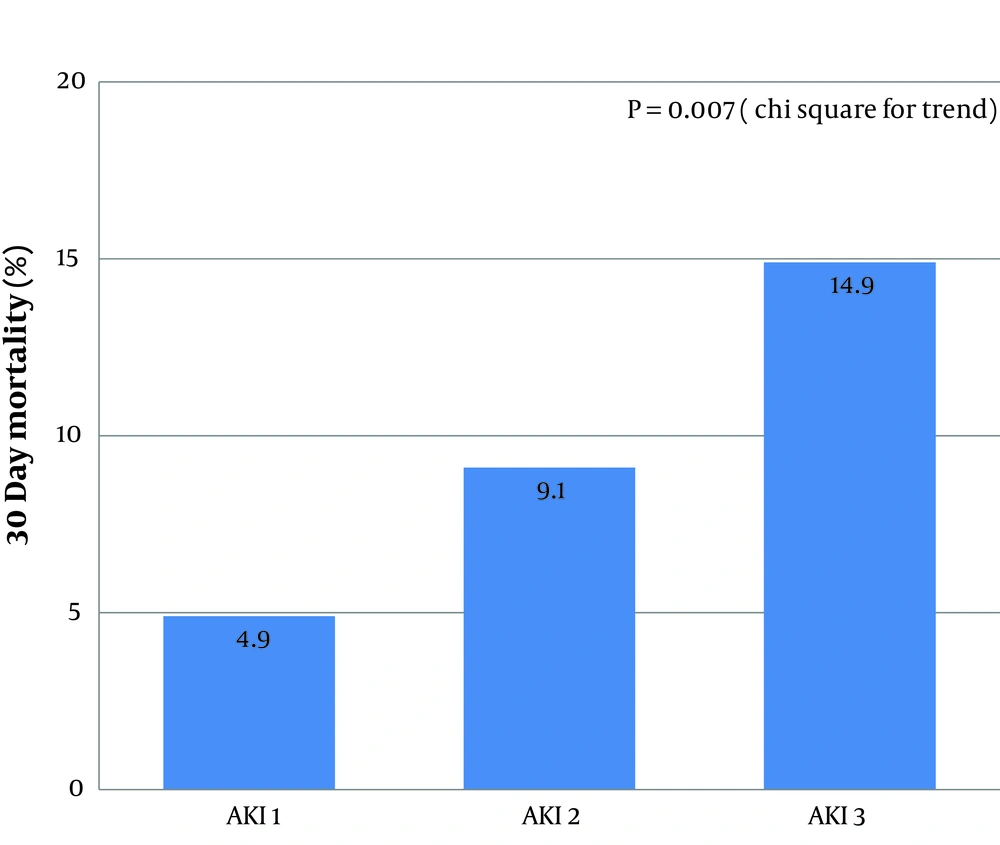
This study provides the first description of the epidemiology of AKI in a urology population, using current diagnostic criteria and including all subgroups of patients. The results confirm that AKI is common in this population and its severity remains associated with mortality, although this appears lower than in other groups. However, the rate of renal recovery was low.
The widespread acceptance of the RIFLE, AKIN and more recently the KDIGO definitions for AKI have emphasised its high incidence and extremely poor outcomes (
5-
7).
In part, a better appreciation of its high incidence stems from the recognition that even small declines in renal function are associated with increased mortality, longer hospital stays and increased healthcare costs (
10). Using current definitions, the reported incidence of AKI in general hospitalised patients varies between five and twenty percent of all admissions with overall mortality rates above 20% (
1,
2). Mortality rates increase with severity of AKI being greater than 30% in stage 3 AKI, rising to more than 45% in critical care patients requiring renal replacement therapy (
3). AKI rarely occurs in isolation and co-existing acute illness is often implicated in its aetiology as well as having a strong impact on patient outcome (
11). However, AKI is more than a simple marker of illness severity; it is increasingly appreciated that AKI has distant effects that contribute to organ dysfunction and negatively impact on overall outcomes (
12).
