1. Context
Coronaviruses (CoVs) are single-stranded RNA viruses with genomes ranging from 26 to 32 kb. CoVs belong to the Coronaviridae family and the Orthocoronavirinae subfamily. Based on the different protein sequences, the Orthocoronavirinae are classified into the following genera: Alpha, Beta, Gamma, and Delta. It has been identified that Coronaviruses could be potentially pathogenic to different species of animals and humans (1). In recent years, several types of coronavirus, including the severe acute respiratory syndrome (SARS) epidemic in China the Middle East Respiratory Syndrome Coronavirus (MERS-CoV) epidemic in Saudi Arabia, caused pandemics throughout the world (2). The new CoV virus that emerged at the end of 2019 in Wuhan, China, has been named SARS-CoV-2 (Severe Acute Respiratory Syndrome Coronavirus 2) by the World Health Organization (WHO). The disease caused by SARS-CoV-2 was called Coronavirus disease 2019 (COVID-19). SARS-CoV-2 belongs to the Betacoronavirus genus. Although it is quite similar to SARS-COV (2), it is highly pathogenic, transmissible, and causes acute respiratory illness. SARS-CoV-2 infects host cells directly via a similar receptor for SARS-CoV and results in pathophysiological properties (3). Viral transmission mainly occurs through direct, indirect, or close contact with infected people or infected samples, droplets, or airborne. Symptoms may include fever, chills, cough, fatigue, and difficulty breathing (2). COVID-19 could be asymptomatic or have mild symptoms such as the flu, severe respiratory failure, septic shock, multi-organ failure, and death in extreme cases. Older age, diabetes, cardiovascular and respiratory disease, and cancer were associated with a more severe SARS-CoV-2 (4). RNA of the SARS-CoV-2 virus is located in a nucleocapsid surrounded by a viral envelope, and this envelope is composed of various proteins such as membrane proteins and spike glycoproteins. Electron microscopic studies of these spikes revealed that the appearance of crowns on the virus surface is essential for the onset of infection in humans (5). SARS-CoV-2 promotes vital organ (such as the lungs and kidneys) dysfunction directly via binding to its receptors (6). Due to the epidemic of COVID-19 and its effects on multiple organs, there is a growing concern worldwide about its impact on reproductive organs and reproductive care. Hence, it is necessary to understand possible mechanisms by which SARS-CoV-2 can damage the reproductive system and cause infertility. The current review summarizes the current knowledge of the effects of the SARS CoV-2 virus on the human reproductive system.
2. Evidence Acquisition
This study reviews articles published on the COVID-19 and its relevance to the human reproductive system with the following keywords of SARS-CoV-2, COVID-19, human reproductive system, testis, ovary, semen, using PubMed, Science-Direct, Google Scholar, and Scopus databases from 2019 to 2021.
