1. Background
Phosphorus is a critical element causing vascular calcification and the deposition of minerals on the walls of arteries and veins in patients with renal failure. Accordingly, hyperphosphatemia as a significant pathophysiology accounts for high mortality risk in chronic kidney disease (CKD) patients (1).
Limiting phosphate intake is not sufficient to manage hyperphosphatemia in pre-dialysis CKD patients. Such patients require additional phosphate binders for the treatment of hyperphosphatemia. The frequently used phosphate binders include those encompassing calcium, such as calcium carbonate and calcium acetate, and calcium-free ones such as aluminium hydroxide, sevelamer, nicotinic acid, magnesium hydroxide, and others. Calcium-based phosphate binders are frequently used; however, they increase the risk of hypercalcemia (2). Although sevelamer is costlier than calcium-based binders, its main advantage is that it is a metal- and calcium-free, nonabsorbable phosphate binder with a low risk of hypercalcemia, bone disease, and parathyroid suppression. It improves the quality of life and decreases mortality (3-5). In addition to reducing phosphorus absorption, other pleiotropic effects of this sevelamer, including lipid-lowering effect, reduced accumulation of advanced glycation end products (AGEs), and amelioration of metabolic acidosis in pre-dialysis CKD have also been reported (6).
Comparatively, calcium-containing phosphate binders lead to the progression of aortic vascular calcification and coronary artery calcification (7). Several studies have documented improved survival rates in CKD patients using sevelamer over calcium-based phosphate binders (8-10).
In India, there is no epidemiological data on the magnitude of vascular calcification in patients with pre- end-stage renal disease (ESRD) (CKD stages 4 and 5). This open-label comparative study was conducted to compare the effects of sevelamer carbonate and calcium acetate on aortic vascular calcification.
2. Objectives
This study aimed to compare the effects of calcium acetate and sevelamer carbonate on the progression of aortic vascular calcification in patients with CKD stages 4 and 5.
3. Methods
A clinical study with the prospective and randomized design was conducted at the centre-PGIMER and Dr. Ram Manohar Lohia Hospital, New Delhi. The study lasted for 18 months. Individual patients were assessed at baseline, 6 months, and 12 months. The study included ambulatory patients of both genders aged above 18 years. The included patients were diagnosed with stage 4 or 5 CKD, hyperphosphatemia (for stage 4 CKD - serum phosphorus level > 4.6 mg/dL, for stage 5 CKD - serum phosphorus level > 5.5 mg/dL). We also excluded patients with symptomatic coronary artery disease or arrhythmia, those on dialysis (peritoneal dialysis or hemodialysis), as well as those with acute infections such as leukocytosis, fever, or chronic infection such as hepatitis B or C, tuberculosis, AIDS, or history of kidney transplant.
The study was performed according to the guidelines presented in the Declaration of Helsinki at the International Conference on Harmonization (ICH). The Ethics Committee of PGIMER and Dr. R.M.L. Hospital, Delhi, reviewed and approved the study protocol. Informed written consent was obtained from the participants.
Among those attending the nephrology OPD and wards of Dr. R.M.L. Hospital and PGIMER, New Delhi, one-hundred-fifty patients diagnosed with CKD stages 4 and 5 were screened for the calcification of the abdominal aorta using a digital X-ray and a multi-slice CT scan. Of the screened individuals, fifty CKD stages 4 and 5 patients with evidence of vascular calcification were selected and randomly assigned into two groups (n = 25 per group) with a computer-based randomization software. The patients who were previously taking phosphate binders were given two weeks washout period for phosphate binders. Calcium acetate was prescribed at starting dose of 667 mg TDS, and sevelamer carbonates were prescribed at starting dose of 400 mg TDS. The dose of phosphate binders was titrated at two-week intervals to attain a target serum phosphorus within a range of ≥ 2.7 - ≤ 4.6 mg/dL for stage 4 CKD patients and ≤ 5.5 mg/dL for stage 5 CKD patients. Every patient was on a phosphate diet. During the study procedure, serum calcium levels were maintained between 8.5 mg/dL and 10.5 mg/dL. For one year, the two groups were followed up to detect the effects of calcium acetate and sevelamer on vascular calcification using a multi-slice CT scan, serum biochemistry, and inflammatory markers at baseline, six months, and 12 months. Digital X-ray lumbar spine (lateral view) was performed at baseline, and Kauppila score was calculated (11). The non-contrast multi-slice CT scan of the abdominal aorta was carried out using Philips Extended Brilliance Workspace, and the aortic calcification index (ACI) was calculated using software (Figure 1).