Although urology patients are generally accepted to be a high risk population for AKI, no previous studies have described its epidemiology in this group. Some reports describe changes in renal function in relation to a specific urological condition but few have employed current diagnostic criteria. One study used a modification of the AKIN criteria to describe the features of AKI secondary to ureteral calculi, but the authors did not report the severity of AKI or examine its effect on outcomes (
8). In another example, AKI in bilateral ureteric obstruction was defined as ≥ 33% decrease in serum creatinine after intervention (
9). The use of such alternative descriptions makes comparisons between studies very difficult.
Our results confirm that AKI is a common clinical problem facing urologists although within this population there are several distinct groups. In those undergoing elective surgery, almost half of the patients classified as AKI had undergone partial or radical nephrectomy. In these patients, the more severe stages of AKI were rare with no mortality. This good early prognosis is obviously at odds with the outcomes of AKI in other clinical situations and highlights that AKI is not a diagnosis in itself but a syndrome describing changes in renal function. Its implications therefore depend on the underlying aetiology and co-existing acute conditions (
11). Although ‘apparent AKI’ in stable patients undergoing nephrectomy may sometimes occur as a result of nephron loss, post-operative patients may develop AKI because of other complications, which in our series was largely due to obstructive causes or sepsis. The other two elective procedures in which AKI was observed with greatest frequency were TURBT and cystectomy. As with any major surgery, the latter is recognised to have a significant complication rate (
13) but it is not possible to determine from our results whether AKI associated with TURBT reflects the larger number of cases performed or whether there is an increased risk of AKI specific to this procedure.
Overall mortality rates were lower than seen in studies of other AKI populations (
1,
2,
14). This may reflect a greater proportion of elective patients (which included those undergoing nephrectomy), which is in contrast to general hospitalised AKI patients in whom over 90% are admitted as an emergency and AKI occurs as part of acute illness (
15). In addition, urinary obstruction was a common underlying cause in which AKI may be more easily reversible by radiological or surgical drainage (
9). Despite this, it is important to note that mortality progressively increased with AKI stage. The presence and severity of AKI remains a marker of the ‘unwell patient’ who requires additional clinical attention.
Two co-morbid conditions had appeared to have an influence on outcomes. Firstly, we observed an association between CKD and mortality, which is particularly important as CKD is a strong risk factor for developing AKI in this first instance. Secondly, there was also a strong link between malignancy and mortality, which was present in two-thirds of non-survivors and accounted for all but one of the deaths in the elective group. These results are consistent with other reports in which a malignant cause of obstructive uropathy conferred a significantly worse prognosis (
9). In contrast to historical data, more recent studies in general AKI populations suggest that malignancy is now the third most common cause of death (
15). In part, this may reflect that a significant proportion of patients are admitted to hospital to receive end of life care (
16) and in some AKI may occur as part of the terminal phase of their illness.
The low rate of renal recovery by time of hospital discharge may in part reflect the inclusion of nephrectomy patients. It is important to note that AKI occurring after partial nephrectomy increases the likelihood of developing subsequent CKD (
17), which in turn contributes to increased cardiovascular risk (
18-
20). However, renal surgery was not the only explanation for non-recovery by time of hospital discharge. This observation is consistent with the burgeoning evidence that links episodes of AKI to the onset and progression of CKD. Although our follow up time was short, it would appear that this paradigm may also apply to urological patients (
4).
There are some weaknesses to our study, in particular its observational nature precludes the determination of causality of the reported associations. Patients’ co-morbidity was derived from hospital coding data which may introduce some inaccuracies.
Finally, our methodology prevented the inclusion of a control group without AKI. We are also aware that the cohort of patients is probably too heterogeneous to allow for definite conclusions from simple parametric and non-parametric statistical testing. However, although this was a heterogeneous group, and although the conclusions are to be expected, these have not previously been described and they contribute to raise awareness of AKI in Urology.
Conclusion: This study is the first to describe incidence, aetiology and outcome of AKI occurring in elective and emergency urology admissions. It is clear that the underlying aetiology of AKI must be taken into consideration when assessing the potential impact of AKI but that the presence of AKI remains a marker of the unwell patient. Specific strategies to target improvements in renal recovery following AKI may be particularly pertinent in this patient group.