1. Introduction
Diabetic nephropathy is the leading cause of end-stage renal disease in developed countries (1). It is also a major risk factor for development of cardiovascular diseases (2). In patients with diabetic nephropathy, appropriate management of blood glucose level is important to delay the progression to end-stage renal disease and prevent the development of cardiovascular disease (3, 4). Although many kinds of hypoglycemic agents are currently available for the management of blood glucose level, most agents cannot be used in patients with advanced-stage diabetic nephropathy because decreased renal function delays their elimination from the body, leading to adverse effects.
Glucagon-like peptide-1 (GLP-1) receptor agonists are analogs of human GLP-1 that have a high homology to the endogenous human GLP-1 (5). GLP-1 receptor agonists enhance glucose-dependent insulin secretion from the pancreatic beta cell, and suppress inappropriate glucagon secretion leading to decreased blood glucose levels (6). They also delay gastric emptying by suppressing antro-duodenal motility and stimulating pyloric contractions, leading to improved postprandial hyperglycemia (7). Dulaglutide, a long-acting GLP-1 receptor agonist, can be used by once-weekly administration in patients with diabetes mellitus and impaired renal function without any dose adjustment (8-10). Dulaglutide showed better glucose control compared to liraglutide, a once-daily GLP-1 receptor agonist, in patients with type 2 diabetes mellitus and normal renal function (11). Dulaglutide has also been reported to show nephroprotective effects in patients with type 2 diabetes mellitus with mild renal impairment (12). However, the efficacy of dulaglutide for glycemic control and nephroprotection in patients with advanced-stage diabetic nephropathy has not yet been reported. We aimed to investigate retrospectively the efficacy of once-weekly dulaglutide for glycemic control and nephroprotection compared to once-daily liraglutide in patients with advanced-stage diabetic nephropathy.
2. Methods
2.1. Ethical Considerations
This study was performed in accordance with the ethical principles contained in the declaration of Helsinki and was approved by the ethics committee of chiba cerebral and cardiovascular center. Written informed consent was obtained from all patients.
2.2. Patients
We collected and analyzed data from study patients between 2012 and 2016. Inclusion criteria included the followings: patients aged older than 20 years, diagnosed with type 2 diabetic nephropathy with an estimated glomerular filtration rate (eGFR) ≤ 60 (mL/min/1.73 m2), urine protein to creatinine ratio (UACR) ≥ 0.5 (g/gCr), and taking liraglutide for more than 24 months or dulaglutide more than 12 months after taking liraglutide for more than 12 months. Exclusion criteria included the followings: patients diagnosed with type 1 diabetes mellitus or secondary diabetes mellitus, patients with malignancy, severe infection, steroid therapy, and renal replacement therapies such as hemodialysis, peritoneal dialysis, and renal transplantation.