3.1. Statistical Analysis
Data analysis was performed with the statistical package for social sciences (SPSS) software version 19. The collected data were represented as standard deviation and mean (normally distributed data), median, interquartile range, frequency, and percentage. The continuous variables were compared using student's t-test, while Fisher's exact test was used for the categorical variables. Differences between the two groups were assessed using student’s t-test or Mann-Whitney U-test. A comparative assessment of the data was performed for the three groups using ANOVA. In this study, P < 0.05 was set as the significance level.
4. Results
In the present study, the participants were randomly divided into two groups (n = 25 per group); group A received calcium acetate, and group 2 patients received sevelamer carbonate.
Table 1 presents the baseline characteristics of both groups. The mean age of the patients was 57.0 years in group A and 63.3 years in group B. The predominance of males was noticed in both groups (68% and 44%, respectively). The mean duration of CKD was more significant in group A than group B (3.2 and 4.24 years, respectively). The patients in both groups were hyperphosphatemicand had high levels of inflammatory markers, intact parathyroid hormone (iPTH); and lower vitamin D levels. There was no statistically significant difference between the two groups; however, patients in the sevelamer group were older and had higher ACI (P = 0.035) and Kauppila scores (P = 0.04), and elevated serum calcium (P = 0.04), Ca X PO4 (P = 0.006), and vitamin D. Thirteen patients (52%) in group A and 10 patients (40%) in group B used statins to control dyslipidemia at baseline; however, the difference was not significant. The patients from both groups used vitamin D analogs (cholecalciferol, calcitriol), and the difference between the two groups was not significant (Table 1).
| Baseline Parameters | Group A (n = 25) | Group B (n = 25) | P Value |
|---|---|---|---|
| Age (y) | 57 ± 10.98 | 63.32 | 0.04 |
| Gender | |||
| Male | 17 ± 68 | 11 ± 44 | NS |
| Female | 8 ± 32 | 14 ± 56 | NS |
| Diabetes mellitus | 13 (52) | 15 (60) | NS |
| Hypertension | 23 (92) | 21 (84) | NS |
| History of CVD | 10 (40) | 15 (60) | NS |
| Duration of CKD (y) | 3.2 ± 1.42 | 4.24 ± 1.92 | 0.04 |
| SBP (mmHg) | 134.68 ± 12.55 | 137.12 ± 9.03 | NS |
| DBP (mmHg) | 79.56 ± 6.25 | 82.36 ± 7.26 | NS |
| Hemoglobin (gm/dL) | 9.92 ± 1.77 | 10.612 ± 1.51 | NS |
| e-GFR (mL/min/1.73 m2) | 18.60 ± 6.74 | 18.00 ± 7.31 | NS |
| Serum albumin (gm/dL) | 3.77 ± 0.62 | 3.96 ± 0.42 | NS |
| Serum phosphate (mg/dL) | 5.96 ± 0.27 | 6 ± 0.26 | NS |
| Albumin corrected serum calcium (mg/dL) | 9.16 ± 0.43 | 9.45 ± 0.58 | 0.04 |
| Calcium X phosphate (mg/dL) | 54.21 ± 2.51 | 56.67 ± 3.49 | 0.006 |
| Serum alkaline phospate (IU/mL) | 196.76 ± 106.04 | 220.92 ± 261.07 | NS |
| Total cholesterol (mg/dL) | 166.84 ± 48.22 | 162.96 ± 42.53 | NS |
| HDL-cholesterol (mg/dL) | 40.76 ± 11.69 | 42.40 ± 6.64 | NS |
| LDL-cholesterol (mg/dL) | 100 ± 33.95 | 93 ± 26.99 | NS |
| TG (mg/dL) | 151.44 ± 81.58 | 145.44 ± 75.23 | NS |
| Serum iPTH (pg/mL) | 306.55 ± 226.46 | 378.03 ± 296.23 | NS |
| Serum 25 (OH) Vit.D3 (ng/mL) | 13.39 ± 9.29 | 20.87 ± 16.47 | 0.056 |
| Serum hs-CRP (mg/L) | 5.98 ± 2.24 | 6.83 ± 2.28 | NS |
| Serum IL-6 (pg/mL) | 10.12 ± 3.66 | 11.34 ± 4.23 | NS |
| X ray Kauppila score (mean ± SE) | 5.48 ± 0.7 | 8.4 ± 0.99 | 0.02 |
| ACI (mean ± SE) | 18.85 ± 0.97 | 23.45 ± 1.89 | 0.035 |
| Statins | 13 (52) | 10 (40) | NS |
| Vitamin D (ng/mL) | 15 (60) | 20 (80) | NS |
Abbreviations: CVD, cardiovascular disease; CKD, chronic kidney disease; hs-CRP, high sensitivity C-reactive protein; IL, interleukin; DBP, diastolic blood pressure; SBP, systolic blood pressure; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; iPTH, intact parathyroid hormone; TG, triglycerides.
a Values are expressed as mean ± SD or No. (%) unless otherwise indicated.
Table 2 shows biochemical variables and ACI at baseline, six months, and one-year follow-up in group A. The mean glomerular filtration rate (e-GFR) decreased significantly during six months and one year (P < 0.05 and P < 0.001, respectively). The mean serum phosphorus level significantly declined from baseline (5.96 mg/dL) to six months (4.49 mg/dL) and one year (4.49 mg/dL) (P < 0.01). Calcium acetate-controlled serum phosphorus values were assessed according to the guidelines of the kidney disease outcomes quality initiative (KDOQI). There was no significant change in serum calcium levels (= 9.1 mg/dL). There was a slight increase in serum iPTH levels at six months (338.59 pg/mL), which subsequently decreased at one year (227.45 pg/mL) (P < 0.05). Serum vitamin D level significantly increased during one year (P < 0.01); this increase may be attributed to the patients' intake of vitamin D analogs. There was no significant change during six months and one year in mean serum alkaline phosphate and lipid levels [total cholesterol, low-density lipoprotein (LDL-C), high-density lipoprotein (HDL-C)]. There was a significant increase in mean ACI during six months (20.82) and one year (23.22) in comparison to baseline (18.85) (P < 0.01).