3. Results
3.1. Specific Receptors for SARS-CoV-2 Virus
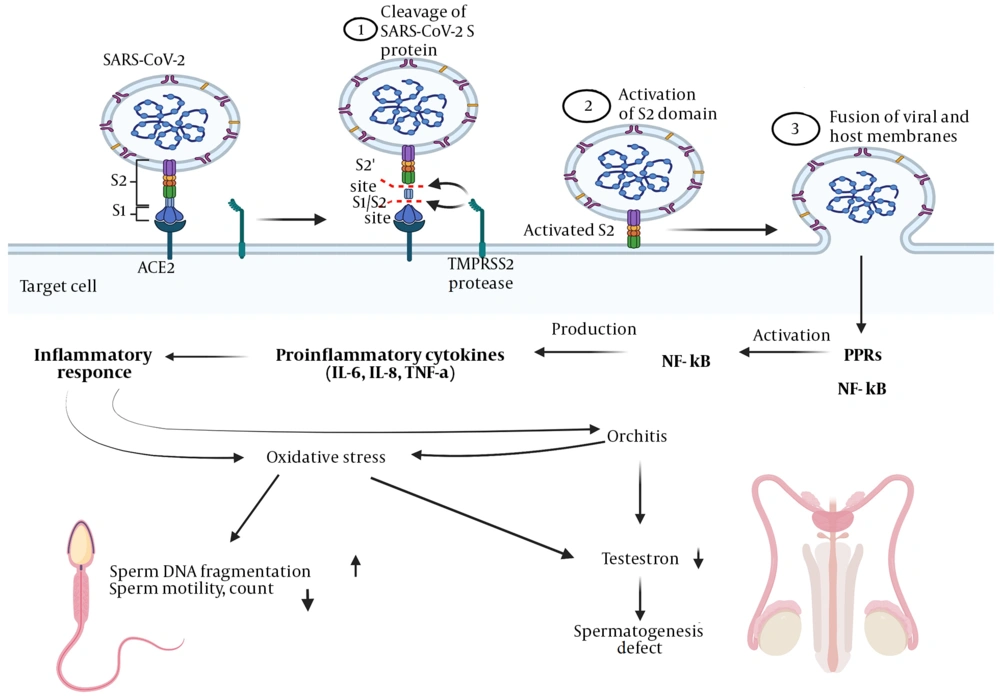
It is believed that the SARS-CoV-2 virus enters host cells via the angiotensin-converting enzyme 2 (ACE2) receptor. As ACE2 is present in approximately all human organs such as the lung, kidney, liver, heart, and testes, cells with a high level of ACE2 expression might be the possible target of the virus. ACE2 is a transmembrane metallopeptidase with a high similarity score to classical ACE. ACE isoforms are part of the renin-angiotensin-aldosterone system (RAAS), which performs vital functions in regulating blood pressure and fluid-electrolyte balance (7). Although ACE2 is the primary receptor for the virus, it is not sufficient to cause uptake of the virus into the host cell. Transmembrane Serine Protease 2 (TMPRSS2) is another potent mediator of host cell invasion by SARS-CoV-2 and SARS-CoV viruses. This important membrane-anchored protein facilitates virus entry through cleavage of the spike protein of SARS-CoV-2 (8).
Furthermore, TMPRSS2 is expressed in many tissues and is known for its dominant expression in prostate epithelium and its function in prostate carcinogenesis. Given these considerations, while TMPRSS2 expression is higher than ACE2 in human tissues, they play a crucial role in virus entering the host cells. Consistent with the result of RNAseq sequencing in human tissues, simultaneous expression of TMPRSS2 and ACE2 in the lung, kidney, and heart has been shown. Hence, these organs are highly susceptible to viral infection (9). The clinical features of COVID-19 are determined by the simultaneous expression of two protein receptors, ACE2 and TMPRSS2, in infected cells (7).
3.2. The Presence of the SARS-CoV-2 Virus and Its Receptors in the Human Reproductive System
Previous studies revealed that ACE2 receptor is expressed in spermatogonia, epididymis, Sertoli, and Leydig cells (10). Furthermore, ACE2 expression in the testes is age-dependent, i.e., middle-aged men (20 to 30) have higher expression, whereas it is lower in patients over 60 years of age. It indicated that young men are more at risk of testicular damage than older men (7). It has been determined that a minor ratio of spermatogonial stem cells express both specific receptors; therefore, sperms may not be vulnerable to viral infection (11). Currently, there was little evidence regarding the direct cell infection or possible entry of SARS-CoV-2 into testes through ACE2 or other mechanisms (However, However, it should be considered that the virus could be present in semen because semen contains other cells besides sperms, including germ cells and leukocytes, which express ACE2 and TMPRSS2. On the other hand, these protein receptors are expressed simultaneously in the seminal vesicle; so the virus can exist in the semen. In addition, it was demonstrated that SARS-CoV-2-associated protein receptors are co-expressed in other male reproductive organs, such as glands (12). Research indicated that the TMPRSS2 receptor is widely distributed in prostate epithelial cells and released as a part of the proteasome in semen (13). ACE2 has not been expressed in prostate cells, despite the fact that studies suggest the virus can be transmitted in semen through prostate-derived compounds and cells (12). Different studies on the existence of the virus in semen have expressed controversial results. For instance, two studies conducted in patients with COVID-19 have reported that the virus may not be present in semen (14). Moreover, a study of a population of 38 male patients with COVID-19 showed that the SARS-CoV-2 virus was present in the semen of four of 15 patients during the acute phase of infection and two of 23 patients during the recovery phase. There is no evidence for the existence of the virus in the semen of asymptomatic male patients. Notably, semen could contain the virus and be considered one of the possible transmission routes. It would be useful to investigate the presence of the virus in semen samples from men with different degrees of disease, both symptomatic and asymptomatic (7) (Table 1).
