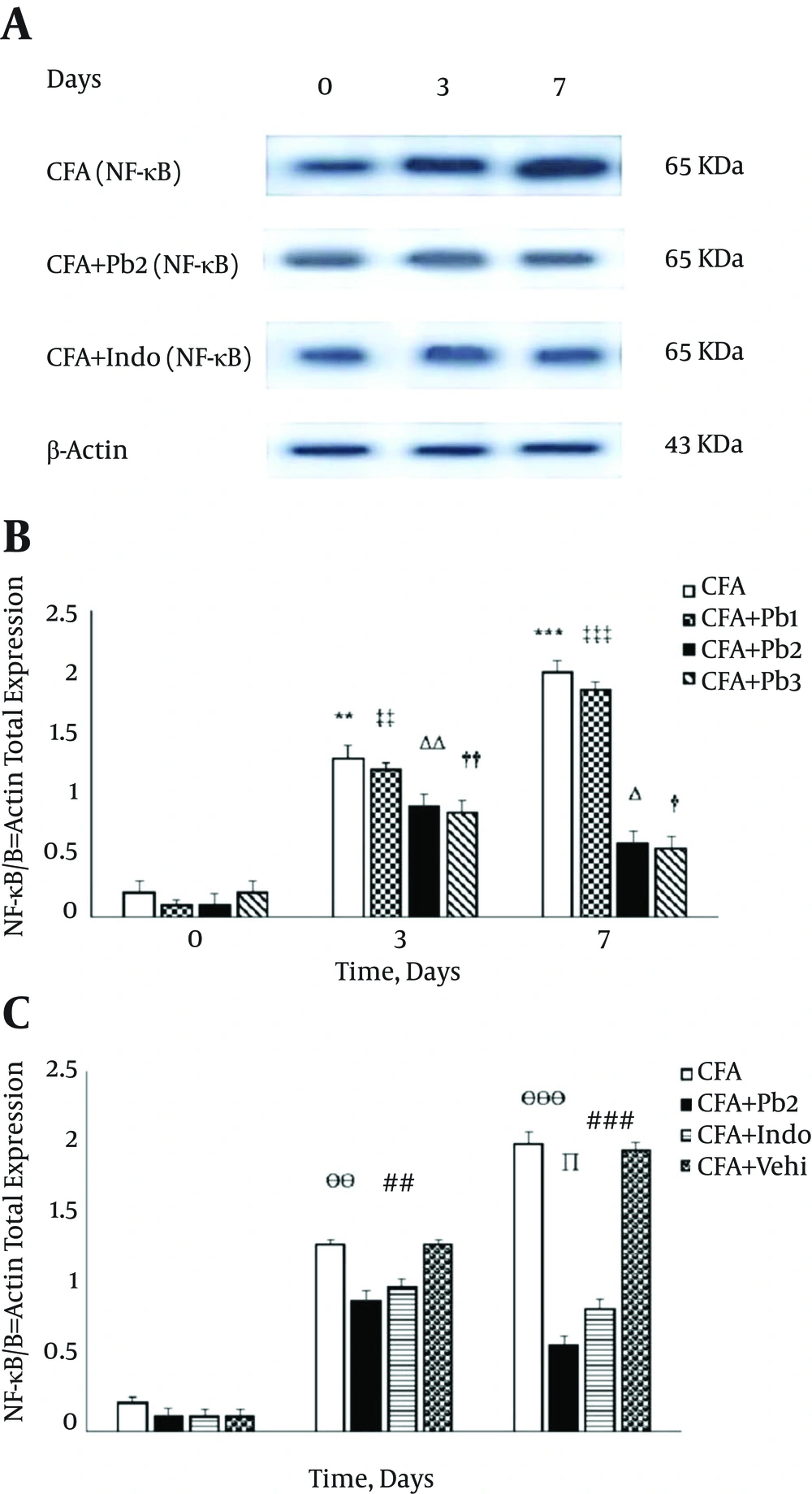
4.5. Variations in Spinal NF-κB Expression During the Acute Phase of Inflammation
Protein expression of NF-κB showed similar bands with molecular masses of approximately 65 kDa in the spinal cord tissues of all experimental groups. To normalize differences in protein-loading, all data were expressed as NF-κB/β-actin ratios. Densitometry of the data, which were obtained from the Western blotting technique demonstrated that the NF-κB/β-actin ratios significantly increased on 3 and 7 days after the CFA injection compared to day 0 in the CFA group (P = 0.0081 for day 3 and P < 0.001 for day 7) (Figure 6A). There was no significant difference in the expression of the spinal NF-κB/β-actin compared to the baseline in the CFA control group during the different stages of this study (data not shown). Expression of the spinal NF-κB/β-actin significantly increased on the 3rd and 7th days of the study compared to the baseline in the CFA + Pb1 group (P = 0.0078 for day 3 and P < 0.001 for day 7). Oral administration of probiotics (1/250) did not have a significant effect on changes of expression of the spinal NF-κB/β-actin in the CFA + Pb1 group compared to the CFA group during the acute phase of inflammation. There was no significant difference in changes of expression of the spinal NF-κB/β-actin following the administration of doses of 1/500 and 1/1000 of probiotics (Figure 6B). Furthermore, oral administration of probiotics with a dosage of 1/500 as an effective dose caused a significant reduction in the activity of the spinal NF-κB/β-actin in the CFA + Pb2 group, compared to the CFA group (P = 0.0067 for day 3 and P < 0.001 for day 7). Also, administration of indomethacin significantly reduced the expression of the spinal NF-κB/β-actin in the CFA + Indo group compared to the CFA group (P = 0.0074 for day 3 and P < 0.001 for day 7). Comparison of the two groups, the CFA + Pb2 and CFA + Indo, showed that probiotics (1/500) significantly reduced expression of the spinal NF-κB/β-actin on day 7 compared to the same day in the CFA + Indo group (P = 0.023) (Figure 6C).