| Parameters | Calcium Acetate (n = 25) | P Value | ||
|---|---|---|---|---|
| Baseline | 6 Months | 1 Year | ||
| e-GFR (mL/min/1.73 m2) | 18.6 ± 6.74 | 17.28 ± 5.93 | 14.56 ± 5.17 | < 0.05 b, < 0.001 c |
| Serum albumin (gm/dL) | 3.77 ± 0.62 | 3.76 ± 0.52 | 3.69 ± 0.44 | NS |
| Serum PO4 (mg/dL) | 5.96 ± 0.27 | 4.49 ± 0.66 | 4.49 ± 0.75 | < 0.01 b, c |
| Albumin corrected serum calcium (mg/dL) | 9.16 ± 0.43 | 9.16 ± 0.32 | 9.15 ± 0.27 | NS |
| Calcium X phosphate (mg/dL) | 54.21 ± 2.51 | 41.1 ± 6.04 | 41.02 ± 6.83 | < 0.01 b, c |
| Serum alkaline phosphate (IU/mL) | 196.76 ± 106.04 | 182.13 ± 128.92 | 175.96 ± 156.11 | NS |
| Total cholesterol (mg/dL) | 166.84 ± 48.22 | 161.02 ± 39.83 | 154 ± 46.64 | NS |
| HDL-cholesterol (mg/dL) | 40.76 ± 11.69 | 40.73 ± 11.50 | 38.88 ± 9.95 | NS |
| LDL-cholesterol (mg/dL) | 100 ± 33.95 | 100.86 ± 27.75 | 93.99 ± 30.37 | NS |
| TG (mg/dL) | 151.44 ± 81.58 | 151.16 ± 95.15 | 166.80 ± 115.96 | NS |
| Serum iPTH (pg/mL) | 306.55 ± 226.46 | 338.59 ± 253.68 | 227.45 ± 136.48 | < 0.05 d |
| Serum 25 (OH) Vit.D3 (ng/mL) | 13.39 ± 9.29 | 16.1 ± 9.69 | 24.84 ± 11.73 | < 0.01 c |
| Serum hs-CRP (mg/L) | 5.98 ± 2.24 | 6.73 ± 2.36 | 7.13 ± 3.13 | < 0.01 c |
| Serum IL-6 (pg/mL) | 10.12 ± 3.66 | 10.46 ± 4.26 | 11.25 ± 4.66 | < 0.01 c |
| ACI (mean ± SE) | 18.85 ± 0.97 | 20.82 ± 1.06 | 23.22 ± 1.19 | < 0.01 b, c, d |
Abbreviations: e-GFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglyceride; iPTH, intact parathyroid hormone; hs-CRP, high sensitivity C-reactive protein; IL, interleukin; ACI, aortic calcification index.
a Values are expressed as mean ± SD unless otherwise indicated.
b Comparison between baseline and 6-months.
c A comparison between baseline and one year.
d A comparison between six months and one year.
