1. Background
Impaired fasting glucose (IFG), and impaired glucose tolerance (IGT) are two forms of abnormal glucose metabolism which are located between the normal glucose tolerance (NGT), and type 2 diabetes. These two disorders have significant pathophysiological effects on insulin sensitivity, and secretion as well as cardiovascular diseases (1, 2). Epidemiological studies have considered microalbuminuria (MAU) as a risk factor for atherosclerosis, coronary artery disease, and other vascular disorders in patients with type 2 diabetes, and IGT (3-5). Microalbuminuria refers to a slight increase in secretion of albumin in urine, and is a sign of progression towards nephropathy in patients with diabetes (6). It is a clue which helps us predict the occurrence of cardiovascular disorders in both patients with or without diabetes (7, 8). The risk of microalbuminuria is correlated with plasma glucose level, and the duration of hyperglycemia in patients with diabetes (9, 10). Glycemic control in these patients can prevent the development, and progression of microalbuminuria, but this issue has not been well-documented about IGT and IFG-related disorders yet (11). Some studies conducted in this regard have shown that IGT is a more important risk factor than IFG for developing microalbuminuria (5). At present, no special treatment and diagnostic measures is advised in patients with IGT and IFG.
2. Objectives
Considering that early diagnosis and control of microalbuminuria can slow its progression towards macroalbuminuria and renal failure, and also prevent cardiovascular complications, the present study was designed to investigate the probable correlation between microalbuminuria and IFG and IGT.
3. Patients and Methods
This cohort study was performed from 2009 to 2011 in university hospital of Mazandaran university of medical sciences, Sari, Iran. Exclusion criteria were as follows: overt diabetes mellitus (12), hypertension, heart failure, renal failure, previous history of proteinuria, recent urinary tract infection (UTI), and treatment with corticosteroids or spironolactone, ARBs (angiotensin receptor blockers), and ACEIs (angiotensin converting enzyme inhibitors). The study population was sequentially selected based on the results of glucose tolerance test (GTT, 75 g), and the level of FBS. FBS ≥ 100mg/dL and less than 126 was considered as IFG. IGT was defined between 140-199 mg/dL of serum glucose after two-hour oral 75 g glucose consumption. The study was approved by the local ethics committee, and informed consent was obtained from all participants.
The quantitative urine albumin-creatinine (Cr) ratio in morning spot urine samples were used for standard microalbuminuria determination. For these measurements, the automated clinical chemistry analyzer by immunotorbidometry assay with prstige24i device (auto analyzer, Japan), Pars Azmon Kit (Iran) were used. microalbuminuria was defined as 30-300 mg/g Cr (13) in two random measurements with a month interval.
The subjects with different results in these two random tests (one positive and one negative) were asked for testing the third sample. All of our cases were followed up for two years, we did not have any drop out in our patients follow, and their blood sugar and urine albumin levels were measured every 6 months. For each participant, HbA1C was requested at least two times in a two-year follow up. We used chromatography method with Bio-system Kit (Italy, CV = 5%) for this test.
The obtained data was analyzed by SPSS software. ANOVA, X2 and fisher exact tests were used for comparison of data between the groups. Correlation between blood glucose and urine albumin was evaluated by the Pearson correlation coefficient. P value less than 0.05 was considered as statistically significant.
4. Results
The mean (SD) age of the subjects was 52.5 (± 11.2), 51.4 (± 12.2) and 40.2 (± 12.0) years in the IFG, IGT and control groups respectively (P < 0.001). Of forty five subjects with normal glucose tolerance, 12 (26.7%) were male, and 33 (73.3%) were female. Thirty four (76.6%) of subjects with IFG, and 38 (84.4%) with IGT were female (P = 0.4). Basic fasting blood sugar and blood glucose 2 hours after 75 gr oral glucose in three groups (IFG, IGT and control groups) were shown in Table 1.
