1. Background
Urinary tract infection (UTI) is a common childhood infection in hospitalized and non-hospitalized children (1). Urinary tract infection is the most common bacterial infection of the urinary tract system (2), which is the most common in children after viral flu (3). This infection may be seen in the lower or upper urinary tract or both (2). Most UTIs originate from the bladder, resulting from the proliferation of pathogenic bacteria in the perineal. These bacteria migrate to the bladder to cause a bladder infection. If the bladder infection is not treated, pathogenic bacteria can reach the kidneys through the ureter and cause secondary kidney infections, such as acute pyelonephritis, which can lead to kidney scar or permanent kidney damage. In very severe cases of pyelonephritis, invading bacteria can penetrate the bloodstream by gaping in the epileptic and endothelial dams, causing bacteremia, systemic infections, sepsis, and serious and fatal complications (4).
Urinary tract infections are divided into simple and complex types. In simple UTIs, which are related to the lower urinary tract and are the most common type of UTI, there is no disorder in the physical system of the urinary tract, and none of the medical devices that remove the immune system (such as a catheter) do not play a role in creating it. Also, there is no relationship between simple UTI and anatomical and functional disorders like diabetes and pregnancy (5, 6). A significant percentage of patients of any age are re-infected after antimicrobial treatments. During the first six months after the first period of UTI, 25% of young women are re-infected with UTI (6). Also, 3% to 5% of girls and 1% of boys develop UTI during childhood. In the first year of life, boys (3.7%) are more susceptible to UTI than girls (2%). However, after the first year of life, the prevalence of this infection is significantly greater in girls (2).
The prevalence of UTI is 1.25 per 1000 live births. Symptomatic and asymptomatic UTIs are seen in 1 - 2% of school-age girls and are more common between the ages of 7 and 11, but they are rare in boys in these age ranges (7-9). Some bacteria, including Proteus, Enterococcus, Klebsiella, and Staphylococcus, can infect the urinary tract. Nevertheless, more than 80% of simple UTIs are caused by Escherichia coli (4).
Some studies have shown that Proteus is more common in boys older than one year than E. coli. Staphylococcus aureus can cause UTIs in both girls and boys. Viral infections, especially adenoviruses, can also cause UTIs (9-11). Urinary tract infection in infancy is a potentially dangerous infection that can cause permanent problems such as kidney scars, urine reflux, and kidney failure in the elderly. According to the importance of prevention, diagnosis, and treatment of UTIs, it is necessary to invest in risk factors to prevent children from UTIs, secondary kidney damage, and irreparable complications (12-16).
2. Objectives
There are several risk factors for UTIs, some of which are related to the prenatal period, pregnancy, or parental illness, while others are related to birth circumstances and the baby's life. These include gestational diabetes, maternal age, parental smoking, multiple births, prenatal care, chronic hypertension, hypertension, maternal BMI, infant sex, infant weight, duration of pregnancy, type of delivery, circumcision status of boys, type of breast milk, and history of UTI. Therefore, considering the importance of prevention, timely diagnosis, and treatment of UTIs in preventing kidney damage and proper and timely use of appropriate methods of diagnosis and treatment, it is necessary to consider the risk factors contributing to infections. Urine tract infections should be identified, and necessary measures should be taken to reduce the incidence of this disease in children. Therefore, we decided to identify the factors and risk factors affecting UTIs in infants and children to prevent, treat, and reduce possible complications in the future.
3. Methods
3.1. Study Design and Subjects
This descriptive cross-sectional study was accomplished on children's parents referred to Amir Kabir hospital in Arak. After the certification of the design and approval of the study by the Ethics Committee of Arak University of Medical Sciences, sampling was done. Educated Iranian mothers living with their husbands were entered into this research. They also had no medical restrictions on breastfeeding their infants.
Inclusion criteria were hospitalization in Amir Kabir Hospital in Arak due to UTIs, absence of congenital anomalies based on tests (VCUG and ultrasound) and examinations, not taking drugs weakening the immune system, age between one day and 12 years, and parents' consent to interview. In case of unwillingness and incomplete questionnaires, they were excluded from the study.