Table 3 presents biochemical variables and ACI at baseline, six months, and one-year follow-up in group B. The mean serum phosphorus level at baseline was 6.00 mg/dL. Similar to group A, sevelamer-regulated serum phosphorus values were according to the KDOQI guidelines. Serum phosphate, calcium phosphate, serum alkaline phosphate, and e-GFR significantly decreased from baseline to 1 year in group B. The mean serum calcium level in group B decreased significantly in one year from baseline (9.45 vs. 9.19 mg/dL; P < 0.01).
| Parameters | Sevelamer (n = 25) | P Value | ||
|---|---|---|---|---|
| Baseline | 6 Months | 1 Year | ||
| e-GFR (mL/min/1.73 m2) | 18 ± 7.31 | 17.32 ± 7.81 | 15.48 ± 6.85 | < 0.01 c |
| Serum albumin (gm/dL) | 3.96 ± 0.42 | 3.93 ± 0.32 | 4.03 ± 0.40 | NS |
| Serum PO4 (mg/dL) | 6 ± 0.26 | 4.43 ± 0.58 | 4.45 ± 0.52 | < 0.01 b, c |
| Albumin corrected serum Calcium (mg/dL) | 9.45 ± 0.57 | 9.46 ± 0.59 | 9.19 ± 0.39 | < 0.01 d |
| Calcium X phosphate (mg/dL) | 56.67 ± 3.49 | 41.97 ± 6.29 | 40.84 ± 4.42 | < 0.01 b, c |
| Serum alkaline phosphate (IU/mL) | 220.92 ± 261.07 | 175.28 ± 146.85 | 141.84 ± 91.89 | < 0.05 c |
| Total cholesterol (mg/dL) | 162.96 ± 42.53 | 157.76 ± 49.04 | 137.44 ± 36.70 | < 0.01 c, d |
| HDL-cholesterol (mg/dL) | 42.40 ± 6.64 | 43.98 ± 7.16 | 43.38 ± 6.73 | NS |
| LDL-cholesterol (mg/dL) | 93 ± 26.99 | 91.28 ± 26.32 | 76.20 ± 21.59 | < 0.01 c, d |
| TG (mg/dL) | 145.44 ± 75.23 | 139.16 ± 65.88 | 118.60 ± 51.8 | < 0.01 c |
| Serum iPTH (pg/mL) | 378.04 ± 296.23 | 314.02 ± 228.63 | 221.60 ± 137.61 | < 0.01 c |
| Serum 25 (OH) Vit.D3 (ng/mL) | 20.81 ± 16.47 | 18.98 ± 11.56 | 22.79 ± 9.37 | NS |
| Serum hs-CRP (mg/L) | 6.82 ± 2.27 | 4.88 ± 1.83 | 3.69 ± 1.59 | < 0.01 b, c |
| Serum IL-6 (pg/mL) | 11.34 ± 4.23 | 10.65 ± 4.51 | 8.06 ± 3.71 | < 0.01 c, d |
| ACI (mean ± SE) | 23.45 ± 1.89 | 23.46 ± 1.89 | 23.48 ± 1.88 | NS |
Abbreviations: e-GFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglyceride; iPTH, intact parathyroid hormone; hs-CRP, high sensitivity C-reactive protein; IL, interleukin; ACI, aortic calcification index.