2.3. Study Protocol
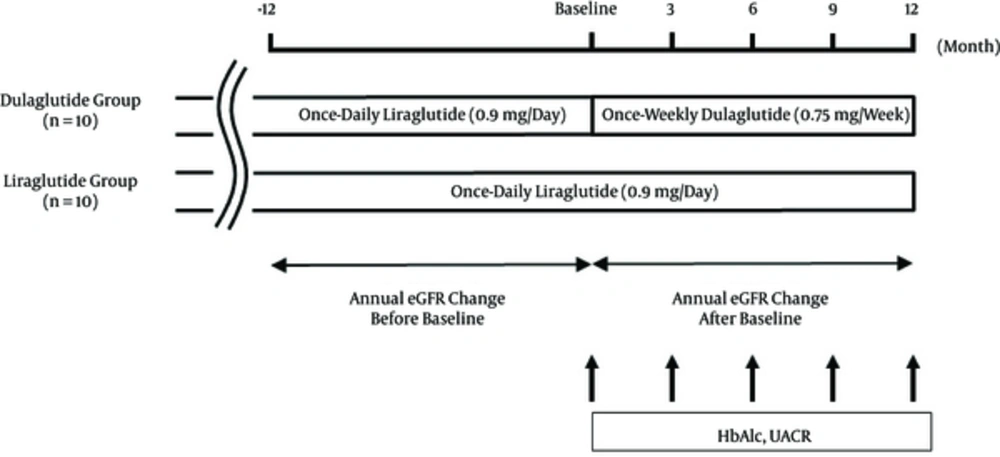
Figure 1 shows the diagram of the study design. The study is a retrospective comparative study involving 20 patients. We categorized 10 of the patients who had previously received once-weekly dulaglutide (0.75 mg/week) for more than 12 months after once-daily liraglutide (0.9 mg/day) for more than 12 months to the dulaglutide group. We allocated the other 10 patients to the liraglutide group, which consisted of patients previously treated with once-daily liraglutide (0.9 mg/day) for more than 24 months. The baselines of each patient in the dulaglutide group were set up on the days in which liraglutide was changed to dulaglutide between September 1, 2015 and November 30, 2015. In the liraglutide group, baselines were also set up during the same period on the days when patients visited hospital. Dulaglutide was subcutaneously injected once weekly on the same day of the week. Liraglutide was subcutaneously injected once daily at the same time each day. Changes in the clinical data levels of glycosylated hemoglobin (HbA1c) and UACR at 3, 6, 9, and 12 months were compared with those at baseline in each group. The annual changes in eGFR (mL/min/1.73 m2/year) were evaluated 12 months before and after the baseline in each group.
2.4. Laboratory Methods
The eGFR was calculated using a modified version of the Modification of Diet in Renal Disease formula of the Japanese society of nephrology: eGFR (mL/min/1.73 m2) = 194 × age-0.287 × serum creatinine−1.094 (multiplied by 0.739 for females) (13). The annual changes in eGFR were determined by linear regression analysis as the slope per month before and after baseline in each group. Blood and urinary parameters were determined by the Department of Clinical Laboratory, Chiba Cerebral, and Cardiovascular Center.
2.5. Statistical Analysis
Statistical analysis was performed using JMP 11 (SAS Institute Inc., Cary, NC, USA). Data were expressed as means ± standard deviation. Comparisons of component ratios were performed using Fisher's exact test. Comparisons between the two groups were performed using Mann–Whitney U test. Comparisons of clinical parameters within each group were performed using repeated-measures analysis of variance with Tukey’s test. The comparisons of the annual change in eGFR before and after baseline in each group were performed using paired t-test. Differences with P < 0.05 were considered statistically significant.
3. Results
Twenty patients were analyzed (11 males, 9 females, mean age of 68.8 ± 10.6 years). No patient was initiated renal replacement therapies during study period. Ten patients were categorized to the dulaglutide group and 10 to the liraglutide group. Table 1 summarizes baseline characteristics and other hypoglycemic agents used by the patients in each group. Most of the clinical data were similar between the two groups except for high-density lipoprotein cholesterol and triglyceride (Table 1). During the study period, the dosage of other drugs including other hypoglycemic and antihypertensive agents did not change.