A, Immunoblots of spinal NF-κB enzyme expression (65kDa) throughout different stages of inflammation (days 0, 3 and 7) in experimental groups; All densitometry data were demonstrated as NF-κB/β-actin ratios; data are represented as mean ± SEM (n = 6/group). B, Oral administration of probiotics reduced NF-κB/β-actin ratio caused by the plantar injection of CFA in acute inflammation dose dependently. Results were expressed as mean ± SEM and n = 6 / group; ** P ≤ 0.01 and *** P ≤ 0.001: comparison of NF-κB/β-actin ratio in different days in the CFA group with day zero; ΔΔ P ≤ 0.01 and ΔΔΔ P ≤ 0.001: comparison of NF-κB/β-actin ratio in different days in the CFA + Pb1 group with day zero; † P ≤ 0.05 and †† P ≤ 0.01: comparison of NF-κB/β-actin ratio in different days in the CFA + Pb2 group with day zero; ‡ P ≤ 0.05 and ‡‡ P ≤ 0.01: comparison of NF-κB/β-actin ratio in different days in the CFA + Pb3 group with day zero. C, Effective doses of probiotics (1/500) reduced NF-κB/β-actin ratio significantly during acute inflammation in the CFA + Pb2 group compared to the CFA + indo group; results were expressed as mean ± SEM and n = 6 / group; ӨӨ P ≤ 0.01 and ӨӨӨ P ≤ 0.001: comparing the changes of NF-κB/β-actin ratio between the CFA and CFA + Pb2 groups during different days of the study; ## P ≤ 0.01 and ### P ≤ 0.001: comparing the changes of NF-κB/β-actin ratio between the CFA and CFA + Indo groups during different days of the study; ∏ P < 0.05: comparing the changes of NF-κB/β-actin ratio between the CFA + Indo and CFA + Pb2 (1/500) groups during different days of the study.
5. Discussion
Our results revealed a role of probiotics in reducing hyperalgesia, edema, serum levels of IL-1β and TNF-α and spinal P38MAPK activity during the acute phase of inflammatory pain caused by CFA. Probiotics with a low dose did not change inflammatory symptoms, serum levels of IL-1β and TNF-α and NF-κB expression significantly compared to the CFA group. Plantar injection of CFA in this study induced inflammation, hyperalgesia and edema in the hind paw, which continued up to day 7.
Pain perception is triggered by noxious stimuli, such as an injury, inflammation or diseases. Acute pain is initially associated with specific autonomous and somatic reflexes, but these disappear in patients with chronic pain (1). The CFA-induced inflammation model is used as an inflammatory pain model, which has long been accepted as a useful tool for investigating pathophysiological and pharmacological changes during inflammatory pain and new anti-inflammatory drugs (11). Scientists showed that plantar injection of CFA causes mechanical and thermal hyperalgesia that accompanies long-lasting inflammation in the affected paw. In the acute phase of CFA-induced arthritis, secretion of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α were increased and caused hyperalgesia and edema (15). According to Hargreaves, the rat, due to its inflammation induced by plantar injection of CFA, had withdrawn his inflamed paw from the motivating factor, but some other studies have shown that motor behaviors because of the inflammation are normal and no significant changes in motor activities are observed (3). Rezazadeh et al. (11) have shown that edema and hyperalgesia induced by CFA two hours after the injection in the rat’s hind paw were started and continued for at least a month. Our results showed that the CFA-injected paw was clarified by a rapid onset of paw edema and hyperalgesia obvious within 24 hours and increased up to day 7 after the injection.