aAbbreviations: IFG, Impaired fasting glucose; IGT, impaired glucose tolerance; NGT, normal glucose tolerance
aAbbreviations: IFG, Impaired fasting glucose; IGT, impaired glucose tolerance; NGT, normal glucose tolerance
aAbbreviations: IFG, Impaired fasting glucose; IGT, impaired glucose tolerance; NGT, normal glucose tolerance
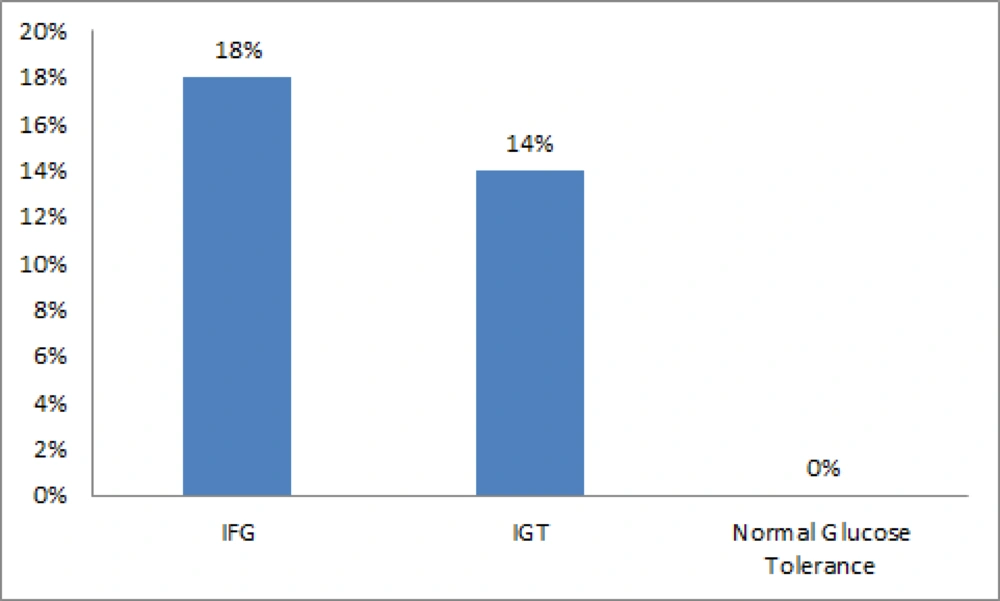
Microalbuminuria was not seen in the control group. The prevalence rate of microalbuminuria was 15.5% in prediabetic subjects (P = 0.005). The prevalence of microalbuminuria in patients with IFG or IGT was not significantly different (P = 0.4) (Figure 1).
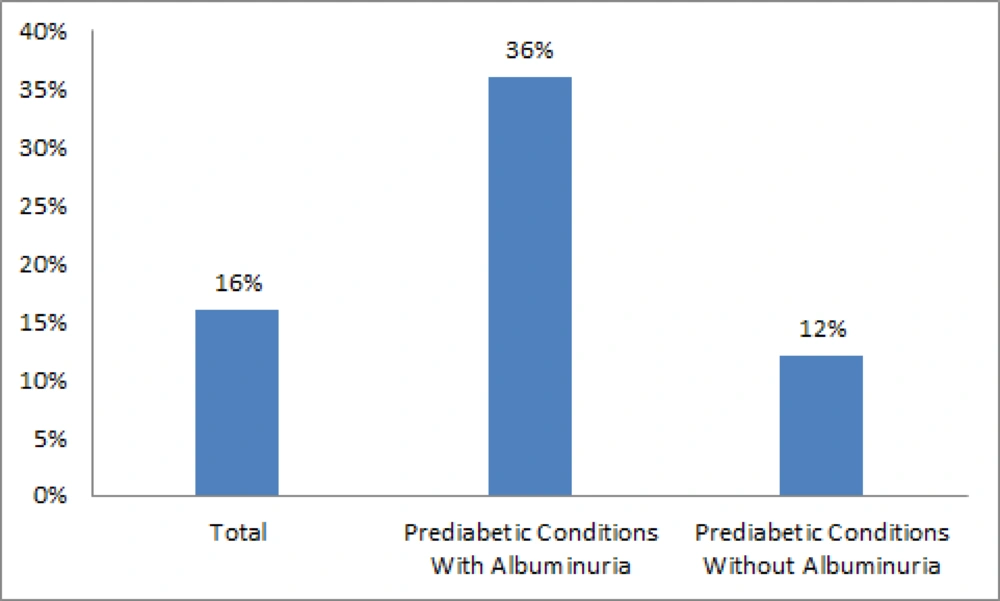
Urine albumin secretion in both samples measurement was significantly correlated with FBS (r = 0.32, P < 0.0001; r = 0.3, P < 0.001; r = 0.32, P < 0.0001), and BS2hpp (r = 0.3, P = 0.002; r = 0.3, P = 0.001). Fourteen subjects (4 in IFG group and 10 in IGT group) developed diabetes mellitus within a 2-year follow-up period (P = 0.1). [according to the ADA 2011 definition of diabetes mellitus (14)]. Prediabetic subjects with albuminuria were four times at risk of progression to diabetes mellitus than subjects without proteinuria during a 2-year follow-up (P = 0.02, Odd ratio = 4.1; CI95%, 1.13-15.1) (Figure 2). The distribution of diabetes mellitus in albuminuric subjects with IFG and IGT were 7.1% and 28.6 % respectively (P = 0.03).
5. Discussion
This study investigated the presence of microalbuminuria in patients with IFG and IGT, and compared them to the control group (NGT). The prevalence of microalbuminuria in prediabetic subjects was significantly higher than normal glucose tolerance cases, but the prevalence of microalbuminuria was not significantly different between the two groups with IFG and IGT. In Monica study on Italian subjects, the prevalence of microalbuminuria were 6.9%, 5.6%, and 4.3% in IFG, IGT and NGT groups, respectively (15). The prevalence of microalbuminuria was 8.3% in IFG, 9.9% in IGT, and 4.3% in NGT groups in Robyn study in Australia (16). The difference in prevalence reported by different studies can be attributed to the differences in population indexes such as race, laboratory techniques for urine albumin measurement, and the differences in the definition of microalbuminuria, IFG, and diabetes mellitus.
