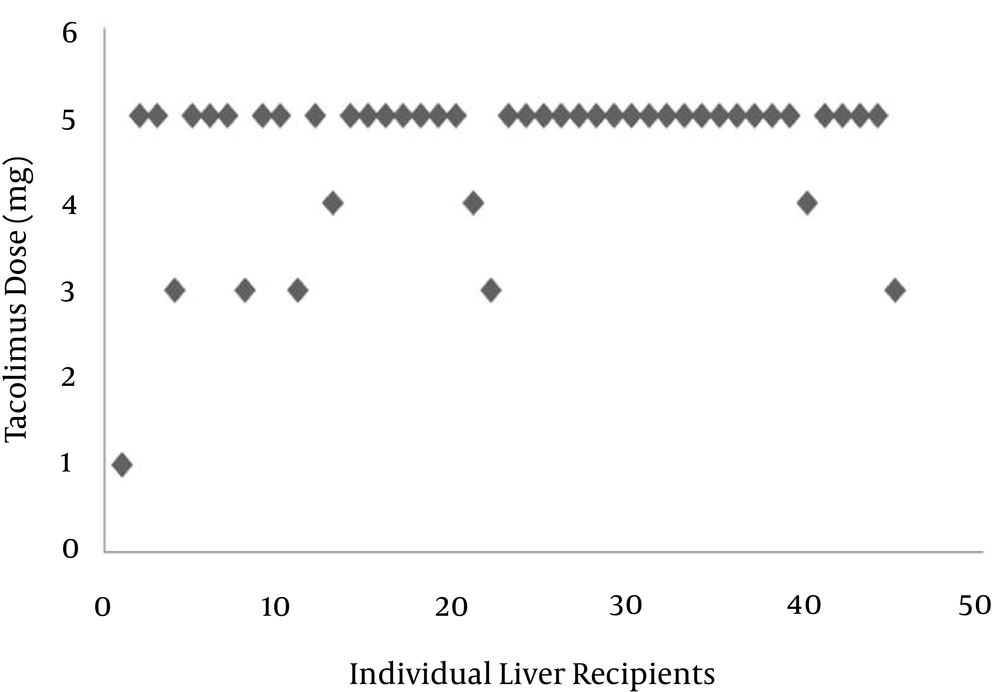
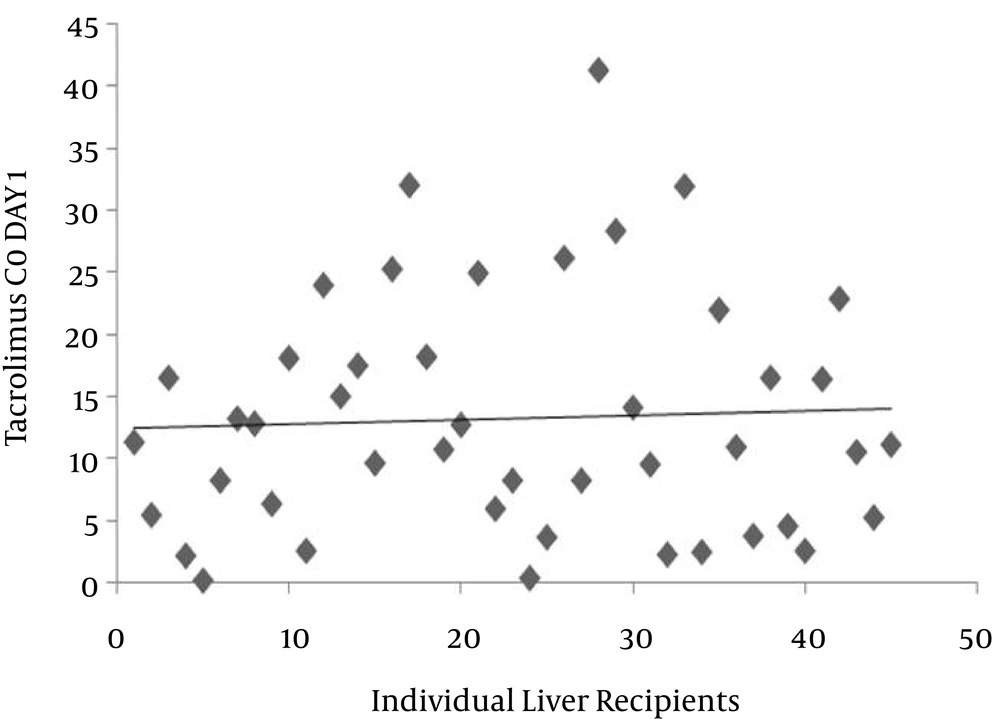
The distribution of immunosuppressive trough levels in the population studied was highly variable. As shown in
Figure 1, the mean dosage of the drug was 4.6 mg (range: 1 - 5 mg). Out of 45 patients, the doses of tacrolimus in 80% were 5 mg. Because the management of tacrolimus was connected with marked unpredictability and a large disparity in C0, thus, the efficiency and neurotoxicity seem to be associated with blood levels. As shown in
Figure 2, the mean C0 of tacrolimus was 13.2 mg/L, with a minimum value of 0.1 and a maximum value of 41.4 mg/L. Trough levels of tacrolimus in 20.4% of patients was lower than the suggested therapeutic level. In 35% of the patients studied, the level was more than 15 mg/L (neurotoxic border). There was no correlation between dose and trough levels (P = 0.270, r = 0.168). The pattern of pharmacotherapy for selected patients is described as follows (
Box 1): A twenty-four-year-old male with code 45, due to autoimmune hepatitis, received prednisone, and azathioprine eight years prior to liver transplantation. After surgery, the patient presented with a herpetic lesion on his lip that had been treated with intravenous acyclovir followed by the oral form. The drug regimen after transplantation was as follows: Prograf 3 mg every 12 hours, tablet; CellCept 1 gm every 12 hours, capsule; fluconazole 100 mg every 12 hours, tablet; co-trimoxazole 1 g daily, tablet; folic acid 5 mg 1 daily, tablet; prednisone 20 mg every morning, tablet; pantoprazole 40 mg every 12 hours, tablet; calcium D every 8 hours, tablet; and acyclovir every 12 hours. Tacrolimus C0 was reported as 6 mg/mL. In another twenty-eight-year-old male transplanted due to primary sclerosing colangitis (PSC) with code 89, the drug regimen was as follows: amp cefitizidime 1 gm; metronidazol 500 gm every 6 hours; pantoprazole 40 gm every 12 hours; methylprednisolone 1 gm a day after transplant for two days; heparin 10000 iu for 24 hours; and mesalasin-albumin 1 vial. Evidence of acute rejection was noted in the patient’s medical record, and then the patient was treated with 3 gm methylprednisolone. The patient was discharged with the following drug regimen: tacrolimus 7 mg; CellCept 2 gm; and prednisone 20 mg. Tacrolimus C0 was reported as 5 mg/mL. Another patient, with code 68, underwent liver transplantation due to Wilson’s disease. After the operation, the patient showed psychosis and episodes of rejection. The drug regimen was as follows: Prograf 4 mg every 12 hours; CellCept 2 gm every 12 hours; and prednisone 20 mg every 12 hours.