| Dulaglutide Group (n = 10) | Liraglutide Group (n = 10) | P Value | |
|---|---|---|---|
| Age, y | 67.6 ± 9.9 | 69.9 ± 11.6 | 0.73 |
| Sex (male/female) | 6/4 | 5/5 | 1.00 |
| Body weight, kg | 65.4 ± 9.7 | 75.1 ± 24.2 | 0.34 |
| Body mass index, kg/m2 | 25.7 ± 3.2 | 28.9± 8.8 | 0.60 |
| Systolic blood pressure, mmHg | 136.8 ± 17.1 | 137.7 ± 13.5 | 0.97 |
| Diastolic blood pressure, mmHg | 75.6 ± 11.2 | 73.6 ± 11.5 | 0.62 |
| Serum creatinine, mg/dL | 2.8 ± 1.6 | 2.3 ± 1.3 | 0.45 |
| eGFR, mL/min/1.73 m2 | 20.3 ± 10.4 | 26.2 ± 12.0 | 0.27 |
| Urinary protein excretion, g/gCr | 2.8 ± 3.7 | 2.0 ± 2.9 | 0.82 |
| HbA1c, % | 7.2 ± 0.8 | 6.6 ± 0.9 | 0.08 |
| LDL-cholesterol, mg/dL | 81.4 ± 35.2 | 89.0 ± 32.3 | 0.34 |
| HDL-cholesterol, mg/dL | 38.4 ± 16.6 | 50.4 ± 14.4 | *0.02 |
| Triglyceride, mg/dL | 241.9 ± 96.9 | 141.1 ± 36.1 | *0.1 |
| Uric acid, mg/dL | 6.3 ± 0.8 | 5.9 ± 1.2 | 0.65 |
| Albumin, g/dL | 4.0 ± 0.5 | 3.7 ± 0.6 | 0.52 |
| Hemoglobin, g/dL | 11.6 ± 1.1 | 11.7 ± 1.1 | 0.97 |
| Brain natriuretic peptide, pg/mL | 71.6 ± 147.4 | 59.1 ± 42.8 | 0.17 |
| Estimated salt intake, g/d | 10.3 ± 1.9 | 10.4 ± 3.1 | 0.91 |
| Antihyperglycemic drugs | 8 (80) | 5 (50) | 0.35 |
| Insulin agents | 2 0) | 5 (50) | 0.35 |
| Antihypertensive drugs | 9 (90) | 0 (100) | 1.00 |
| RAS inhibitors | 7 (70) | 8 (80) | 1.00 |
| Diuretics | 3 (30) | 1 (10) | 0.58 |
| Antihyperlipidemic drugs | 8 (80) | 8 (80) | 1.00 |
| Statins | 7 (70) | 6 (60) | 1.00 |
| Antihyperuricemic drugs | 4 (40) | 6 (60) | 0.66 |
Abbreviations: eGFR, estimated glomerular filtration rate; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; RAS, renin–angiotensin system.
3.1. Effects of Once-Weekly Dulaglutide on Glycemic Control
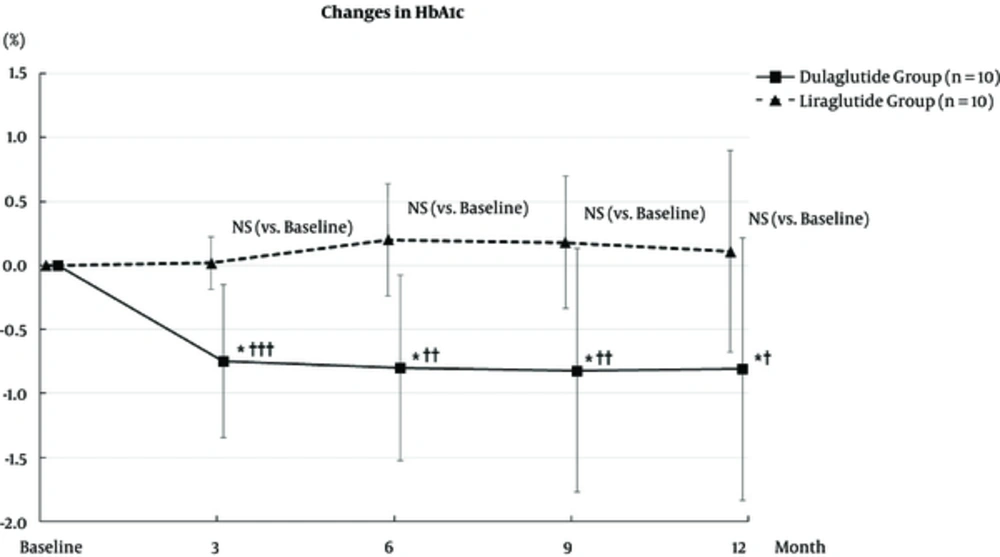
HbA1c levels of the dulaglutide group significantly decreased from 7.21 ± 0.84% at baseline to 6.46 ± 0.58% at 3 months (P < 0.05), 6.41 ± 0.53% at 6 months (P < 0.05), 6.39 ± 0.49% at 9 months (P < 0.05), and 6.40 ± 0.57% at 12 months (P < 0.05). HbA1c levels of liraglutide group at 3, 6, 9, and 12 months did not change compared to the baseline. The levels of HbA1c at 3, 6, 9, and 12 months from the baseline in the dulaglutide group decreased more significantly than those in the liraglutide group (Figure 2).
