1. Introduction
Since the rise of coronavirus disease -2019 (COVID-19), a respiratory disease caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), there has been a high rate of morbidity and mortality reported all around the globe. As of March 28, 2022, 481 million cases and 6.12 million deaths have been reported by the world health organization (WHO) worldwide. However, the initiation of vaccines as a preventive measure against this disease has played a huge role in bringing the pandemic under control. The vaccines used against SARS-CoV-2 were reported to be well tolerated, and the most common short-term side effects included tenderness at the injection site, fever, fatigue, body aches, and headaches (1). Reported cases of long-term and severe reactions have been rare and among them include myocarditis, newly diagnosed or relapsed glomerulonephritis, and minimal change disease (2-5). Also, there have been few cases reports on antineutrophil cytoplasmic antibody (ANCA)-associated glomerulonephritis after receiving COVID-19 vaccines (1, 6-9).
In this study, we report a case of ANCA-associated glomerulonephritis (AAGN) in a 15-year-old patient after receiving the second dose of the BBIBP-CorV vaccine (also known as the Sinopharm COVID-19 vaccine).
2. Case Presentation
A 15-year-old man without any past medical history presented with colic-like non-positional pain of both flanks from 1 month ago. The pain was without any radiation and had no relationship with eating and defecation. Nausea, darkening of the urine color, and loss of appetite were also reported. There were no reports of hematuria, oliguria and dysuria. On further inspection, it was learned that the patient had experienced flu-like symptoms such as sneezing and rhinorrhea after receiving his second dose of the Sinopharm COVID-19 vaccine one month before admission on October 28, 2021. The first dose of the vaccine was well-tolerated. Cetirizine tabs were used twice daily for a week to subdue these signs and symptoms. The patient also reported taking beclomethasone nasal spray. The allergy history of the patient indicated an allergy to pollen from childhood. The patient did not have a past history of using drugs, drinking alcohol, and smoking.
On examination, his pulse rate (PR) was 84 bpm, blood pressure (BP) was 120/80 mmHg, temperature (T) was 36.8, and respiratory rate (RR) was 18/min. The findings of physical examination were normal. The initial laboratory results showed creatinine of 3 mg/dL, blood urea nitrogen of 31.5 mg/dL, hemoglobin of 10.8 g/dL, and hematocrit of 31.9%. Other items of hematology and biochemistry tests such as white blood cells, platelets, fasting blood sugar, sodium and potassium were within the normal range. Urine analysis reported yellow-orange color and semi turbid appearance along with 3+ proteinuria, 4+ hematuria, and white blood cell count of 8 - 10, many red blood cells, and granular casts of 0 - 1. Urine culture results were negative. Additional immunology studies showed high levels of P-ANCA (Anti-MPO) > 100 Au/mL (positive anti-MPO). The result for C-ANCA (Anti PR3) was negative. Due to these findings, AAGN was suspected. The results of the patient's echocardiogram were normal, and the ejection fraction rate was 55%. The patient was hospitalized due to a high level of creatinine.
Renal ultrasound revealed the size of the right kidney was 147 mm, and the parenchyma width was reported to be 17 mm. The size of the left kidney was 130 mm, and its parenchyma width was 13 mm. There was a reduction of differentiation of corticomedullary junction. Reports showed no signs of stones and hydronephrosis.
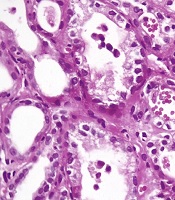
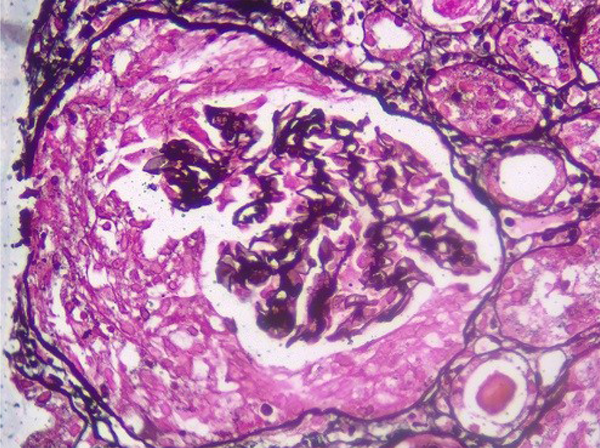
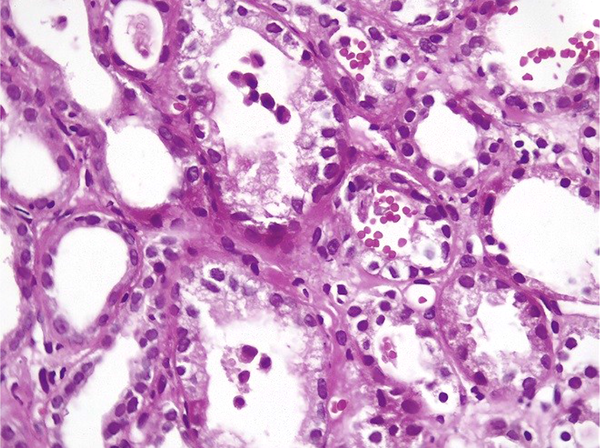
Kidney needle biopsy was performed on the patient. Surgical pathology report indicated diagnosis of pauci-immune crescentic and necrotizing glomerulonephritis and mild tubulointerstitial nephritis with foci of acute tubular injury (Figures 1 and 2).