| Studies | Sample | None of COVID-19 Patients | COVID-19 mRNA Detection |
|---|---|---|---|
| Pan et al. (2020) (14) | 12 Semen, 1Testes | 13 | - |
| Li et al. (2020) (15) | Semen | 38 | 15.8% |
| Holtmann et al. (2020) (11) | Semen | 34 | - |
| Pan et al. (2020) (14) | Semen | 34 | - |
| Kayaaslan et al. (2020) (16) | Semen | 16 | - |
| Guo et al. (2021) (17) | Semen | 23 | - |
| Gacci et al. (2021) (18) | Semen | 43 | < 1% |
| Ma et al. (2021) (19) | Semen | 34 | - |
| Ruan et al. (2021) (20) | Semen | 74 | - |
| Yang et al. (2020) (21) | Testicular tissue | 12 | 1 from 12 cases |
| Achua et al. (2021) (22) | Testicular tissue | 6 | 33% |
| Demirel et al. (2021) (23) | Follicular fluid | Case report | - |
| Fossard et al. (2021) (24) | Follicular fluid | 183 | - |
| Rajput et al. (2020) (25) | Follicular fluid | 156 | - |
| Qiu et al. (2020) (26) | Vaginal fluid | 10 | - |
| Aslan et al. (2020) (27) | Vaginal fluid | 12 | - |
| Dong et al. (2020) (28) | Vaginal fluid | one | - |
It has been established that ACE2 mRNA transcripts are abundantly expressed in fertile and postmenopausal women (29). The ACE2 expression level in oocytes is also relatively high. Thus, the ovary and the oocyte could be potential goals for COVID-19. Angiotensin II is involved in steroid secretion, follicular growth, oocyte maturation, maintenance of the corpus luteum, and the ovulation process. Angiotensin 1 - 7 stimulates estradiol and progesterone production, increases ovulation, and induces the resumption of meiosis in the egg. It should be noted that ACE2 has a role in balancing angiotensin II and angiotensin 1 - 7. ACE2 mRNA transcripts were recognized in the human and rat uterus, and the levels of its transcripts were found to be higher in epithelial cells than stromal cells during the secretory stage (30).
As mentioned above, the ability of SARS-CoV-2 to invade tissues and infect cells depends on the simultaneous expression of TMPRSS2 and ACE2; these genes are expressed in most parts of the female reproductive system, including the ovary, cumulus cells, endometrium, and even in the early stages of fetal development. The presence of these receptors at the protein level in these tissues has not been confirmed yet (31). To date, no information is available on the existence of the SARS-CoV-2 in the ova of infected women. For instance, a study was performed to identify RNA of the SARS-CoV-2 virus in the oocytes of two ovulation-stimulated women. Although their PCR test was positive on the day of oocyte collection, no viral RNA was detected in 16 oocytes from these two patients (31).
Despite many published studies, the effects of SARS-CoV-2 infection on female reproductive activity have remained in doubt. In particular, it is still unclear whether the virus can infect oocytes and thus transmit the disease to the fetus or not. A significant concern in assisted reproductive processes during the COVID-19 epidemic is the possibility of spreading SARS-CoV-2 infection through germ cells and pre-implanted embryos. According to the high levels of ACE2 in human germ cells and early embryos, it could be possible that SARS-CoV-2 has a direct effect on oocytes and embryos. With this in mind, more molecular or cellular tests are needed to assess it (32).