The sample size was calculated by the following formula. Based on a dropout rate of 10%, the minimum sample size was calculated as 100 people.
We considered a confidence interval of 0.95, test power of 80, Z1 of 1.96, and Z2 of 0.84. Based on the prevalence difference in the base studies, the p value was considered equal to 0.50. The highest number of samples was achieved. Also, d was the minimum relative abundance of each factor. It would show a statistically significant difference (P ≤ 0.2). At least 100 people participated in the present study. The objectives of the study were explained to the samples. Written consent was obtained after explaining the study's aims and information confidentiality.
3.2. Data Collection
After the approval of the research plan by the Research Council and obtaining permission from the Ethics Committee of Arak University of Medical Sciences, sampling was performed. Permission was obtained from the children's parents, and the relevant checklist was completed through interviews. The data collection tool was a valid questionnaire to gather the child's characteristics, including age, sex, type of nutrition, circumcision, and age of circumcision, as well as information about fetal birth weight and chronic maternal diseases such as diabetes and stress, blood, and residence place (rural/urban). Content validity method was used to determine the validity of the questionnaire, and the test-retest was used for reliability. Cronbach's alpha was 0.84.
3.3. Data Analysis
SPSS 22 was used for data analysis. The Kolmogorov-Smirnov test evaluated the normality of data. Independent t-test (quantitative variables), chi-square test (nominal variables), and Mann-Whitney test (non-parametric, ordinal variables) were used to describe the information obtained from descriptive and inferential statistics. In addition, logistic regression was used to evaluate the simultaneous effect of factors related to physical domestic violence. A significance level of 0.05 was considered.
4. Results
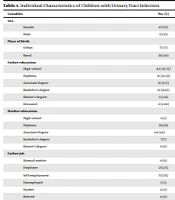
One hundred children and infants with UTIs were included in this study. Of them, 87% were female, and 71% were from urban areas. Also, 43.75% of fathers had undergraduate education, and 38% of mothers had diplomas. Besides, 55% of fathers were self-employed, and 96% of mothers were housewives. In addition, 53% owned villas (Table 1).
| Variables | No. (%) |
|---|---|
| Sex | |
| Female | 87 (87) |
| Male | 13 (13) |
| Place of birth | |
| Urban | 71 (71) |
| Rural | 29 (29) |
| Father education | |
| High school | 42 (43.75) |
| Diploma | 31 (32.29) |
| Associate degree | 9 (9.37) |
| Bachelor's degree | 11 (11.45) |
| Master's degree | 3 (3.12) |
| Deceased | 2 (2.08) |
| Mather education | |
| High school | 2 (2) |
| Diploma | 38 (38) |
| Associate degree | 44 (44) |
| Bachelor's degree | 7 (7) |
| Master's degree | 9 (9) |
| Father job | |
| Manual worker | 9 (9) |
| Employee | 25 (25) |
| Self-employment | 55 (55) |
| Unemployed | 3 (3) |
| Farmer | 4 (4) |
| Retired | 4 (4) |
| Mother job | |
| Employee | 3 (3) |
| Student | 1 (1) |
| Housewife | 96 (96) |
| Mother's smoking | |
| Smoking | 25 (26.04) |
| Non-smokers | 71 (73.95) |
| Delivery | |
| Cesarean | 62 (62) |
| NVD | 38 (38) |
| Medications used during maternal pregnancy | |
| None | 71 (71) |
| Levothyroxine | 6 (6) |
| NSAID | 3 (3) |
| Insulin | 3 (3) |
| Paroxetine Inderal; Sodium valproate; alprazolam citalopram | 1 (1) |
| Iron pills | 1 (1) |
| Diphenhydramine and acetaminophen | 1 (1) |
| Estrogen | 1 (1) |
| Injectable antibiotics | 1 (1) |
| Letizen | 1 (1) |
| Oral antibiotics | 1 (1) |
| Ranitidine | 1 (1) |
| Diclofenac suppositories | 1 (1) |
| Lung infection drug | 1 (1) |
| Acetaminophen | 1 (1) |
| Accommodation | |
| Villa | 53 |
| Apartment | 31 |
| House type | |
| Landlord | 52 |
| Tenant | 26 |
We found that 67% of mothers did not have a disease, 8% had diabetes, and 6% were hypothyroid (Table 2). In 86% of cases, the child's siblings did not have a disease. Also, 42% were the second child, and 62% of children experienced the first episode of UTI, but 27% experienced the second episode of UTI. Moreover, 37% did not take medications in the last two weeks, and 26% did not take antibiotics. Also, 55% had no medical history. Based on the results, 41% of children were aged 2-6 years, and 39% were under two years (Table 3). The mean age was 47.05 months, and the height was 92.66 cm. The head circumference was 42.85 cm, and the weight was 15.57 kg. The average duration of breastfeeding was 18.15 months. The mean duration of complementary food was 5.85 months.