Based on the serologic and biopsy findings, a diagnosis of AAGN was suspected.
The patient underwent medication therapy with daily 50 mg tabs of prednisolone and 500 mg CellCept tabs twice daily for three days. Creatinine level was reduced to 2.3 - 2.5 mg/dL during the hospital stay, and the patient reported a decrease in pain intensity. The laboratory results showed a creatinine level of 2.2 mg/dL at discharge. The patient was discharged with orders of oral tabs of prednisolone and CellCept. Follow-ups were done for the patient monthly. During the follow-ups, he received two doses of cefotaxime/sulbactam (Zitax) injection. Currently, the patient is still under regular follow-up. In the last follow-up, he had a stable condition with reduced urine protein and creatinine level of 1.1 mg/dL.
3. Discussion
Vaccines are being under widespread use as a defense against coronavirus disease-2019 (COVID-19), a respiratory disease caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) originated in Wuhan, China, in late December 2019, which led to a pandemic with a high mortality rate. COVID-19 vaccines went under various clinical trials and have been proven to have a high safety profile and effectiveness against the disease (10). Since the first initiation of COVID-19 vaccines, there have been reports of mild short-term side effects, including tenderness at the injection site, fever, fatigue, body aches, and headaches. These signs and symptoms might vary according to the type of the vaccine. However, reports of severe and long-term effects were rare (1). The AAGN was among the few side effects, which were rarely reported among those who received COVID-19 vaccines (1, 6, 7).
ANCA-associated vasculitis (AAV) is a group of multisystemic autoimmune diseases affecting small to medium-sized arteries by infiltration of mononuclear cells. It can involve organs such as kidneys and respiratory tracts (10). Eighty percent of all AAV cases might lead to renal manifestations, mainly as AAGN and further, end stage renal disease (ESRD). There are major subtypes of this disease, including granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), and eosinophilic granulomatosis with polyangiitis (EGPA) (11). The mentioned types have a peak incidence rate at the age of 65 to 75 years but may occur at any age, and are mainly male-dominated (12).
Myeloperoxidase (MPO) and proteinase 3 (PR3) are the major autoantigens for ANCA. They are proteins in the primary granules of neutrophils and lysosomes of monocytes. These antigens can be seen in patients with ANCA, while absent in healthy subjects. In AAV, antibodies are generated against the mentioned antigens in cytoplasmic granules of neutrophils (13). However, the exact mechanisms which lead to the initiation of AAV are not well understood. Considering previous studies, it was reported that some aspects such as infectious agents, genetic factors, environmental exposures, and various specific drugs such as hydralazine can play as triggers of autoimmunity and are involved in the pathogenesis of the disease. In our case, there was no possible known initial trigger or previous autoimmune factors that could lead to AAV (6, 13).
Previously, there have been reports of AAV after receiving seasonal influenza vaccine. In a study, an increase in production of ANCA in response to an RNA-based influenza vaccine was noted. This finding can hint at a possible association between the autoimmune response and influenza RNA vaccine (14). New-onset and relapsed AAV and glomerulonephritis have been reported in some cases after receiving COVID-19 vaccines such as Pfizer, Moderna, and AstraZeneca (1, 6-9, 15). In our case, this condition was developed one month after receiving the second dose of Sinopharm COVID-19 vaccine. This vaccine, also known as Inactivated COVID-19 (VERO CELL) vaccine, is produced in vero cells and inactivated with ß-propiolactone. The SARS-CoV-2 antigen is purified and then adsorbed with Aluminum Hydroxide to make this vaccine (16). While there were multiple reports on this occurrence in mRNA COVID-19 vaccines such as Pfizer and Moderna, reports on development of AAV after Sinopharm vaccine are few (17, 18).
Furthermore, some studies reported an increase in ANCA, leading to AAV and autoimmune reactions in patients suffering from COVID-19. These findings can further indicate the possibility of autoimmune reactions to both RNA of the SARS-CoV-2 virus (7, 8).
The temporal relationship between AAV and receiving COVID-19 vaccines could be explained by theoretical mechanisms, such as molecular mimicry, polyclonal activation, or a transient systemic proinflammatory cytokine response. However, there is no definitive mechanism explaining the pathophysiology of this phenomenon (1). Manifestations of AAGN, clarified with serology and pathology results, further indicate the presence of this condition. The occurrence of this immune reaction in a short time after receiving Sinopharm vaccine, raises the question of whether there might be a relationship between development of AAGN and receiving Sinopharm COVID-19 vaccine? However, there is no test to prove the causality of this incidence.
Considering the importance of administering COVID-19 vaccines in reducing the number of cases and the wide extent of COVID-19 vaccines being used, it is important to investigate their possible side effects. There have been rare autoimmune side effects reported to date. Our case is one of few patients in whom AAGN developed shortly after receiving the Sinopharm COVID-19 vaccine. Moreover, there was no possible trigger that could have led to this autoimmune reaction. We introduce the hypothesis that the AAV, in this case, is related to the recent Sinopharm COVID-19 vaccination. Although causality can be difficult to establish, the development of this autoimmune reaction after both natural SARS-CoV-2 infection and following other vaccinations may support our hypothesis.