3.3. Possible Pathogenic Mechanisms of SARS-COV-2 Virus on the Male Reproductive System
A wide range of viruses, including immunodeficiency virus (HIV), mumps virus, influenza virus, and Zika virus (ZIKV), can invade the testes and impair reproductive function. Besides the direct impact on the testes, viruses can also affect testosterone levels or sperm production through many other agents such as inflammation, fever, and hypothalamic-pituitary-gonadal axis dysfunction (33, 34). Respiratory viral infections are generally accompanied by inflammation, cell death, and other pathophysiologic processes that contribute to oxidative stress. Overexpression of reactive oxygen species and deficiency of antioxidant mechanisms have been critical for virus replication and the progression of virus-related diseases (35). COVID-19 is determined by fever, severe pneumonia, and acute heart damage. In patients with COVID-19, elevated circulating levels of chemokines and cytokines are observed. This "cytokine storm" develops a pre-inflammatory environment that is acutely associated with severe tissue destruction causing fetal consequences in patients with COVID-19 (36). As mentioned above, ACE2 takes a prominent role in the pathogenesis of COVID-19 throughout the direct host cell damage. The SARS-COV-2 attacks the cell by binding to the ACE2 receptor (37, 38). It can be inferred that cells with a higher number of ACE2 on their surface are more vulnerable to SAR-CoV2 infection (38). Accordingly, the SARS-COV-2 virus could affect the male reproductive tissues directly and indirectly through cytokine storms and inflammation. The following are some complications in the testicles upon exposure to this virus.
3.3.1. Inflammation of the Testicle (Orchitis)
Testicular inflammation caused by the mumps virus occurs primarily in adult patients. Under these conditions, the virus invades the testicles immediately and destroys the testicular parenchyma, causing germ cell destruction, leukocyte secretion, and collagen deposition (39). In addition to HIV and mumps, other viruses such as hepatitis B and C viruses and papillomavirus cause inflammation of the testicles (40). According to studies, SARS is another viral disease that induces testicular inflammation (39). Investigations after the SARS-CoV outbreak in 2002 revealed that testicular inflammation is a side effect of this disease that can lead to impaired spermatogenesis and germ cell apoptosis, thereby affecting sperm parameters (39). Inflammation was detected in testicular autopsies of six men who had died from SARS-CoV infection. Also, histopathological signs showed that the inflammatory secretions were mainly in the Sertoli cells, seminiferous interstitium, and degraded germ cells (39). All of this implies that virus-mediated inflammatory and immunological reactions are vital in testicular damage (41) (Figure 1). In a study performed on six testicular tissues from COVID-19 patients, pathological alterations and impaired spermatogenesis were found in three of them, and microscopic evaluation revealed that SARS-CoV-2 was detected in the testes of one of the patients. There is a straight correlation between elevated ACE2 levels in the testis and impaired spermatogenesis (22). In addition, the study by Gacci et al. showed that a high percentage of COVID-19 patients had oligo-crypto-azoospermia after recovery, and the level of interleukin-8 in their semen was pathologically high, which may indicate inflammation of the male genital tract (18). The postmortem examination results of 12 testes showed pathological changes, including mild inflammation and infiltration of T lymphocytes in the testes, and SARS-CoV-2 virus was observed in 10% of these samples (21). Of the numerous studies conducted with semen from COVID-19 patients, Li et al. found the virus in 15.8% of the semen samples from these men (15). In addition, in the study by Gacci et al., only a small amount of semen of 170 COVID-19 patients contained the SARS-CoV-2 virus (18) (Table 1). It can be concluded that the SARS-CoV-2 virus can, directly and indirectly, cause testicular inflammation and lead to a dramatic decrease in testicular function.