| Mother Disease During Pregnancy | No. (%) |
|---|---|
| None | 67 (67) |
| Diabetic | 8 (8) |
| Hypothyroid | 6 (6) |
| Gestational diabetes | 3 (3) |
| Seizure | 1 (1) |
| Other diseases | 15 (15) |
| Variables | No. (%) |
|---|---|
| Child's sibling diseases | |
| None | 86 (86) |
| UTI | 3 (3) |
| Allergic disease | 2 (2) |
| Other diseases | 9 (9) |
| Age (y) | |
| < 2 | 39 (39) |
| 2 - 6 | 41 (41) |
| > 6 | 20 (20) |
| UTI re-admission | |
| First | 62 (62) |
| Second | 27 (27) |
| Third or more | 11 (11) |
| Two-week medical history | |
| No | 62 (62) |
| Common cold | 27 (27) |
| GI | 2 (2) |
| Other | 11 (11) |
| Two-week drug history | |
| None | 37 (37) |
| AB | 26 (26) |
| NSAID | 10 (10) |
| Vaccination | 8 (8) |
| Other | 19 (19) |
| Medical History | |
| None | 55 (55) |
| Hypothyroid | 3 (3) |
| Allergic disease | 2 (2) |
| Renal disease | 6 (6) |
| GERD | 4 (4) |
| Icterus | 6 (6) |
| Seizure | 4 (4) |
| Anemia | 3 (3) |
| Lung infections | 2 (2) |
Blood tests showed mean white blood cells of 11.82 × 1000 per microliter, red blood cells of 4.50 × 1000 per microliter, hemoglobin of 12.05 g/dL, hematocrit of 36.28%, MCV of 24.80 f Lit, MCH of 26.86 pg, platelets of 3.17 × 1000 per microliter, RDW of 13.72%, neutrophils of 57.21%, lymphocytes of 35.73%, monocytes of 2.85%, eosinophils of 1.36%, pH of 6.02, urine white blood cells of 41.15, red blood cells of 11.02, and epithelial cells of 3.55. Urinary test abnormalities were not observed in most children. Besides, 64% of children had no urine culture, and 19% had negative culture. Of them, 88% had not traveled in the last two weeks.
The growth status of children was normal in 79% of cases. Also, 64% of children had a normal ultrasound. Besides, 22% had hydronephrosis and nephrolithiasis on ultrasound, and 84% did not have antibiograms. The average thickness and size of the right kidney were 71.75 and 9.16, respectively, and the thickness and size of the printed kidney were 9.16 and 9.50, respectively.