a Values are expressed as mean ± SD unless otherwise indicated.
b Comparison between baseline and 6 months.
c Comparison between baseline and one year.
d Comparison between six months and one year.
In another group, mean total cholesterol and LDL-C significantly decreased one year from baseline and six months (P < 0.01). The mean triglyceride and serum iPTH levels significantly decreased from baseline to one year (145.44 vs. 118.60 mg/dL and 378.04 vs. 221.60 pg/mL; P < 0.01). The high sensitivity of C-reactive protein (hs-CRP) and interleukin-6 (IL-6), the inflammatory markers of atherosclerosis, significantly decreased at six months and one year from baseline in group B (P < 0.01). However, no significant change was observed in mean ACI from baseline to six months and one year in group B (P > 0.05).
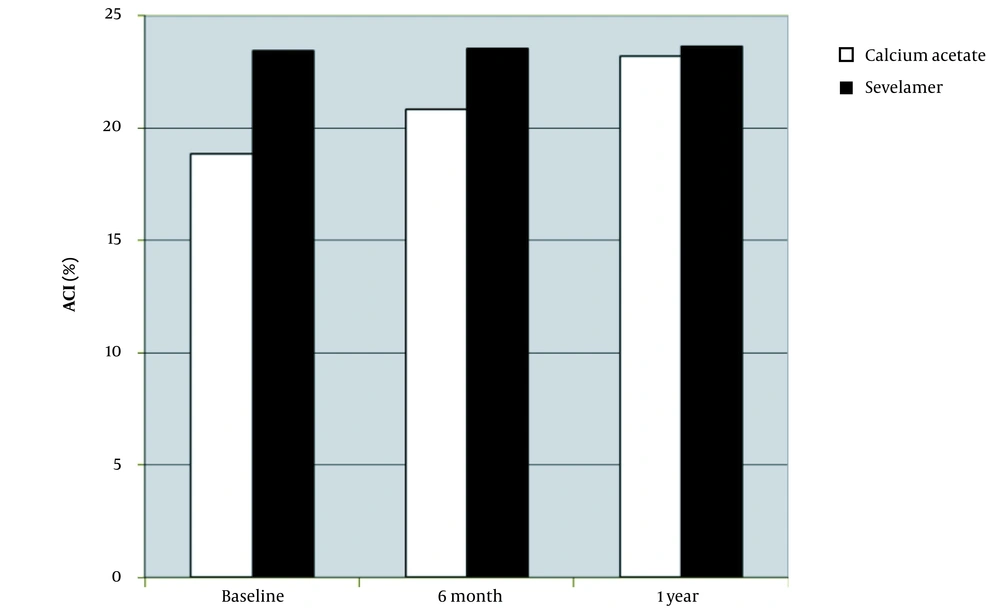
The mean ACI was significantly higher in the sevelamer group than in the calcium acetate group at baseline (23.453 ± 1.891% vs. 18.848 ± 0.972%; P = 0.035). After six months (23.46 ± 1.889% vs 20.824 ± 1.062%; P = NS) and one year (23.483 ± 1.887% vs 23.216 ± 1.188%; P = NS), the mean ACI in both calcium acetate and sevelamer groups were comparable (Figure 2). In the calcium acetate group, the mean ACI increased significantly at six months and one year (P < 0.01); however, no significant increase was observed in the sevelamer group. Accordingly, sevelamer was more effective in retarding the progression of vascular calcification of the aorta in patients with CKD stages 4 and 5.