3.3.2 Increased Levels of Oxidative Stress
It has been found that there may be a direct link between oxidative stress and the severity of many viral infections. Several studies indicate that antioxidant system depletion and overproduction of reactive oxygen species (ROS) have great significance in the SARS-CoV pathogenesis and are associated with the development and severity of respiratory disease. Studies in laboratory animals have shown increased levels of ROS and impaired antioxidant defense as a result of SARS-CoV infection (42). Based on studies, SARS-CoV-2 may lead to male reproductive dysfunction through several possible mechanisms. It has been proposed that activating sensitive oxidative pathways through inflammatory reactions provides a common pathological mechanism for degrading physiological functions in host tissues.
Oxidative stress leads to lipid peroxidation of membranes, DNA fragmentation, and apoptotic processes, which generally impair sperm quality (41, 43). Sperm cells are susceptible to ROS attack due to their small amount of cytoplasm and thereby the small number of antioxidants and rich in unsaturated fatty acids in their plasma membrane (44, 45). SARS-CoV with over-production of ROS can activate Toll-like receptor (TLR-4) and NF-κB signaling pathway, leading to a higher release of pro-inflammatory cytokines and inflammatory responses (46). Lin et al. represented that a protein called 3CL, a SARS-CoV main protease, significantly increased ROS production and is effective in 3CL-induced cell apoptosis (47). Based on recent global experience, SARS-CoV-2 appears to have a higher risk of morbidity and mortality than the typical seasonal influenza. Influenza viruses at the cellular level increase sensitive oxidant pathways and activate pathogenic mechanisms that lead to oxidative stress (35). Increased oxidative stress (41, 48) through decreased progressive sperm motility and an associated increase in sperm DNA fragmentation is one of the recognized causes of infertility in men (49). Overall, we can assume that SARS-CoV-2 is able to increase sperm DNA fragmentation by activating pathogenic pathways and affecting the sperm fertilization potential (50). In addition to the direct link between infection and oxidative stress of SARS-CoV-2, animal studies have revealed that antiviral drugs such as ribavirin (a treatment option for COVID-19) could induce oxidative stress, decrease testosterone levels, and severely disturb spermatogenesis (51, 52). A considerable decrease in sperm count was observed in ribavirin treatment (53). Moreover, sperm DNA fragmentation (SDF) was present in these patients up to 8 months after discontinuing medication (32, 54). A study on the effect of SARS-CoV-2 infection on sperm parameters and oxidative status showed that the percentage of motility and morphology and ROS level and DFI percentage improved significantly 120 days post-infection compared to 14 days post disease (55). Another investigation on semen parameters of men with SARS-CoV-2 infection compared to healthy individuals (control) showed that the percentage of sperm DNA fragmentation increased significantly, and sperm motility, viability, and concentration decreased significantly in patients compared to control (56). These studies demonstrate the effect of SARS-CoV-2 virus on increasing semen oxidative stress. A recent study has shown that sperm parameters (motility, volume, morphology) significantly decreased in men after SARS-CoV-2 infection, suggesting that coronavirus affects male fertility and alters the normal function of the male reproductive system (57).