Increasing evidence has indicated the notable role of pro-inflammatory cytokines, particularly TNF-α and IL-1β in the acute phase of inflammation and involvement of these factors in inflammatory pain induction and modulation by interfering with nociceptive transduction, conduction, and transmission (12, 16). In acute conditions, TNF-α has been reported to sensitize nociceptive neurons indirectly through the induction of pro-inflammatory cytokines cascade involving IL-1β and IL-6 resulting in the release of inflammatory mediators from immune cells (17). Also, IL-1β has an important role in induction of hyperalgesia through direct action on neurons, stimulation of pain receptors and its central role in joint and cartilage inflammation and destruction (18). Accordingly, the results of this study showed that the inflammation caused by the plantar injection of CFA significantly increased serum levels of IL-1β and TNF-α in the acute phase of inflammation. In this regard, our results revealed that inflammatory symptoms such as edema and hyperalgesia reduced after oral administration of probiotics with doses of 1/500 and 1/1000 compared to the CFA group. In addition, oral administration of 1/500 and 1/1000 doses of probiotics significantly decreased serum levels of IL-1β and TNF-α during the acute phase of inflammatory pain compared to the CFA group.
Human studies have applied different strains of probiotics, and all of them have reported functional improvement or subjective well-being in those receiving the treatment with different strains of probiotics. Scientists have illustrated that probiotics reduced pro-inflammatory cytokines (i.e., interleukins-1β, 2, 6, 12, and 17, interferon-γ, and TNF-α), and increased regulatory cytokines (interleukin-10 and transforming growth factor-β) (8, 19).
During the early stages of the inflammation, inflammatory mediators and cytokines change the receptors sensitivity and declined activation threshold for conducting ion-channels. Cytokines are potent hyperalgesic agents which may act directly on nociceptors or indirectly, stimulating the release of inflammatory mediators (5, 20). During the acute phase of inflammatory pain, most of the inflammatory factors linked to RA have been shown to be regulated by the intracellular signaling pathway agents such as NF-κB (18). The NF-κB agent regulates gene transcription of many pro-inflammatory cytokines and is a key regulator of TNF-α gene activation (21). A dysregulation of NF-κB has been illustrated to play a notable role in many inflammatory diseases, including RA and osteoarthritis (22). Accordingly, our findings showed that the inflammation caused by the CFA injection significantly increased NF-κB expression in the acute phase of inflammation. In summary, considering different aspects of hyperalgesia inducement, it can be concluded that it can also be resulted that during acute inflammatory pain, a part of NF-κB effects on hyperalgesia is mediated by TNF-α and IL-1β expression.
Probiotic bacteria are able to increase levels of anti-inflammatory cytokines and immunoglobulins, autoimmune modulation and immune stimulation against pathogenic bacteria. Also, probiotics can increase immune cell proliferation and decrease the pro-inflammatory cytokines production (3, 23). It is evident that NF-κB is involved in controlling cellular response to various stimuli and plays a particularly considerable role in response to inflammation by triggering production of transiently expressed protein synthesis and protein processing (24-26). Several lines of evidence support that activation of intracellular signaling pathways and inflammatory cytokines have an important role in pain induction during inflammation. Thus, it seems that inhibition of pro-inflammatory cytokines like IL-1β and TNF-α and intracellular signaling pathways like NF-κB could be a promising strategy to control inflammatory symptoms in RA (14, 27). Our results also highlighted that diminution of NF-κB following oral administration of probiotics revealed that probiotics inhibits upstream of the NF-κB expression and subsequently can reduce inflammatory pain. Our results illustrated that NF-κB as a result of oral administration of probiotics with doses of 1/500 and 1/1000 unlike 1/250 in the inflammatory phase of arthritic inflammation induced by CFA adjuvant was reduced, with a decrease in edema and hyperalgesia subsequently. In this study, it seems that diminution of the NF-κB expression can be considered as an anti-inflammatory mechanism due to oral administration of probiotics.
In conclusion, the present study shows that oral administration of probiotics can significantly reduce the serum levels of TNF-α and IL-1β and NF-κB, which is aligned with a significant reduction in hyperalgesia and paw edema during the acute phases of CFA-induced inflammation in male rats. However, further studies are needed to evaluate the effect of probiotics on anti-inflammatory cytokines and other pro-inflammatory cytokines and intracellular signaling pathways in acute and chronic phases of inflammatory pain, which can be considered as a limitation of this study because of its design in this part of our research.