The current study revealed that the urine albumin secretion is significantly higher in the IFG and IGT groups than in the NGT group. Although the prevalence and risk of microalbuminuria was higher in IFG group than in IGT group, this difference was not statistically significant in our study. Meigs in Framingham Offspring Study reported a strong correlation between IFG and microalbuminuria (11). In Wang study the prevalence of microalbuminuria in IGT group was higher than IFG group, and IGT was introduced as the most important risk factor for microalbuminuria (5). The mechanism of albuminuria in relation with glycemia involves the glycation of basal membrane proteins, losing the selective power of membrane, glomerular hyperperfusion, and its hyperinfiltration (17). Also, it has been revealed that the relation between hyperglycemia and microalbuminuria is independent from the blood pressure (5). In our study, the cases with hypertension were excluded from the study. Fourteen subjects (10.4%) in our study developed diabetes within a 2-year follow-up were 4 in the IFG, and 10 in the IGT groups respectively. This suggests that the risk of diabetes was greater for the IGT group, whereas our study revealed more correlation between IFG and risk of microalbuminuria. The limitation of our study was that we did not repeat the glucose tolerance test during the two year follow up for diabetes mellitus diagnosis, although we measured HbA1C for this mention (14). In our study the body mass index has not been matched between the three groups; despite the fact, according to a large sample study with 20,828 cases by Thoenes, BMI had not any association with albuminuria (18).
Our study showed that the prevalence of microalbuminuria is high in prediabetes conditions such as IFG and IGT, and it correlates with the progression towards diabetes mellitus in this population. We recommend periodic evaluation of urine albumin in addition to HbA1C in patients with prediabetes conditions.