3.3.3. Hypogonadism in Male Patients with COVID-19
One of the possible complications of SARS-CoV-2 infection is hypogonadism. There are currently conflicting reports suggesting that SARS-CoV-2 infection in men may cause acute-phase hypogonadism. A study conducted on 81 men of childbearing age with SARS-CoV-2 disorder indicated that the luteinizing hormone (LH) level was significantly increased in these men. However, testosterone and Follicle stimulator hormone (FSH) levels were significantly decreased. This raises the possibility of hypogonadism in these patients (58). Another study showed that testosterone significantly reduced in men after SARS-CoV-2 infection (57) (We We could not conclude that these changes resulted from the direct or indirect effect of SARS-CoV-2 disease on the testis (58). According to the reports, the first stage required to initiate SARS-CoV-2 infection occurs primarily through TMPRSS2. TMPRSS2 separates the ACE2 receptor from the cell surface to relieve the virus from entering the host cell (59). According to human research, activation of androgen receptors is required to initiate transcription of the TMPRSS2 gene. Since ACE2 and androgen receptor genes are located on the X chromosome, increased X-linked inheritance of genetic polymorphisms and, consequently, endogenous androgenic actions could be a probable mechanism to explain male susceptibility to SARS-CoV-2 infection (60). Various studies in humans and animals show that hypogonadism is mainly associated with elevated inflammatory cytokines such as IL-6, IL-1β, and TNF-α (61, 62), mainly the primary inflammatory mediators in the pathogenesis of SARS- CoV-2 virus. However, a severe inflammatory disease such as COVID-19, may suppress hypothalamic-pituitary-testicular (HPT) axis activity causing a decrease in LH, FSH and testosterone. Although the study on 81 male patients with COVID-19 was inconsistent with this theoretical perception, the serum testosterone level was lower, and the LH level was higher in this study (58). These findings could indicate the direct effects of the SARS-CoV-2 virus on the testis, not the HPG (Hypothalamic-pituitary-gonadal) axis. Therefore, the relationship between SARS-CoV-2 infection and sex hormone fluctuations in men needs further research.
3.4. Possible Mechanisms of SARS-CoV-2 Pathogenesis in the Female Reproductive System
Since there is not much information about the sexual transmission of SARS-CoV-2, more research is needed on the presence of the SARS- CoV-2 virus in semen and follicular fluid, and vaginal fluid. Qiu et al. performed a study to discover the existence of SARS-CoV-2 in the vaginal fluid of 10 women with COVID-19, yet no virus was observed in their examination. Hence, only postmenopausal women were examined, and their vaginal swabs were evaluated 17 days or more after SARS-CoV-2 infection (26). As shown in Table 1, based on the studies performed to date on follicular fluid and vaginal fluid, no evidence of COVID-19 was found in these samples. Furthermore, one study in nine patients showed that SARS-CoV-2 infection did not affect the biomarker of oocyte quality and the concentration of sex-related hormones and follicular function (63). The prevalence of the virus in the lower genital tract and genital secretions may be reduced due to low COVID-19 viremia.
Moreover, analysis of the expression of SARS-CoV-2 associated proteins revealed that they are not present in female genitalia, suggesting that female reproductive organs may not be among the targets of the virus (7). Besides, according to other research, changes in the immunological environment of the female reproductive system could have a significant impact on the process of embryo implantation (64). Thus, due to the cytokine storm of COVID-19, the process of recurrent miscarriages can be possible. It seems necessary to study the effects of SARS-CoV-2 on embryo implantation during in vitro fertilization (IVF) cycles or abortion.
3.5. Gender Differences in Patients with COVID-19
Recently, various studies have declared that gender differences in the incidence of COVID-19 are slightly prominent. Men seem to suffer more from the clinical consequences and have a higher death rate from SARS-COV-2 infection than women. For example, gender-specific variations could be associated with steroids and X-linked genes. However, prior research generally confirms that men and women vary in innate and adaptive immune reactions (61). The immune-related genes on the X chromosome reduce the inflammatory response.
Additionally, women produce higher antibodies that stay in the bloodstream for extended periods, and more significant activity of immune cells is observed in women rather than in men. This is due to TLR7 gene expression and interferon-gamma (IFN-γ) production. TLR7 is expressed more in women and has two alleles. Thus, women's immune response is more robust in comparison to men. Genetic and hormonal differences may increase men's sensitivity (7). According to prior investigations on male fertility, testis approximately contains the highest levels of ACE2 mRNA among other tissues in the body (38). ACE2 mRNA is expressed in seminiferous ducts, spermatogonia, Leydig cells, and Sertoli cells. According to a study by La Vignera et al., androgens improve ACE2 expression, illustrating why men are more susceptible to this virus infection. In addition, less expression of ACE2 was detected in men with prostate cancer who were treated with androgen deprivation therapy (ADT). Therefore, ADT could prevent the penetration of SARS-CoV-2 (62). Various literature suggests that androgens also affect the TMPRSS2 overexpression, which is necessary for the SARS-COV-2 to infect the host (62, 65).