5. Discussion
Calcium-based binders are cost-effective and exhibit disadvantages such as hypercalcemia and progressive vascular calcification, especially in hemodialysis patients; therefore, this study used a non-calcium-based binder sevelamer, which is demonstrated in literature with improved outcomes. The present study compared the effects of calcium, and non-calcium phosphate binders (calcium acetate vs. sevelamer carbonate) on the progression of aortic calcification in patients with CKD stages 4 and 5.
Russo et al. (12) showed that higher serum phosphorus levels were associated with increased coronary arterial calcification, and adverse cardiac events were more prevalent in patients with high coronary artery calcium (CAC) scores.
The present study revealed that sevelamer carbonate had greater effectivity and consistency in delaying the progression of aortic vascular calcification as compared to calcium acetate in patients with CKD stages 4 and 5. Sevelamer carbonate decreased serum calcium-phosphate product levels effectively compared to calcium acetate. Total cholesterol, LDL-C, mean triglyceride, and serum iPTH also decreased from baseline to one year in the sevelamer group. In the sevelamer group, the ACI did not change significantly; however, it progressed significantly in the calcium acetate group. Inflammatory markers such as IL-6 and hs-CRP, as the indicative of atherosclerosis, significantly decreased by sevelamer, whereas the inflammatory markers increased significantly in the calcium acetate group.
The previous studies comparing the effects of calcium-containing phosphate binders and sevelamer on vascular calcification have mainly encompassed dialysis patients. Moreover, the literature on pre-dialysis patients is not rich.
Block et al.’s study on patients with a mean age of 66 years (n = 30), suggested that calcium phosphate binders in moderate-to-advanced CKD patients led to a significant decrease in serum phosphorus levels and also reduced the development of secondary hyperparathyroidism; however, it enhanced vascular calcification (13). Chertow et al. (14) showed that both coronary artery and aortic calcification progressed significantly in hemodialysis patients treated with calcium-containing phosphate binder; however, it was not observed in those treated with sevelamer. Russo et al. compared the effects of calcium carbonate and sevelamer on CAC in pre-dialysis CKD stages 3 - 5 patients. They concluded that sevelamer had a beneficial effect on lipid profile and slowed down the progression of CAC (15).
The baseline ACI was remarkedly higher in patients treated with sevelamer than those in the calcium acetate group; however, there was no significant progress in vascular calcification in the sevelamer-treated patients. Previous studies have documented that higher baseline calcification scores are associated with a more significant progression (7, 14).
Raggi et al. (16) reported that, without treatment with phosphate binders, CAC progressed more significantly, which was associated with adverse cardiac outcomes in pre-dialysis patients. Despite its normal serum concentration, serum phosphorus might be involved in the progression of vascular calcification. This hypothesis was supported by Kestenbaum et al. (17), who reported lower mortality risk among individuals with lower serum phosphate levels. Ketteler et al. (18) showed the high effectivity and well-tolerability of sevelamer carbonate in regulating serum phosphorous levels in hyperphosphatemia pre-dialysis CKD patients.
To retard the vascular calcification process, it is essential to achieve low or normal serum phosphorus concentration. Moreover, calcium, PTH, and the calcium-phosphorus product are of paramount importance in slowing down the calcification process. It is worth noting that both sevelamer and calcium acetate Ca X PO4 product, and iPTH values were as recommended by KDOQI; however, aortic vascular calcification was significantly higher in the calcium acetate group. In contrast, the increase was not significant in the sevelamer group, indicating that the vascular calcification process is multi-factorial, and the beneficial effect of sevelamer on vascular calcification is due to its additional pleiotropic properties (9).