3.2. Effects of Once-Weekly Dulaglutide on UACR and Annual Change in eGFR
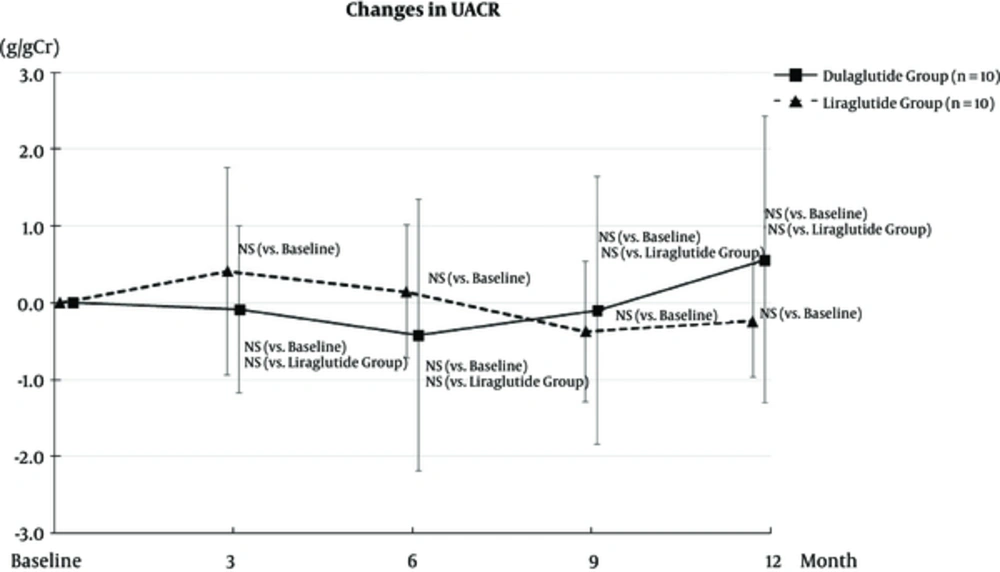
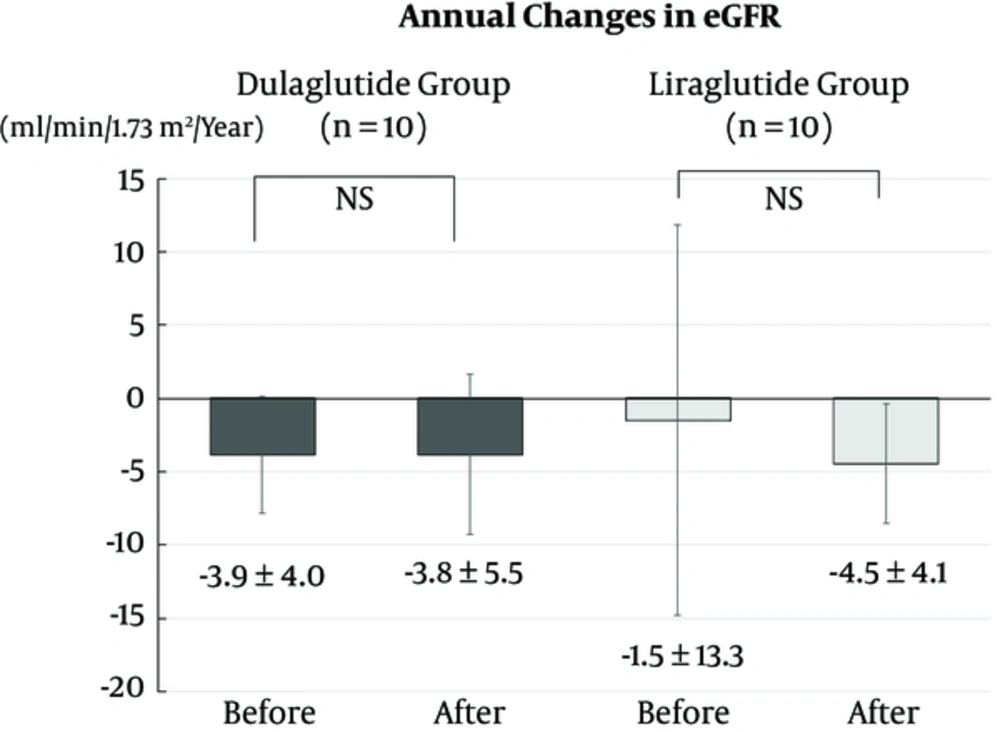
UACR at 3, 6, 9, and 12 months did not change from the baseline in both dulaglutide and liraglutide groups (Figure 3). The annual change in eGFR did not differ from before and after baseline levels in both the dulaglutide and liraglutide groups. In addition, the annual change in eGFR after the baseline did not differ between the groups (Figure 4).