3.6. Programs and Recommendations for IVF Laboratories During COVID-19 Pandemic
Based on information on COVID-19 transmission through semen, appropriate actions should be taken for semen collection in the IVF laboratory to avoid virus transmission. Before any experiment, one of the necessary tests is to take the COVID-19 test for men who refer to infertility treatment. Then only patients whose test results are negative should be allowed to continue their infertility treatment. In addition to patient control, it is necessary to take caution during the preparation of sperm to reduce the possibility of transmission. It is recommended that semen collection and analysis be performed using the method used for samples from HIV-positive individuals to reduce the risk of possible transmission in assisted reproductive techniques (ART) (66). Also, in the case of semen freezing, a separate tank should assign for COVID-19 positive men. Since the SARS-CoV-2 virus is stored in liquid nitrogen and maintains its pathogenic properties, assessing the presence of the viruses in the semen is essential before freezing (67). If possible, exceptional facilities and equipment should be provided for patients with COVID-19, and the surfaces and equipment should be disinfected entirely after the experiment. In addition, proper filtration systems for air conditioning is needed in the andrology laboratories.
Moreover, during the pandemic, prevent the transport of frozen samples between medical centers. Everyone should avoid expelling follicular fluid without closing the lid when working with follicular fluid. Based on the latest updates from the Centers for Disease Control in the United States and other countries in Europe, SARS-CoV-2 infection has no detrimental effect on pregnancy. Furthermore, some medications used to treat COVID-19 in pregnancy are not recommended. Frozen Embryo Transfer is one of the appropriate options for women who cannot delay their ovarian stimulation, such as those seeking cancer treatment, having poor ovarian reserve, or old age women. Any risk of the spreading viral infection in the gametes or embryos in the IVF laboratory could be minimized if repeated washing protocols are followed (68).
3.7. Recommendations About Prevention of Reproductive System Dysfunction
Investigations show the presence of SARS-CoV-2 in semen samples (15) and its impact on sperm parameters (56, 57). There are concerns about subfertility or infertility due to severe COVID-19 (69). We can conclude that prevention and timely treatment of the early stages of the disease can reduce its direct or indirect effects on fertility. Conventional therapies for treated COVID-19 patients include antiviral, antibiotic, steroid, and oxygen therapies (70). It is recommended to alleviate the impact of SARS-CoV-2 on fertility; testosterone levels were measured in men positive for SARS-CoV-2, and appropriate testosterone treatment is given if needed (71). Also, anti-inflammatory drugs reduce inflammation and tissue damage in COVID-19 patients (70). There is no report on the effects of COVID-19 vaccination on fertility (72). Thus, it is reasonable to assume that vaccination may have a preventive effect.
4. Conclusions
Considering the global epidemic of COVID-19, it is vital to recognize all potential transmission routes of the SARS-CoV-2 virus. So far, many viruses have been identified in human semen, and their potential of being sexually transmitted has been confirmed. Additionally, viruses capable of triggering viremia can cross the testicular barrier and survive in the male reproductive system (73). Consequently, it can be postulated that the testis is a potential purpose for the SARS-CoV-2 virus, and testicular tissue damage and infertility after SARS-CoV-2 infection might be the side effects. Invasion of SARS-CoV-2 by ACE2 receptors may result in direct testicular damage or indirectly cause impaired testicular function through inflammatory and immunological reactions.
Further clinical studies are required to test these possibilities related to the reproductive functions among male patients. There are no definitive data from the reproductive function in the male patients recovering from COVID-19. Furthermore, no study has been presented on the presence of the SARS CoV-2 virus in the various parts of the female reproductive system and its effects on female fertility yet. Hence, further studies are needed at different stages of the COVID-19 to understand the complexity of the virus and mechanisms that have a devastating effect on the world's population.