5. Discussion
One hundred children and infants with UTIs were included in the study. Of them, 87% were female, and 71% were from urban areas. Also, 43.75% of fathers had undergraduate education, and 38% of mothers had diplomas. Besides, 55% of fathers were self-employed, and 96% of mothers were housewives. In addition, 53% owned villas. Also, 67% of mothers had no disease, while 8% were diabetic. Moreover, 87% of the siblings did not have the disease. Of them, 42% were the second child. 62% of children experienced the first episode of UTI, but 27% experienced a second episode of UTI. 37% did not take medications in the last two weeks, and 26% did not take antibiotics. 55% had no medical history. 41% of children were aged 2 - 6 years, and 39% were under two years. The mean age was 47.05, the height was 92.66 cm, the head circumference was 42.85, and the weight was 15.57 kg. The incidence of UTIs in the first year of life was equal in both girls and boys and then became more common in girls (17). Most children had no clinical symptoms. The average duration of breastfeeding was 18.15 months. Three studies reported that breastfeeding could affect the occurrence of UTI (18). Breastfeeding and normal weight may reduce the risk of UTI in childhood (19). The mean of complementary food was 5.85 months. Two studies showed that the recurrent UTI risk factors were infrequent voiding and poor fluid intake (20, 21).
The mean blood tests were 11.82 for white blood cells, 4.50 for red blood cells, 12.05 for hemoglobin, 36.28 for hematocrit, 80.24 for MCV, 26.86 for MCH, and 3.17 for platelets. It also showed RDW of 13.72, neutrophil 57.21, lymphocyte 35.73, monocyte 2.85, and eosinophil 1.36. Also, pH was 6.02, white blood cells in urine were 41.15, red blood cells were 11.02, and epithelial cells were 3.55. Urinary test abnormalities were not observed in most children. 64% of children did not have a urine culture, and 19% had negative culture. The most common organism was Escherichia coli. 62% did not have the disease in the last two weeks. 88% had not traveled in the last two weeks. 37% did not take medications in the last two weeks, and 26% did not take antibiotics. 55% did not have a history of the disease. 27% of mothers did not take medications. The growth status of children was normal in 79% of cases. 64% of children had normal ultrasounds. 22% had hydronephrosis and nephrolithiasis on ultrasound.
84% did not have an antibiogram. The average thickness and size of the right kidney were 71.75 and 9.16, respectively, and the thickness and size of the printed kidney were 9.16 and 9.50, respectively. Jahanshahifard et al. conducted a case-control study on 114 infants up to 14 years old. In this study, the prevalence of UTIs was higher in girls (77.2%). The highest prevalence of UTIs was in the age group of 0-1 years (38.6%). In this study, 79.8% of the participants in the non-infected group were breastfed. There was a significant relationship between breastfeeding and not getting a UTI (14). The results of their study are in line with our study.
Kavitha et al. in a case-control study assessed the various risk factors and profiles of UTI (16). In this study, boys that were not circumcised, recent hospitalization, recent catheterization, common toilet usage, cleaning perineum from back to front, constipation, usage of tight underclothing, diaper usage, worm infestation, neurological abnormality, voluntary withholding of urine, and reduced water intake constitute were mentioned as risk factors. They showed that recurrent infection was most often due to unresolved bacteriuria or persistent bacteriuria than reinfection and common organism causing UTI was E. coli. They also found that the risk of UTI depended on socioeconomic status and cultural habits.
The results of their study are consistent with our study. In our study, E. coli was the most common pathogen. Kizilca et al. assessed 344 children with UTIs and showed that microorganisms causing infection were ESBL-producing in 148 patients and not ESBL-producing in 196 patients. The most important cause of UTI in this study was E. coli (22). The results of our study were consistent with the study of Kizilca et al. In clinical practice, it is less likely to protect children from the first UTI.
5.1. Conclusions
More than half of the children had a normal ultrasound. Also half of them had hydronephrosis and nephrolithiasis on ultrasound. A significant percentage did not have an antibiogram. The mean thickness and size of the right kidney were 71.75 and 9.16, and the thickness and size of the left kidney were 9.16 and 9.50, respectively. Escherichia coli was the most common microorganism. It is essential to prevent, timely diagnose, and treat UTIs to prevent kidney injuries. Hence, using the presented factors to improve and make early diagnosis is necessary.