Long-term exposure to calcium-based binder therapy leads to "calcium load," which may promote the development and progression of vascular calcification and adynamic bone disease. In patients treated with sevelamer, baseline serum calcium, and vitamin D levels were remarkably higher. This finding is consistent with the high baseline calcification scores in sevelamer-treated patients. The patients in both groups frequently used vitamin D analogues, and there was no significant difference in this regard. This study showed a slight decrease in serum calcium levels at one year (9.45 vs. 9.19) as well as no hypocalcemia episode in patients assigned to the sevelamer group. There was no significant change in serum calcium levels in patients assigned to the calcium acetate group with the absence of hypercalcemic episodes. Hypercalcemia episodes were reported to be rare in patients treated with sevelamer (19). Block et al. (7) and Chertow et al. (14) suggested that the potential features of sevelamer responsible for slowing down the progression of vascular and valvular calcification encompassed fewer episodes of hypercalcemia and improved lipid profile.
CKD is associated with abnormalities in lipoprotein metabolism, an increase in serum triglycerides, LDL-C, and a decrease in serum HDL-C (20). Dyslipidemia in patients with CKD contribute to atheromatous calcification (21). Sevelamer shows lipid-lowering features, decreases LDL-C and LDL/HDL ratio, and raises HDL-C in dialysis patients (7, 14, 19). In the sevelamer group, the mean concentration decreased by 15% for total cholesterol, 18% for triglycerides, and 18% for LDL. The patients in the sevelamer group required statins less frequently to control dyslipidemia than the calcium acetate group. During 1-year follow-up,the statins were withdrawn in three (out of 10) patients in the sevelamer group, while no patient stopped statins in the calcium acetate group. According to the calcium acetate renagel evaluation-2 study, CKD patients on hemodialysis treated with calcium-containing phosphate binders or sevelamer experienced its effects on vascular calcification with intensive lipid-lowering (22). Accordingly, lipid-lowering therapy may favourably affect progressive vascular calcification.
Inflammatory markers have been associated with increased mortality and vascular calcification in CKD patients on dialysis due to their involvement in the initiation and progression of atherosclerosis and vascular calcification (23-25). In the present study, the inflammatory markers of atherosclerosis (hs-CRP, IL-6) decreased significantly from their baseline values at six months and one year in sevelamer-treated patients. Yamada et al. (26) documented the anti-inflammatory features of sevelamer in the CKD patients on hemodialysis. Nguyen-Khoa et al. hypothesized that sevelamer decreased vascular inflammation due to its antioxidant effect (27). In the present study, the beneficial effect of sevelamer on the progression of abdominal aortic calcification may be associated with its anti-inflammatory property.
Acidosis is common in patients with advanced CKD, who are not receiving dialysis. Acidosis has a negative effect on bone metabolism, inflammation, increased malnutrition, and atherosclerosis. Ketteler et al. claimed that metabolic acidosis could be corrected by increasing serum bicarbonate and sevelamer carbonate (18).
The limitations of this study were the inclusion of a relatively small number of CKD stage 4 and 5 patients, which is difficult to generalize in a large group of patients. Larger prospective multicentric trials should be conducted in pre-ESRD patients to confirm the present study results.
5.1. Conclusions
The prevalence of vascular (abdominal aortic) calcification was 75% patients with CKD in stages 4 and 5 (69.5% in CKD stage 4 and 85.72% in CKD stage 5). Abdominal aortic calcification increased significantly in calcium acetate-treated patients during six months and one year; however, the increase was not significant in the sevelamer group. In patients with CKD stages 4 and 5, sevelamer demonstrates higher efficacy and consistency in halting aortic vascular calcification progression than calcium acetate. The beneficial effect of sevelamer in retarding vascular calcification progression beyond its phosphate-binding property may be associated with its lipid-lowering and anti-inflammatory properties.