3.3. Changes in Other Clinical Parameters
Other clinical and laboratory parameters, including body weight, systolic and diastolic blood pressure, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, uric acid, albumin, and hemoglobin, did not significantly change at 3, 6, 9, and 12 months compared to the baseline levels in both dulaglutide and liraglutide groups (data not shown).
3.4. Adverse Events
No adverse events, including hypoglycemia, severe hyperglycemia, decreased appetite, nausea, diarrhea, severe constipation, or acute pancreatitis, were observed in either the dulaglutide or liraglutide groups during the study period.
4. Discussion
We found that once-weekly dulaglutide decreased HbA1c levels in patients with advanced-stage diabetic nephropathy after changing from once-daily liraglutide. However, once-weekly dulaglutide did not affect UACR or the annual change of eGFR, nor did it show any adverse effects on the patients.
Dulaglutide is a long-acting GLP-1 receptor agonist with a half-life of approximately 5 days and has a recommended dosage of once-weekly (8-10). Randomized phase II and III clinical studies have reported that once-weekly dulaglutide shows no differences in terms of safety and effectiveness between patients with normal renal function and patients with decreased renal function (10). Hence, it requires no dose adjustment for use in patients with renal impairment. Furthermore, a phase III clinical study reported that once-weekly dulaglutide significantly reduced HbA1c levels in type 2 diabetes mellitus patients with normal renal function compared to once-daily liraglutide after 52 weeks (11). In our study, once-weekly dulaglutide significantly reduced HbA1c levels with no adverse effects in patients with advanced-stage diabetic nephropathy after changing from once-daily liraglutide. These results suggest that once-weekly dulaglutide is more advantageous for glycemic control compared to once-daily liraglutide, and it can be used safely in patients with advanced-stage diabetic nephropathy.
Studies have reported that GLP-1 receptor agonists have nephroprotective effects, which are independent of the glucose-lowering effects (14, 15). Liraglutide has been reported to decrease proteinuria and attenuate the progression of renal dysfunction (16-18). Recently, integrated data from phase II and III trials showed that dulaglutide also reduced urinary albumin excretion in patients with type 2 diabetes and mild renal impairment (12). In our study, we did not observe these nephroprotective effects after changing liraglutide to dulaglutide in patients with advanced-stage diabetic nephropathy. These results suggest that dulaglutide may not have superior effects on nephroprotection compared to liraglutide in patients with advanced-stage diabetic nephropathy. Large scale and long-term clinical studies investigating the nephroprotective effects of dulaglutide on diabetic nephropathy are required.
There are several limitations in our study. First, this is a retrospective observational study, which may be subjected to substantial selection bias. Second, this study is based on a small number of patients from a single center, which limits the possibility of generalizing our findings. Therefore, large-scale, double-blind trials are required to confirm the efficacy of dulaglutide on glycemic control as well as on nephroprotection in patients with advanced-stage diabetic nephropathy.
In conclusion, once-weekly dulaglutide improved glycemic control without showing any adverse effects in patients with advanced-stage diabetic nephropathy after changing from once-daily liraglutide. These results suggest that once-weekly dulaglutide is more advantageous for glycemic control compared to once-daily liraglutide and it could be used safely in patients with advanced-stage diabetic nephropathy.